Abstract
Cationic surfactants (CSs) are surface-active compounds containing a positively charged polar group and at least one alkyl chain as a nonpolar group. Due to their structure, they tend to adsorb on negatively charged surfaces and interact with biopolyanions. It leads to their wide use as disinfectants, cleaning agents, fabric softeners, hair care products, emulsifiers, corrosion inhibitors, etc. Considering their extensive use and also their toxicity, fast, simple, and accurate CSs determination is crucial in industrial process control, product quality assurance, and environmental monitoring. Potentiometric sensors meet all these requirements, so they stand as the primary method for CSs determination. In this review, numerous potentiometric methods for CSs determination have been described, with a particular focus on methods published in the period from 2000 to 2024. Due to their simplicity and good analytical performance, solid-state electrodes are the most commonly used type of sensor for CSs determination.
Graphical abstract
Abbreviations
- AChE
-
acetylcholinesterase
- APAS
-
alkylpyridinium-alkylsulfates
- APTPB
-
alkylpyridiniumtetraphenylborates
- AS
-
anionic surfactant
- BDDA
-
benzyldimethyldodecylammonium
- BDHA
-
benzyldimethylhexadecylammonium
- BDHAC
-
benzyldimethylhexadecyl ammonium chloride
- BDTA
-
benzyldimethyltetradecylammonium
- CMA
-
cetyltrimethylammonium
- CMC
-
critical micellar concentration
- CP
-
cetylpyridinium
- CPC
-
cetylpyridinium chloride
- CP-FTC
-
cetylpyridinium ferric thiocyanate
- CP-IM
-
cetylpyridinium iodomercurate
- CP-SnP
-
cetylpyridinium based Sn(iv) phosphate
- CS
-
cationic surfactant
- CTA
-
hexadecyltrimethylammonium
- CTAB
-
hexadecyltrimethylammonium bromide
- DBP
-
dibutyl phthalate
- DDAC
-
N,N-didecyl-N,N-dimethylammonium chloride
- DDS
-
dodecyl sulfate
- DHBI–Br
-
1,3-dihexadecyl-1H-benzo[d]imidazol-3-ium bromide
- DHDA
-
dodecyl(2-hydroxyethyl)dimethylammonium
- DMI
-
1,3-didecyl-2-methylimidazolium
- DMIC
-
1,3-didecyl-2-methylimidazolium chloride
- DMTB
-
1-dodecyl-5-methyl-1H-benzo[d][1,2,3]triazol-1-ium bromide
- DNSA
-
dinonylnaphthalene sulfonic acid
- DOAC
-
N,N-dioctyl-N,N-dimethylammonium chloride
- DODI
-
1,3-dioctadecyl-1H-imidazol-3-ium
- DOP
-
bis(2-ethylhexyl) phthalate
- DTA
-
dodecyltrimethylammonium
- EO
-
etoxylated
- FET
-
field-effect transistor
- f-PNPE
-
2-fluorophenyl 2-nitrophenyl ether
- HCR
-
high concentration range
- HDP
-
hexadecylpyridinium
- Hyamine
-
benzethonium chloride
- ISE
-
ion selective electrode
- ISFET
-
ion-sensitive field-effect transistor
- KTCPB
-
potassium tetrakis(4-chlorophenyl)borate
- LCR
-
low concentration range
- LOD
-
limit of detection
- MPB
-
1,4-bis-(8-mercaptooctyloxy)-benzene
- MWCNT
-
multi-walled carbon nanotube
- MWCNT-OSO3 −CP+
-
multi-walled carbon nanotubes functionalized with a sulfate group and cetylpyridinium
- NaTPB
-
sodium tetraphenylborate
- NPDOE
-
o-nitrophenyl dodecyl ether
- NS
-
nonionic surfactant
- NTAB
-
nonyltrimethylammonium bromide
- NTO
-
N,N,N′N′-tetracyclohexyl-3-oxapentanediamide
- o-NPOE
-
o-nitrophenyl octyl ether
- PAMPS
-
poly-2-acrylamido-2-methylpropanesulfonic acid
- PDMS
-
polydimethylsiloxane
- PTFE
-
polytetrafluoroethylene
- PVB
-
poly(vinyl butyral)
- PVC
-
polyvinyl chloride
- s-BT
-
s-benzylthiuronium
- Septonex
-
1-(ethoxycarbonyl)pentadecyltrimethylammonium bromide
- SWCNT
-
single-walled carbon nanotube
- TCP
-
tricresyl phosphate
- TFPB
-
tetrakis[3,5-bis(trifluoromethyl)phenyl]borate
- THA
-
tetrahexadecylammonium
- THF
-
tetrahydrofuran
- TPB
-
tetraphenylborate
- TTA
-
tetradecyltrimethylammonium
- WWTP
-
wastewater treatment plants
- Zephiramine
-
benzyldimethyltetradecylammonium
- α-CD
-
α-cyclodextrin
1 Introduction
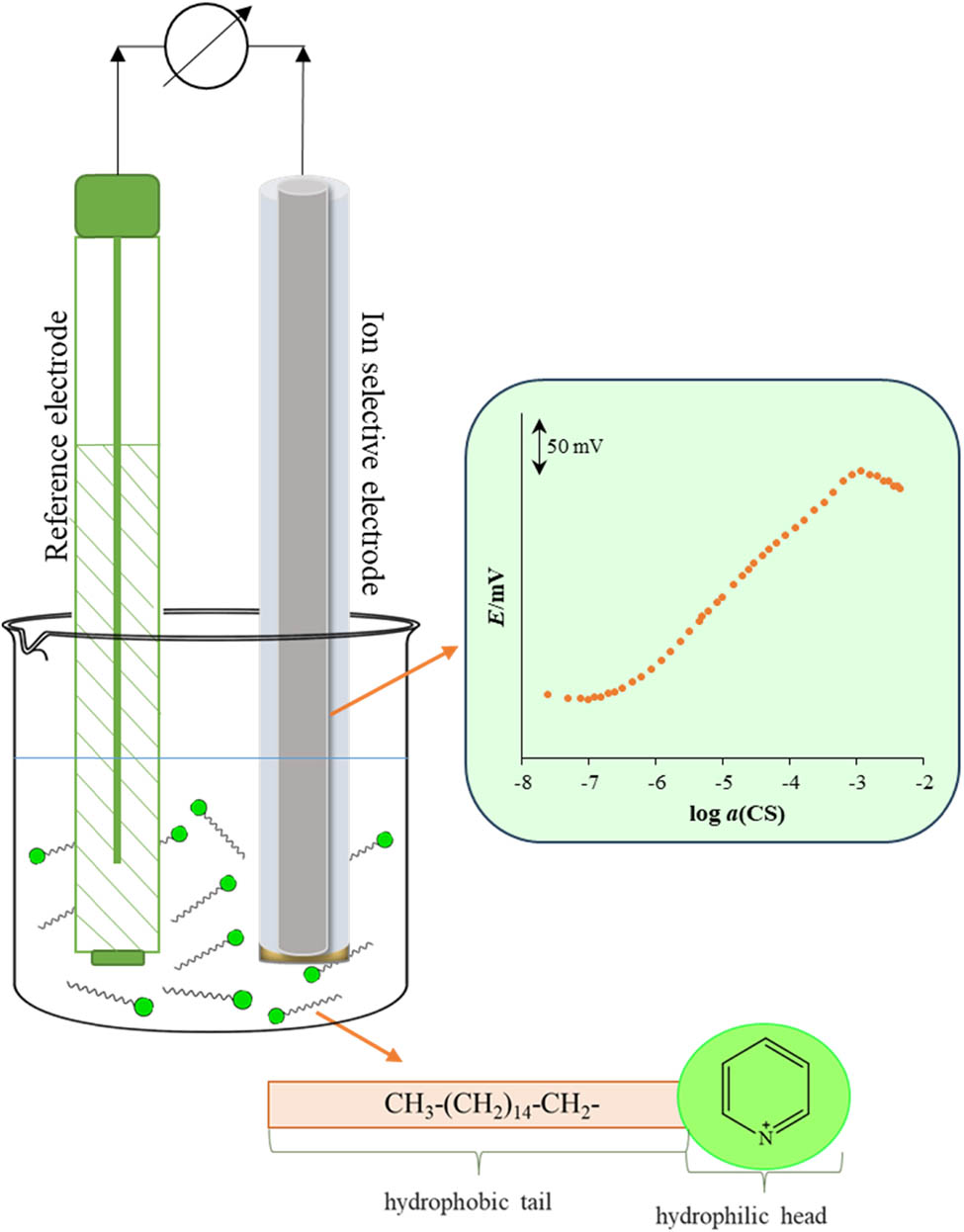
Surfactants are compounds containing a nonpolar hydrophobic group (tail) and polar hydrophilic group (head) in their molecular structure. Due to their dual nature, they can adsorb at the phase boundary between two immiscible phases and reduce surface tension, and they have a high tendency to self-assembly (Figure 1). These two properties are the main reasons for their wide application. Surfactants can be classified as cationic, anionic, nonionic, or amphoteric, according to the charge of hydrophilic groups in their molecules. Considering the increasing population growth and consequently increasing environmental and technological requirements, production and consumption of surfactants have been consistently growing [1]. They are widely used in industry, personal care products, and house hold products due to their properties. In 2022, the global surfactant market is estimated at USD 43.5 billion, at a compound annual growth rate of 4.9% [2].

Visualization of surfactants dual nature, adsorption, and self-assembly to micelles and its relation to the possibility of their potentiometric determination.
Cationic surfactants (CSs) are a class of surfactants with a positively charged polar group. In most CSs, that is the quaternary ammonium group. As hydrophobic group, they contain at least one alkyl chain. Because most solid surfaces carry a negative charge, the polar group of CSs exhibits a tendency to adsorb on these surfaces through electrostatic attraction [3]. Also, due to their polar group, CSs have high affinity toward biopolyanions such as nucleic acids and certain proteins [4].
Although they only account for approximately 5% of the total surfactant market, they are very important and widely used as cleaning agents, fabric softeners, hair care products, emulsifiers in road construction, corrosion inhibitors, etc. [3]. Also, they have the ability to interact with negatively charged phospholipids in the cell membrane of microorganisms which leads to inhibition or denaturation of membrane proteins. This property is responsible for their extensive use as disinfectants, bactericides, and fungicides, their effectiveness against enveloped viruses and tumor cells, and for their toxicity [5,6]. During the coronavirus disease (COVID-19) period, CSs were extensively used due to their antiviral activity. The spike protein (S) found on the lipid membrane of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is an essential glycoprotein within the membrane of the virus that facilitates its entry into host cells. Binding to its receptor on a host cell is followed by conformational alterations, leading to the fusion of the viral and cellular membranes. Virions with disrupted lipid membranes are unable to infiltrate cells and initiate infection. Among different CSs, those with increased length and number of hydrophobic tails and a benzene ring in their structure have enhanced antiviral activity [7]. Also, CSs exhibit lysosomotropic action which ultimately leads to blocking virus entry [8].
Some nonconventional applications of CSs include the use of cleavable surfactants as microbicides [9]. Their positively charged polar heads interact with the negatively charged bacterial membrane, causing disruption of the bacterial cell membrane through a combination of electrostatic and hydrophobic interactions, which leads to the leakage of cytoplasmic constituents and consequently to cell death. Due to the higher percentage of negative charge on bacterial cell membranes compared to mammalian cells, the positive charge of CSs enables enhanced interaction with the bacterial membrane. The main advantage of cleavable surfactants compared to non-cleavable ones is that they contain weak linkage between their polar and non-polar groups, which results in improved biodegradability [10]. Also, CSs can interact with nucleic acids and be used as non-viral delivery systems for DNA and RNA molecules into the cell nucleus, which can be used in gene therapy for diseases caused by defective genes or mutations, such as cancer [11]. Gene therapy is based on the repair or replacement of damaged or absent genes by new DNA in the cell nucleus. It can be done using viral or non-viral delivery systems (vectors). The first step in gene (DNA) delivery using non-viral vectors, such as CSs, is DNA-vector complexation and the formation of the lipoplex. It is formed due to electrostatic interactions between the positively charged polar groups in CSs molecules and the negatively charged phosphate backbone of DNA. It is followed by the binding of the formed complex to the cell plasma membrane, entering the cell by endocytosis, traveling through the cytoplasm, and entering the nucleus. Once inside the nucleus, the DNA is released from the DNA-vector complex and can reach the transcription machinery, resulting in its expression as a protein and the therapeutic effect [12]. Another use of CSs includes them as templates for the synthesis of mesoporous materials, where the choice of surfactants and reaction conditions directly determine the material pore size. Usually CSs and silica interact in order to get a composite material, which is followed by the removal of CSs using extraction or calcination in order to get mesoporous silica [13]. The resulting mesoporous materials can be widely used in optical devices, as catalysts, drug carriers, etc.
However, the toxicity of CSs is considered to be the main limiting factor for their wider use. It is mainly manifested as eye or skin irritation. Compared to other classes of surfactants, CSs exhibit higher aquatic toxicity and cause more skin and eye irritations [6]. It can be explained by the overall negative charge of most skin proteins at physiological pH, which causes stronger interactions with CSs [14]. Also, if consumed in significant amounts, they can affect the human central nervous system inducing agitation, weakness, lack of coordination, and decreased consciousness [15]. It is believed that individual CSs molecules (monomers) cause more toxic effects compared with those aggregated into micelles. Consequently, CSs with a higher value of critical micellar concentration (CMC) are more toxic [14].
CSs and their degradation products are continuously released into the environment as effluents from wastewater treatment plants (WWTP), through sewage sludge applied on land, and through industrial discharging into surface water. Due to their amphiphilic nature and affinity to adsorb on the surface of particulate matter, CSs pass through WWTP with limited treatment [16]. They are biodegradable, but only under aerobic conditions. Their anaerobic non-biodegradability can be explained by CSs toxicity toward relevant anaerobic microorganisms [16]. The biodegradation time of CSs and their aquatic toxicity are directly proportional to the length of the hydrophobic group in the CSs molecule [3]. Furthermore, the introduction of a benzyl group in the CSs molecule reduces the rate of biodegradation [17]. Once they enter the environment, CSs can affect organisms such as bacteria, algae, invertebrates, and fish, which leads to disruption of the natural balance of ecosystems [18]. Therefore, the emphasis is on synthesis of ecologically friendly CSs with low toxicity and a high biodegradation rate.
Considering the wide consumption of CSs, their monitoring is essential for the control of industrial processes as well as for the quality control of products based on CSs. Also, due to their high toxicity, it is essential to monitor CSs concentration in the environment. For that purpose, two-phase titration [19] is used as a reference method. This method is based on the formation of ionic pair of anionic surfactant (AS) and pink cationic dye, and its extraction in organic solvent. It is followed by titration using CS as a titrant. As the titration proceeds, cationic titrant forms ionic pair with AS, and cationic dye is displaced into the water layer due to its insolubility in organic solvent. The faint grayish blue organic solvent indicates complete replacement of cationic dye with CSs and the end point of titration. Although two-phase titration has been used for many years as a reference method for CSs determination, this method has many drawbacks, such as a high level of subjectivity, difficult CSs determination in nontransparent samples, a tedious procedure, and the use of large volumes of chloroform as an organic solvent, which leads to a negative impact on the environment and increased expenditure on hazardous waste disposal. Therefore, it is necessary to have a reliable, accurate, simple, and low-cost analytical method for the determination of CSs in various samples. Also, the method must be sensitive and selective, in order to be used for CSs determination in real complex samples with low concentrations of CSs and where CSs are combined with nonionic surfactants (NSs) and other substances.
CSs can be determined using different methods such as spectrophotometry [20,21,22] and chromatography [23,24,25]. However, both of these methods involve the use of organic solvents, expensive devices, and time-consuming sample preparations and procedures. Therefore today, the most commonly used method for CSs determination is potentiometry. Using ion selective electrodes (ISEs) as sensors for potentiometric measurements presents an excellent alternative to all the abovementioned methods due to their simplicity, accuracy, sensitivity, short response time, low cost and portable instrumentation, low reagents consumption, non-use of toxic solvents, no need for time-consuming and complex sample preparation, etc.
This study presents a review of potentiometric methods for CSs determination. There are several review papers on the topic of CSs, but they are mainly based on the properties of CSs [3,26,27], their synthesis [28], or their adsorption [29,30,31]. To the best of our knowledge, there are no reviews on potentiometric methods for CSs determination. The research for this review relied on Scopus, Web of Science, and PubMed databases. The review focuses on methods published in the period from 2000 to 2024.
2 Fundamental concepts for potentiometric determination of CSs
Potentiometry is a frequently used electroanalytical method based on measuring potential within the system without or with negligible current flow. Potentiometric determination can be based on direct potentiometry and potentiometric titration. Direct potentiometry implies measurements of the potential in solution of an unknown concentration of the analyte, followed by the determination of the analyte concentration based on a calibration graph. In potentiometric titration, the potential change is measured as a function of the volume of the added titrant whose concentration is known. Then, the concentration of the analyte can be determined based on the titrant volume at the endpoint and the known stoichiometry of the chemical reaction between the analyte and the titrant.
ISEs are potentiometric sensors whose potential is proportional to the logarithm of the activity of the ion to which the electrode is selective. The potential is measured as the difference between the potentials of the ISE and the reference electrode. The part of the ISE that determines its response and selectivity is the membrane. It usually consists of a polymer matrix containing sensing material dissolved in a suitable solvent. The sensing material mainly consists of a large organic cation, usually CS, and a large organic anion, usually AS. It forms as follows:
where
where
The exchange of ions between the solution and the membrane determines the membrane potential. The potentiometric response of the ISE is based on the Nernst equation (Eq. 4)
where E is the measured electrode potential, E 0 is the standard electrode potential, and S is the slope of the sensor. The theoretical Nernstian S value is 59.2 mV per decade of ion activity at 25°C. After the insertion of Eq. 3 into Eq. 4, it follows:
From Eq. 5 it can be concluded that the
Few research groups tried to additionally explain the theory of potentiometric response. Zdrachek and Bakker [32] described ion exchange at membrane ISEs in sample solution containing competing ions. Ivanova et al. [33] proposed a model describing ISEs response and the influence of co-extraction of ions, from the sample solution and the inner electrolyte, on ISEs detection limits.
3 Potentiometric determination of CSs
Due to the disadvantages of two-phase titration described above, there was a need for the development of more convenient method for CSs determination. Potentiometry was a great alternative. Until today, it remained the most significant method for CSs determination. A chronological overview of the key milestones in the development of sensors for potentiometric determination of CSs is presented in Figure 2.

Chronological overview of significant achievements in the development of sensors for CSs determination.
Among the CSs investigated, most belong to the groups of alkyl imidazolines and quaternary ammonium compounds. The most commonly determined CSs using potentiometric measurements are cetylpyridinium chloride (CPC), 1,3-didecyl-2-methylimidazolium chloride (DMIC), hexadecyltrimethylammonium bromide (CTAB), and benzethonium chloride (Hyamine) (Figure 3).
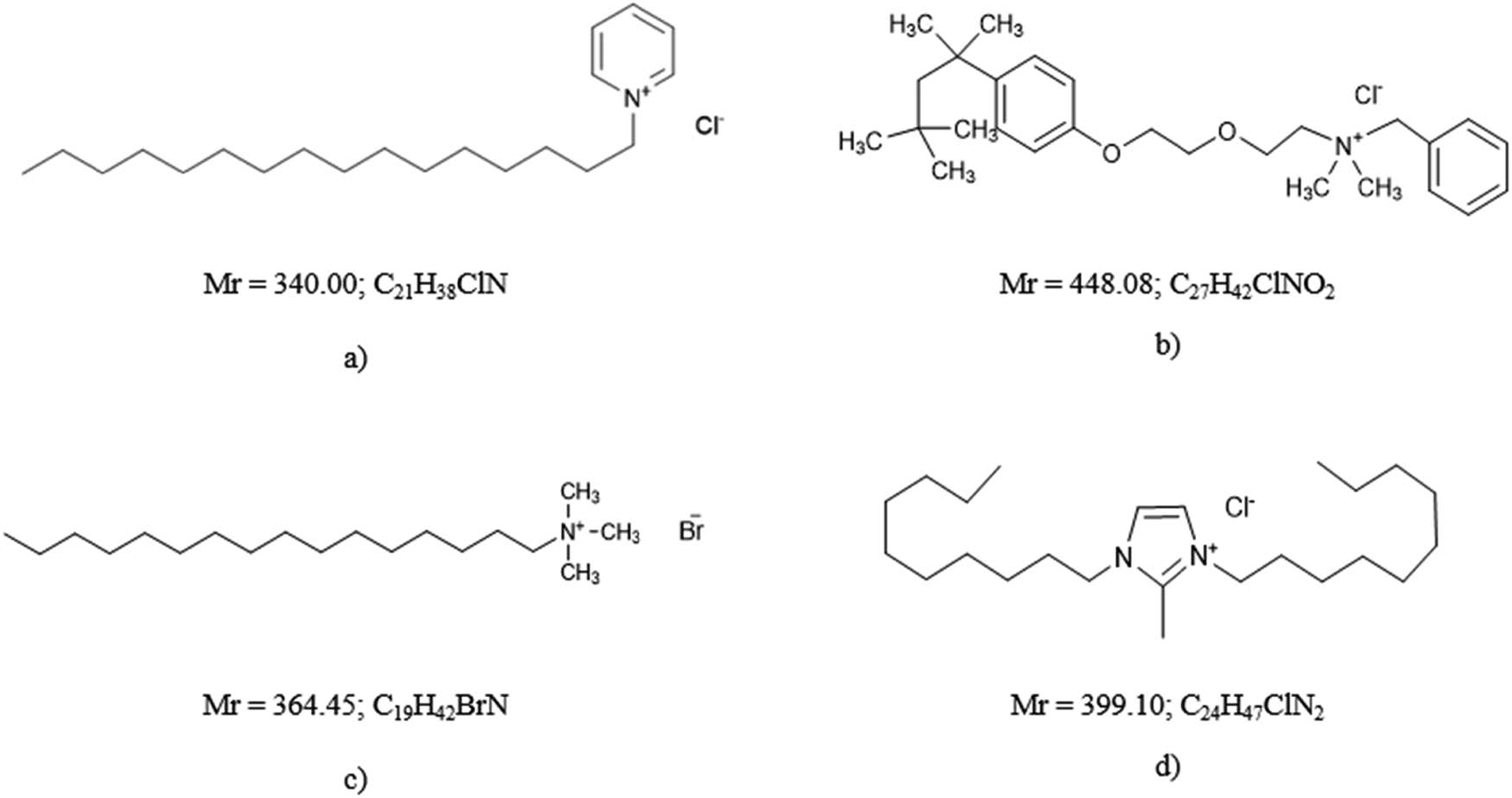
The most commonly determined CSs using potentiometric measurements: (a) CPC, (b) Hyamine, (c) CTAB, and (d) DMIC.
3.1 Conventional ISEs
The conventional ISEs for CSs determination contain a solid electrode body with an inner electrolyte and a liquid membrane (Figure 4a). The liquid membrane means that membrane components are dissolved in liquid solvent, usually tetrahydrofuran (THF), but after solvent evaporation, a thin film is obtained. Then, the membranes of the desired size can be cut out of the film and mounted onto the electrode body. The liquid type of membrane usually consists of sensor material, polymer matrix, and plasticizer. The sensor material is usually an ion pair of CS and AS, but neutral carriers, such as crow ethers, can also be used. It has the greatest influence on characteristics of the electrode, and it determines its selectivity. The membrane usually contains 1% of sensor material. The polymer matrix ensures the mechanical stability of the membrane. It should be inert. Polyvinyl chloride (PVC) is the most often used polymer matrix in ISEs for CSs determination. The plasticizer is the quantitatively dominant component of the membrane. The weight ratio of polymer to plasticizer is usually 1:2. It reduces the viscosity of the membrane and ensures the mobility of the membrane components. Due to its high content in the membrane, the plasticizer also influences the response of ISE and affects its lifetime. The most commonly used plasticizers are o-nitrophenyl octyl ether (o-NPOE) and bis(2-ethylhexyl) phthalate (DOP). Consequently, variations in membrane composition (different components and/or different ratios of components) lead to the development of ISEs with improved analytical characteristics. Therefore, many authors optimized the membrane composition of their electrodes by varying the type of plasticizer and/or content of sensor material [34,35,36].
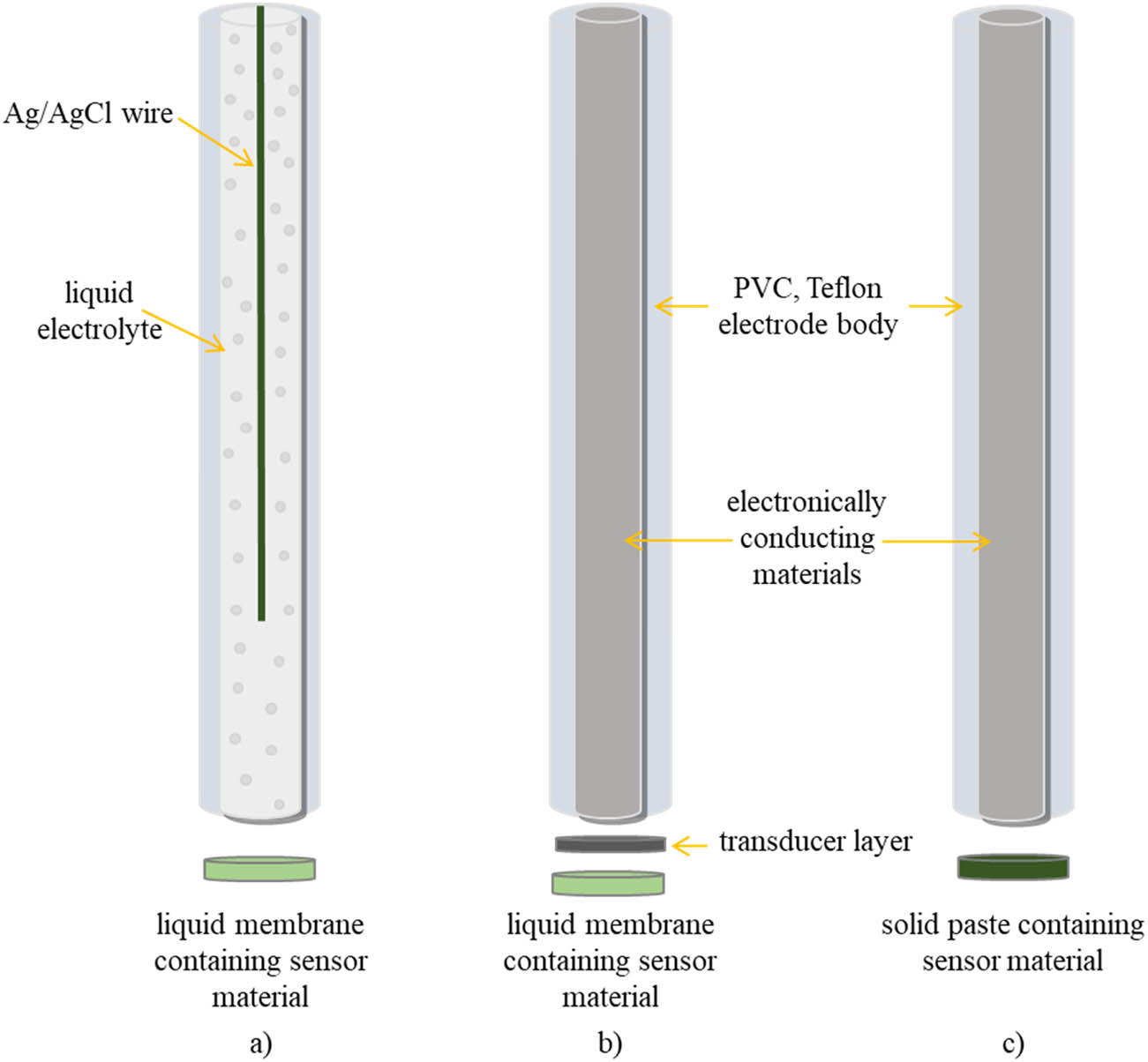
The comparison of the main electrode designs for ISEs for CSs determination: (a) the conventional ISE, (b) solid-contact ISE, and (c) all-solid-state ISE.
In 1970, Gavach and Seta [37] developed and described the first potentiometric sensor for CSs determination. It was an ISE with a liquid membrane. The sensor material in the membrane was an ion pair composed of quaternary ammonium cation and picrate or tetraphenylborate (TPB) anion. Using potentiometric titration with the described ISE as the sensor, it was possible to determine CSs at concentrations on the order of 10−6 M.
This was followed by the development of ISEs for CSs determination based on various sensing materials. These first liquid membranes often gave an unreproducible response and had a short lifetime due to the solubilization of the membrane components in an analyte solution. After Moody et al. [38] developed the calcium-selective electrode with a liquid ion exchanger in a PVC matrix, which led to a significantly prolonged lifetime of the electrode, the development of PVC-matrix electrodes began.
In 1977, Cutler et al. [39] developed electrodes for CS and AS determination with a plasticized PVC membrane based on the complex of CS and AS. The ion-exchange groups were chemically attached to the ends of the PVC chains, which led to a prolonged electrode lifetime due to reduced loss of sensor material by solubilization.
The development of ISEs with PVC membranes that were plasticized with different plasticizers followed. These electrodes showed improved stability. Also, the development of electrodes with different types of sensor materials began.
Khalil et al. [40] developed ISE for Hyamine determination with a PVC membrane, plasticized with diisooctyl phthalate. They used neutral carrier dibenzo-18-crown-6 as the new type of sensor material. The electrode with a membrane contained carrier had improved selectivity compared with the blank electrode, which contained only PVC and plasticizer.
Shoukry [41] applied a new approach for the potentiometric determination of CSs. He developed an accurate and precise potentiometric system with two ISEs, without a conventional reference electrode, for hexadecylpyridinium (HDP) bromide determination. Both ISEs contained plasticized PVC membranes with ion pairs. One was selective to the analyte (CS), and another to the titrant (AS). Instead of the usual sigmoidal titration curve, titration curves with sharp peaks or troughs were obtained. DOP was used as the plasticizer.
Baillarger et al. [42] developed organo-mineral ISE with a heterogeneous membrane for dodecyltrimethylammonium (DTA) ion determination. They used synthetic clay, laponite, as an anionic carrier of CS in PVC membrane plasticized with dinonyl phthalate.
Gerlache et al. [43] developed ISE for CS determination with a PVC membrane plasticized with o-NPOE. The sensor material was an ion pair formed of 1,3-didecyl-2-methylimidazolium (DMI) and dodecyl sulfate (DDS). The proposed ISE could be used for CSs determination using potentiometric titration or for ASs determination using direct potentiometric measurements in an on-line flow system.
Matysik et al. [44] developed two types of electrodes with a polydimethylsiloxane (PDMS) membrane filled with zeolite. One electrode was a conventional electrode with an inner electrolyte, and another was an iridium disk electrode coated with a zeolite-PDMS membrane. The application of zeolites as sensor materials was based on the pores in their crystal lattice, in which guest molecules can penetrate. The results showed that for CSs determination using potentiometric titration, the conventional type of electrode was more convenient.
Saleh [45] developed ISE for CSs determination based on PVC membrane with macrocyclic diimine crown ether as sensor material, and plasticized with o-NPOE. He studied the influence of the concentration of the inner electrolyte on the ISE response.
In 2000, Abbas et al. [46] developed ISE for cetylpyridinium cation (CP) determination. The electrode contained a PVC membrane plasticized with DOP. CP iodomercurate (CP-IM) was used as a sensor material in the membrane. The slope of the ISE was sub-Nernstian which was explained by CP existence in a dimer-like structure. The electrode was successfully applied for CP determination in mouthwash and results were in good agreement with those obtained using the British Pharmacopoeia method. In the next year, the author from the same group [47] developed another ISE for CP determination in mouthwash. This time, he used CP-FTC as sensor material, which led to a reduced limit of detection (LOD) value and a near-Nernstian slope.
In 2004, Mahajan et al. [48] developed two ISEs for two CSs determination using electrodes with PVC membranes plasticized with DOP. The ion pairs of tetradecyltrimethylammonium (TTA) and HDP with DDS were used as sensor materials. The HDP-DDS based electrode exhibited a sub-Nernstian slope. The authors investigated the influence of various additives on CMC and concluded that additives increased the CMC value. It was explained by the formation of complexes between the surfactant and the additive.
In the same year, Masadome et al. [34] investigated the influence of various plasticizers on the response of ISE without sensor material (ion-exchanger), to CSs. As plasticizers, the authors used o-NPOE and its derivatives. Considering that the analytical performance of ISEs with sensor material can be affected by the choice of plasticizer, they assumed that the use of plasticizers with higher hydrophobicity leads to enhanced performance of the ISE. However, the investigated plasticizers reduced LOD, but did not enhance other electrode characteristics. As the best plasticizer, the authors chose o-nitrophenyl dodecyl ether (NPDOE).
In 2006, Mostafa [49] developed ISE for CPC determination, based on the ion pair between s-benzylthiuronium (s-BT) and TPB. He used DOP as the plasticizer. The obtained results, for CPC determination in mouthwash, were in good agreement with those obtained using the British pharmacopoeia method.
In 2008, Madunić-Čačić et al. [50] developed ISE, based on DMI-TPB as sensor material, in a liquid membrane mounted in a commercial Philips electrode body. They used the proposed ISE for CTAB, Hyamine, and CPC determination using potentiometric titration. The authors showed that NSs, which are common in commercial products based on CSs, had negligible influence on the potentiometric titration, using the proposed ISE as an end-point detector, if the mass ratio of NSs to CSs did not exceed 5. The new electrode was successfully applied for CSs determination in disinfectants for the food industry, disinfectants for hospital use, and mouthwashes. The results exhibited satisfactory agreement with those obtained using a commercial surfactant sensor and the standard two-phase titration method. In 2011, the same research group [51] developed the method for simultaneous potentiometric determination of CSs and NSs in commercial cleaning agents and disinfection products, using potentiometric titration. The one-step titration was based on the reaction of an anionic titrant with CS, followed by its reaction with pseudocationic complex, formed from a barium cation and NS. The authors used a DMI-TPB sensor, but instead of o-NPOE, they used DOP as the plasticizer. They investigated two-component mixtures of four CSs and three NSs and concluded that errors appeared when the consumption ratio of titrant for each component in the mixture surpassed 3:1. This problem was solved by the known addition of the surfactant with a lower consumption of the titrant.
In 2011, Najafi et al. [52] developed CSs selective electrode using single-walled carbon nanotubes (SWCNTs), for the first time. SWCNTs and CTAB were mixed and used as an ionophore. Due to the specific properties of SWCNT, the proposed electrode showed lower data scattering and better stability, compared to conventional PVC membrane electrode.
In 2012, Devi and Chattopadhyaya [53] developed ISE for CSs determination, using CP based Sn(iv) phosphate (CP-SnP) as sensor material. The authors determined CPC in mouthwash using direct potentiometry and potentiometric titration. Both results were in good agreement with those obtained using two-phase titration.
In 2014, Zorin et al. [54] demonstrated applicability of using polyelectrolyte-surfactant complex as sensor material in ISE for CSs determination. In developed electrodes, they used poly-2-acrylamido-2-methylpropanesulfonic acid (PAMPS) and different ammonium surfactants.
In 2021, Sakač et al. [55] synthesized a new long-chain quaternary alkyl ammonium salt, 1,3-dihexadecyl-1H-benzo[d]imidazole-3-ium bromide (DHBI-Br), and used its ion pair with TPB (DHBI-TPB), as sensor material in ISE for CSs determination. Also, they used a prepared electrode for simultaneous determination of CSs and NSs in solution containing barium cation, by potentiometric titration. Thus, the first inflexion point was for CS and the second was for NS. The authors showed that increased concentration of etoxylated (EO) NS, as well as the number of EO groups in the NS molecule, negatively affected the shape of the titration curve and the change in potential. The applicability of the sensor for CSs determination in personal care products and disinfectants was demonstrated. The same group of authors [35] investigated the influence of six different plasticizers on the potentiometric response characteristics of the DMI-TPB sensor. The best results were obtained using o-NPOE as the plasticizer. In 2022, they [56] synthesized one more quaternary alkyl ammonium salt, 1,3-dioctadecyl-1H-imidazol-3-ium bromide (DODI-Br). Again, they used its ion pair with TPB (DODI-TPB) as sensor material in ISE for CSs determination. The authors successfully used the proposed sensor for CSs determination in disinfectants and antiseptics. One year later, the same group of authors [57] demonstrated the use of their DODI-TPB sensor for the determination of technical grade quaternary ammonium compounds commonly contained in COVID-19 disinfectants and antiseptics: hyamine, N,N-didecyl-N,N-dimethylammonium chloride (DDAC), and N,N-dioctyl-N,N-dimethylammonium chloride (DOAC).
As it can be seen from above, many different conventional ISEs with liquid type of membrane, for CSs determination were developed. Their main drawback is leakage of the sensor material, which leads to a short lifetime of the ISE, due to changes in analytical performance and potentiometric response. Therefore, many authors tried to optimize the composition of their ISE membrane by using less water-soluble sensing materials. Some of them synthesized new CSs, used as part of sensing material. Some authors have even developed ISEs without sensor materials because gradual saturation of the plasticizer with the ion pair formed during the titration was expected. However, such electrodes usually had a sub-Nernstian slope and a slow response time. Also, it is shown that introducing nanomaterials into membrane composition leads to better electrode stability. Using conventional types of electrodes, CSs were commonly determined in mouthwashes, nose and eye drops, disinfectants, and antiseptics. A chronological overview of the conventional ISEs with liquid membranes for potentiometric determination of CSs is presented in Table 1.
Chronological overview of the conventional ISEs with liquid membranes for potentiometric determination of CSs
| Sensor design | Analyte | Sample | Response parameters | Lifetime (days) | Year | Ref. | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sensor material | Plasticizer | Slope (mV/decade of activity) | R2 | LOD (M) | Measuring range (M) | Response time (s) | Working pH range | Recovery (%) | |||||
| CP-IM | DOP | CP | Mouthwash | 29.0 ± 0.4 | 0.9970 | 4.00 × 10−6 | 2.00 × 10−6–1.00 × 10−3 | 15 (HCR) | 3.0–6.0 | 98.80 | 28 | 2000 | [46] |
| 60 (LCR) | |||||||||||||
| CP-FTC | DOP | CP | Mouthwash | 57.5 ± 0.4 | 0.9900 | 8.00 × 10−7 | 1.00 × 10−6–1.00 × 10−3 | 30 (HCR) | 1.0–6.0 | 98.50 | 28 | 2001 | [47] |
| 60 (LCR) | |||||||||||||
| TTA-DDS | DOP | TTA | Analyte solution | 59.3 | — | — | 3.98 × 10−6–1.00 × 10−3 | 60 | 1.0–6.5 | — | 90 | 2004 | [48] |
| — | NPDOE | DTA | Analyte solution | 57.3 ± 1.2 | 0.9996 | — | 1.00 × 10−6–1.00 × 10−2 | 10 | 2.0–11.0 | — | — | 2004 | [34] |
| s-BT-TPB | DOP | CPC | Synthetic matrices, mouthwash | 57.0 ± 0.4 | 0.9970 | 4.00 × 10−6 | 6.00 × 10−6–1.00 × 10−3 | 10 (HCR) | 6.0–12.0 | 98.00–99.00 | 42 | 2006 | [49] |
| 30 (LCR) | |||||||||||||
| DMI-TPB | o-NPOE | CPC, CTAB, Hyamine | Mouthwashes, disinfectants | 58.6 ± 0.4 | 0.9999 | 1.40 × 10−6 | 2.00 × 10−6–1.00 × 10−4 | 3 (HCR) | 2.0–11.0 | — | Several months | 2008 | [50] |
| 10 (LCR) | |||||||||||||
| DMI-TPB | DOP | CSs and NSs mixture | Cleaning products, disinfection agents | — | — | — | — | — | 3.0–10.0 | 97.7–104.5 | >90 | 2011 | [51] |
| CTAB-SWCNT | Elvaloy 742 | CTAB | — | 57.8 | 0.9710 | — | 3.00 × 10−5–7.00 × 10−4 | 30 (HCR) | — | — | 90 | 2011 | [52] |
| 300 (LCR) | |||||||||||||
| CPC-SnP | DOP | CPC | Mouthwash | 29.1 | 0.9920 | — | 5.00 × 10−6–5.00 × 10−3 | 30 | 2.0–6.0 | — | — | 2012 | [53] |
| PAMPS-CTA | DOP | CTAB | Analyte solution | 57.7 ± 1.4 | — | 1.00 × 10−7 | 4.00 × 10−7–5.00 × 10−4 | 40 | 4.0–9.0 | — | 120–150 | 2014 | [54] |
| DHBI-TPB | o-NPOE | CPC, CTAB, Hyamine | Raw industrial CSs samples, mouthwash, eye drops, nose drops, medical skin disinfectant, hospital disinfectant | 59.2 ± 0.5 | 0.9999 | 0.30 × 10−6 | 2.00 × 10−6–1.00 × 10−4 | few s | 2.0–11.0 | 98.0–102.0 | >120 | 2021 | [55] |
| DMI-TPB | o-NPOE | CPC, CTAB, Hyamine, DMIC | Disinfectants, antiseptics | 59.10 ± 0.5 | 0.9945 | 2.10 × 10−7 | 3.90 × 10−7–3.20 × 10−3 | — | — | 98.2–99.6 | — | 2021 | [35] |
| DODI-TPB | o-NPOE | CPC, CTAB, Hyamine, DMIC | Disinfectants, antiseptics | 58.70 ± 0.5 | 0.9997 | 0.90 × 10−6 | 1.80 × 10−6–1.00 × 10−4 | — | 2.0–10.0 | 98.9–101.2 | — | 2022 | [56] |
| DODI-TPB | o-NPOE | Hyamine, DDAC, DOAC | COVID-19 disinfectants, antiseptics | — | — | — | — | — | — | 96.4–102.2 | — | 2023 | [57] |
3.2 Solid-state ISEs
Although conventional ISEs have been used for CSs determination for a long period of time, they have a problem of leaching sensor material, which causes an unstable potential response. The solid-state ISEs for CSs determination were developed in order to overcome this problem and to further simplify ISEs construction. The absence of an unwanted inner water layer led to enhanced ISEs performance. Also, solid-state ISEs can easily be miniaturized, which leads to lower reagents and samples consumption. A distinction should be made between solid-state ISEs with the liquid membrane (solid-contact ISEs) (Figure 4b) and all-solid-state ISEs (Figure 4c). The most commonly used solid-state concepts for CSs determination include coated-wire, graphite-based, and screen-printed electrodes. CSs determination using solid-state electrodes began in the 1970s.
In 1974, Fogg et al. [58] described the solid-state silicone-rubber membrane ISE for CSs determination, based on the ion pair of CS and AS. The membrane contained hexadecyltrimethylammonium (CTA) DDS, was attached to the lead glass tube. The proposed ISE responded only to CSs, not to ASs.
Coated-wire electrodes are ISEs formed by coating of thin polymer films, containing sensor material, onto a metallic conductor [59]. In order to obtain the desired film thickness, metal wire can be immersed in a membrane cocktail a few times, with the electrode drying period between every immersion. In addition to the fact that the construction of these electrodes is very simple, it is also low cost, and allows for a highly miniaturized design. Also, by changing the content of the coating material or wire construction, they can easily be modified and adopted for various applications. The first coated-wire ISE was developed in 1971 for Ca2+ determination [60]. The optimization of coatings, using different sensor materials and plasticizers, followed.
Vytřas et al. [61] developed an aluminum coated-wire electrode without an ion-exchanger, for the determination of CS and AS using potentiometric titration. The determination was based on the formation of an ion-pair between the analyte and the titrant. They used PVC and poly(vinyl butyral) (PVB) as plastic matrix and obtained better results using PVC. Six different plasticizers were used for membrane optimization, and the best results were obtained using 2-nitrophenyl 2-ethylhexyl ether. They used the same type of electrode for CS determination using indirect titration [62] where the excess sodium TPB (NaTPB) used as a titrant was back-titrated with thallium(i) nitrate solution.
Pinzauti et al. [63] developed a robust silver-silver sulfide solid-state electrode for CSs, sodium halides, and thiols determination using potentiometric titration. CSs were analyzed in antiseptic formulations and contact lens solution.
Dowle et al. [64] developed a graphite electrode with a PVC membrane plasticized with tritolyl phosphate, based on the tetrabutylammonium dodecylsulfate ion pair. Compared to the blank electrode, the electrode with the ion pair had a faster response time and a better slope. Also, the electrode showed good performance in mixed systems containing water and ethanol if the ethanol content was lower than 20%. It was successfully applied for CSs determination in washing-up liquids and washing powders using potentiometric titration. Using the same membrane mixture, Dowle et al. [65] also developed flow-through electrode for CSs determination in routine control analyses. The membrane solution was applied to the inner surface of a graphite tube. The electrode showed a limited lifetime due to the leaching of the sensor material.
In 2000, Matešić-Puač et al. [66] developed and compared two electrodes for CSs determination. The electrodes contained a PVC membrane plasticized with o-NPOE, and the Ca2+ carrier N,N,N′N′-tetracyclohexyl-3-oxapentanediamide (NTO) as the sensing material. The potassium tetrakis (4-chlorophenyl) borate was used as a lipophilic anionic site in order to reduce the coextraction of anions in the membrane. Both electrodes were obtained by coating of membrane mixture on the body of the electrode (graphite-polytetrafluoroethylene (PTFE) universal Ružička selectrode and platinum metallic electrode). Although both electrodes were successfully applied for CSs determination in commercial formulations, better results were obtained using graphite-PTFE electrode. The same group of authors [67] developed and compared four solid-contact ISEs for CSs determination. All ISEs had teflonized graphite as the conducting substrate, but every ISE had a different ion-pair complex as the sensor material. Only the electrode with dodecyldimethylbenzylammonium dodecylbenzenesulfonate exhibited a Nernstian response for both CSs investigated.
Various attempts to improve the selectivity of the electrodes followed. PVC molecular sieves with tailored pore size proved to be suitable for this purpose [68]. Also, zeolites with their unique molecular sieving properties, based on the size and shape of the molecules, can be successfully applied for ISE design [69].
In 2000, Kulapin and Arinushkina [70] developed and compared few solid-contact ISEs. They used graphite and silver leads. CP dodecylsulfate and CP-TPB were used as sensor materials, and dibutyl phthalate (DBP) as a plasticizer. They used the developed ISEs for separate determination of ASs, CSs, and NSs, and for simultaneous determination of CSs and NSs. In 2003, Kulapin et al. [71] applied a new approach for the potentiometric determination of CSs. They developed graphite solid-contact ISE with membrane modified with PVC molecular sieves. Such a concept significantly improved electrode selectivity. Molecular sieves were inert PVC matrices, plasticized with DBP, with different pore diameters. As membrane sensor material, they used CP-TPB. Using such a modified electrode, it was possible to separately determine alkylpyridinium chlorides in binary and ternary mixtures. In 2005, the same group of authors [72] made additional improvements. Using ISEs that contained molecular sieves with predetermined pore sizes in their membranes, they improved selectivity of the sensors. Furthermore, using inselective sensors they developed a multisensor system, like an “electronic tongue” for the separate determination of surfactant homologues in multicomponent solutions. Artificial neural networks were utilized to process analytical signals. They were trained on pure multicomponent solutions. It allowed the determination of composition and concentrations of surfactants in a five-component solution.
In 2002, Matysik et al. [73] developed an all-solid-state disposable planar ISE with a zeolite-PDMS membrane casted on gold leaf, which was fixed to the copper surface of the electrode. Although the electrode had a disposable character due to its low cost and simple preparation procedure, it had good stability. This was a consequence of the structure of zeolite-PDMS membrane, which prevented the leaching of the membrane components. The electrode was successfully used for CSs and ASs determination using potentiometric titration.
In 2006, Plesha et al. [74] developed and compared three coated-wire ISEs with different sensor materials, for CSs determination in cleaning solutions. All ISEs proposed, exhibited a sub-Nernstian response. Also, they investigated the effect of CSs on other ISEs with PVC membranes, and concluded that surfactant concentrations higher than 1 × 10−5 M significantly decreased the slope of the ISE.
At the end of the 2000s, researchers realized that there was a need to develop miniaturized electrodes, that would be suitable for in situ monitoring using portable analyzers. Therefore, they began to develop screen-printed electrodes for CSs determination. Screen-printed electrodes are ISEs produced by printing ink, containing sensor material, on plastic or ceramic substrates [75]. These electrodes are robust, disposable, low cost, and offer versatile electrode designs, including working and the reference electrode on the same substrate, or multiplexed working electrode designs. Furthermore, it is easy to prepare and modify these electrodes, using home-made carbon ink. One of the main advantages of screen-printed electrodes is the possibility of performing measurements in small volumes, which leads to a significant reduction in sample and reagent volumes.
In 2008, Khaled et al. [76] developed the first disposable screen-printed carbon paste electrodes for CSs determination, using home-made printing ink. The electrodes contained both, working and reference electrodes in the same strip. Effects of plasticizer type and content, binding material, carbon content, and solvent were investigated in order to get an optimized printing ink formulation. The optimized screen-printed electrodes were applied for CSs determination in pharmaceutical preparations. The results were in good agreement with those obtained using commercial surfactant electrode and the official method. Also, the analytical performances of the new electrode were compared with those of coated-wire, coated-graphite, carbon paste, and conventional electrodes. The proposed electrodes enabled significant reductions in reagent and analyte volumes. In 2010, Mohamed et al. [77] performed the analytical characterization of their electrodes. They also determined the CTAB concentration in pharmaceutical formulations and spiked sea water. In the same year, Mohamed et al. [78] modified their electrode using tricresylphosphate (TCP) as a plasticizer. The electrode was used for CPC determination. One year later, Mohamed et al. [79] made another modification of their screen-printed carbon paste electrode. They added an ion pair, made of 1-(ethoxycarbonyl)pentadecyltrimethylammonium bromide (Septonex) and TPB, in the carbon ink used for electrode preparation. They used the new electrode for Septonex determination by potentiometric titration and direct potentiometry. In 2014, Ali et al. [80] developed and optimized a carbon paste electrode for dodecyl(2-hydroxyethyl)dimethylammonium (DHDA) determination based on potassium tetrakis(4-chlorophenyl)borate (KTCPB) as the sensor material. They varied the content of the sensor material and the type of the plasticizer, and concluded that the sensor with 7.5% of the sensor material and DOP as the plasticizer, exhibited the best results. The applicability of the sensor for DHDA determination in water samples was demonstrated. In 2015, Ali et al. [81] prepared carbon paste electrode for 1-dodecyl-5-methyl-1H-benzo[d][1,2,3]triazol-1-ium bromide (DMTB) determination. This time, the sensor material was 1,4-bis-(8-mercaptooctyloxy)-benzene (MPB). Again, they varied the content of the sensor material and the type of the plasticizer, but this time, the best results were obtained using 10% of the sensor material and o-NPOE as the plasticizer. In 2017, Khaled et al. [82] applied a completely new approach and optimized their disposable screen-printed electrodes, thus developing enzymatic ISEs for CSs determination. The electrodes were prepared using ion-sensing printing ink printed on graphite/PVC conducting track. The ink cocktail contained α-cyclodextrin (α-CD) as the sensing ionophore, KTCPB as the anionic site, 2-fluorophenyl 2-nitrophenyl ether (f-PNPE) as the plasticizer, THF as the solvent, PVC, and multi-walled carbon nanotubes (MWCNTs) as the ion-to-electron transducer for improving the stability of the electrode potential. The working principle of the proposed biosensor was inhibition of the enzyme acetylcholinesterase (AChE) by surfactants followed by measuring the residual activity of the enzyme using the proposed biosensor. The inhibition of the enzyme was dependent on the nature of the surfactants. The developed biosensor was successfully applied for CSs determination in mouthwash and commercial detergent.
In 2010, Price and Wan [83] tested the Metrohm ionic surfactant electrode for quaternary ammonium compounds determination in eight antimicrobial products, using the potentiometric titration method. They emphasized that the electrode was very sensitive to ethanol and methanol, and that organic solvents caused deterioration of the membrane which significantly reduced the electrode lifetime. It should be taken into consideration due to the presence of ethanol in many antimicrobial products.
In 2012, Gaber et al. [84] developed and optimized an all-solid-state sensor for Hyamine determination. As sensor materials, they tested ion pairs of Hyamine and four heteropoly acids. The best results were obtained using electrode with ion pair composed of Hyamine and phosphomolybdic acid (Hyamine-PM). Also, the authors varied plasticizers. They used five plasticizers, and concluded that the electrodes plasticized by a mixture of plasticizers (DBP and DOP) gave better results, compared to electrodes plasticized by a single plasticizer. The optimized carbon paste electrode was successfully applied for Hyamine determination in eye and ear drops.
In 2016, Issa et al. [85] developed and optimized carbon paste electrode for Hyamine determination based on Hyamine-TPB ion pair as sensor material. In the optimization process, the authors varied the content of the sensor material and the type of the plasticizer. The best results were obtained using the electrode with 5% of sensor material, and tricresyl phosphate (TCP) as the plasticizer. The applicability of the sensor was shown for Hyamine determination in antibacterial spray. The authors also prepared and optimized conventional membrane-sensor, but it exibited worse results than those obtained using carbon paste electrode.
Till the end of the 2010s, it was well known that carbon nanomaterials have unique properties such as a high surface-to-volume ratio, the existence of mobile electrons on the nanotubes surfaces, high hydrophobicity, and the ability to facilitate electron transfer [86]. These properties can lead to the development of stable and reliable ISEs with prolonged lifetime and improved response characteristics [87]. Therefore, the introduction of nanomaterials in ISE design was a promising research area. They can be implemented in ISE design as transducers, as mentioned above, or as part of sensor materials [88].
In 2017, Hajduković et al. [89] applied a new approach for developing potentiometric sensor for CSs determination. They used a hybrid sensor material that consisted of MWCNTs functionalized with a sulfate group and CP cation (MWCNT-OSO3 −CP+). The chemical modification of MWCNTs led to improved membrane stability and signal drift, as well as a prolonged life time of the sensor due to the reduced leaching of the sensor material from the membrane. Also, immobile MWCNT-OSO3 − ion in the membrane formed repulsive electrostatic barrier for the anions. To demonstrate the applicability of the sensor, the authors successfully determined CSs in commercial products and mixtures of two CSs.
In 2023, Frag et al. [36] developed a carbon paste electrode for nonyltrimethylammonium bromide (NTAB) determination in formation water. For the carbon paste optimization, they used five plasticizers and different amounts of four ion pairing agents. The best results were obtained using NaTPB, as an ion pairing agent, and TCP as the plasticizer.
As it can be seen from above, solid-state electrodes were predominant for CSs determination. The coated-wire ISEs are simple, but their performance may show deterioration over time. Disposable strip screen-printed electrodes are easy to mass produce and open the ability for field measurements. Compared to conventional electrodes and coated-wire ISEs, carbon paste electrodes have very low Ohmic resistance, a short response time, and a long lifetime [80]. Also, their preparation is very simple, handmade paste is easy to modify, and the developed electrode can be used immediately after assembly without additional time for solvent evaporation. Many authors made an effort to improve the selectivity of their ISEs by introducing new sensor materials and molecular sieves, or to improve analytical characteristics by optimizing the sensor material and plasticizer content and introducing nanomaterials into their electrode designs. Solid-state ISEs were mainly used for CSs determination in disinfectants, hair rinses, mouthwashes, eye and ear drops, fabric softeners, and cleaning solutions. A chronological overview of the solid-state ISEs for potentiometric determination of CSs is presented in Table 2.
Chronological overview of the solid-state ISEs for potentiometric determination of CSs
| Sensor design | Analyte | Sample | Response parameters | Lifetime (days) | Year | Ref. | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Type of the sensor | Type of membrane | Sensor material | Plasticizer | Slope (mV/decade of activity) | R 2 | LOD (M) | Measuring range (M) | Response time (s) | Working pH range | Recovery (%) | |||||
| Solid-contact (graphite) | Liquid | NTO | o-NPOE | CSs, Hyamine | Commercial products | 55.7 | 0.9997 | 3.00 × 10−6 | 5.00 × 10−6–4.00 × 10−3 | 3–4 (HCR) | 2.0–13.0 | 100.21–101.57 | — | 2000 | [66] |
| 20–30 (LCR) | |||||||||||||||
| Solid-contact (graphite) | Liquid | THA-DDS | o-NPOE | CTA | Analyte solution | 58.6 ± 0.1 | 0.9990 | 2.40 × 10−6 | 2.40 × 10−6–1.00 × 10−2 | 30 (HCR) | — | — | 90–120 | 2004 | [67] |
| 60 (LCR) | |||||||||||||||
| Solid-contact (graphite, silver) | Liquid | CP-DDS and CP-TPB | DBP | CSs, CP, CMA | Hair rinses | 56.0 ± 2.0 | — | — | 1.00 × 10−6–1.00 × 10−3 | — | 2.0–10.0 | 97.67–98.25 | 300–360 | 2000 | [70] |
| Solid-contact (graphite) | Liquid with molecular sieves | CP-TPB | DBP | CSs | Binary and ternary model mixtures of CSs | 56.0 ± 2.0 | — | — | 1.00 × 10−6–1.00 × 10−3 | 120–180 | — | 92.18–107.14 | 180 | 2003 | [71] |
| Solid-contact (graphite) | Liquid with molecular sieves | APAS; APTPB | DBP | CSs | Model mixtures of CSs | 57.0 ± 2.0 | — | 9.00 × 10−7 | 1.00 × 10−6–1.00 × 10−3 | 60–120 | — | — | 300 | 2005 | [72] |
| All-solid-state (gold, copper) | Solid | zeolite-PDMS | — | HDP | Analyte solution | — | — | — | — | — | — | — | 90 | 2002 | [73] |
| Solid-contact (copper) | Liquid | DNSA | DOP | BDTA, BDDA, BDHA | Cleaning solutions | 53.5 ± 0.7 | — | 6.31 × 10−7 | 1.00 × 10−6–1.00 × 10−3 | — | — | — | 50 | 2006 | [74] |
| Screen-printed | — | — | o-NPOE | CSs, CPC | Pharmaceutical formulations | — | — | — | — | 3 (HCR) | — | 98.10–99.00 | Disposable | 2008 | [76] |
| 5 (LCR) | |||||||||||||||
| Screen-printed | — | — | o-NPOE | CTAB | Pharmaceutical formulations, sea water | 58.7 ± 1.3 | — | 6.80 × 10−7 | — | 3 (HCR) | 3.0–8.0 | 98.80–100.20 | Disposable (120) | 2010 | [77] |
| 5 (LCR) | |||||||||||||||
| Screen-printed | — | — | TCP | CPC | Pharmaceutical formulations | 60.66 ± 1.10 | 0.9980 | 8.00 × 10−7 | 1.00 × 10−6–1.00 × 10−2 | 3 (HCR) | 2.0–8.0 | 99.60 | 90 | 2010 | [78] |
| 5 (LCR) | |||||||||||||||
| Screen-printed | — | Septonex-TPB | o-NPOE | Septonex | Pharmaceutical preparations | 59.33 ± 0.85 | 0.9980 | 9.00 × 10−7 | 5.00 × 10−7–1.50 × 10−2 | 3–6 | 2.0–9.0 | 99.00–99.27 | 90 | 2011 | [79] |
| All-solid-state (carbon) | Solid | KTCPB | DOP | DHDA | Water samples | 59.79 ± 0.83 | — | 1.00 × 10−7 | 1.00 × 10−7–1.00 × 10−2 | 8 | 2.0–9.0 | 98.00–99.53 | 110 | 2014 | [80] |
| All-solid-state (carbon) | Solid | MPB | o-NPOE | DMTB | Water samples | 58.59 ± 1.31 | 0.9970 | 1.40 × 10−7 | 1.40 × 10−7–1.00 × 10−2 | 6 | 2.0–9.0 | 99.01 | 63 | 2015 | [81] |
| Screen-printed | — | α-CD | f-PNPE | CPC, BDHAC, CTAB, Hyamine | Mouthwash, commercial detergent | 92.94 ± 1.78 | 0.9991 | 2.06 × 10−7 | 0–2.50 × 10−6 | — | — | 95.40–103.40 | Disposable | 2017 | [82] |
| Solid-contact | — | — | — | CSs, Hyamine | Disinfectants | — | — | — | — | — | — | 90.00–101.00 | — | 2010 | [83] |
| All-solid-state (graphite) | Solid | Hyamine-PM | DBP + DOP | Hyamine | Eye and ear drops | 58.20 ± 0.60 | 0.9880 | 1.00 × 10−7 | 1.00 × 10−7–1.70 × 10−4 | 5–10 | 5.7–8.6 | 97.30–101.8 | Several months | 2012 | [84] |
| All-solid-state (carbon) | Solid | Hyamine-TPB | TCP | Hyamine | Antibacterial spray | 59.85 ± 0.49 | 0.9972 | 3.16 × 10−7 | 5.96 × 10−7–3.03 × 10−3 | < 10 | 1.0–9.0 | 98.57–100.71 | 30 | 2016 | [85] |
| Solid-contact (graphite) | Liquid | MWCNT-OSO3 −CP+ | o-NPOE | CPC, CTAB, Hyamine | Fabric softener, laundry disinfectant, hand sanitizer | 58.90 ± 0.6 | 0.9994 | 1.20 × 10−7 | 1.50 × 10−7–9.10 × 10−4 | 5 | 3.0–10.0 | 100.3–102.6 | 180 | 2017 | [89] |
| All-solid-state (carbon) | Solid | NTA-TPB | TCP | NTAB | Formation water | 58.40 ± 0.8 | 0.9999 | 1.00 × 10−6 | 1.00 × 10−6–1.00 × 10−2 | 10 | 2.5–8.0 | 98.6–100.7 | 37 | 2023 | [36] |
3.3 Ion-sensitive field-effect transistors
An ion-sensitive field-effect transistor (ISFET) is a potentiometric device obtained by coupling ISE and a field-effect transistor (FET). It is based on a change in current through the transistor as a consequence of a change in ion concentration in the analyte solution. ISFETs have fast response and good reproducibility [90]. They are robust and easy to handle. Compared to conventional ISEs, they have a low impedance output signal, are easier to miniaturize, and, due to their small size, require much lower volumes of sample and reagents. Their main advantage compared to conventional ISEs is their compatibility with complementary metal-oxide semiconductor (CMOS) technology, which enables integration of electronics and the sensor on the same chip and leads to low-cost mass fabrication of multi-parametric sensors with low power consumption [90]. Furthermore, using those principles, wearable multianalyte sensing platforms with real-time monitoring can be developed [91]. Also, ISFETs are widely used in continuous flow systems (flow injection analysis and sequential injection analysis), providing automatic sampling and calibration [92,93,94,95]. Considering the above, ISFETs found their application in different areas of analytical chemistry, so it is surprising that only a few authors have developed ISFETs for CSs determination.
Campanella et al. [96] developed three ISFETs for CSs determination based on PVC membranes plasticized with bis(2-ethylhexy1)sebacate. As sensor materials, they used benzyldimethyl-hexadecylammonium reineckate, DTA reineckate and HDP phosphotungstate, respectively. The sensor with the best analytical characteristics was applied for CSs determination in environmental water samples.
Masadome et al. [97] used ISFET to investigate the contribution of the plasticizer to the response of ISE for ionic surfactants determination. They concluded that the plasticizer itself does not respond to surfactants, but a plasticized PVC membrane with no sensor material exhibited a Nernstian response. In 2006, Masadome et al. [98] developed and optimized ISFET for CSs determination. They varied the sensor materials and showed that the ISFET, based on an ion pair formed between the benzyldimethyltetradecylammonium (Zephiramine) and tetrakis[3,5-bis(trifluoromethyl)phenyl]borate (TFPB), exhibited the best analytical performance. o-NPOE was used as the plasticizer. Also, they developed a system for CSs determination, composed of a microfluidic polymer-based microchip embedded with a developed ISFET. Both developed systems exhibited similar results. The obtained slope was 58.5 ± 1.7 mV/decade of activity in the range from 1.00 × 10−6 to 1.00 × 10−3 M. The LOD was 2.90 × 10−7 M. The ISFET could be used in pH range from 3 to 9. Furthermore, the microchip integrated with the developed ISFET, was successfully applied for CPC determination in dental rinses. The accuracies obtained were in the range between 94.70% and 103.30%.
Although ISFETs can be used as fast and reliable devices for CSs determination, they are not widely used in practice. The reason for that could be their long-term drift and hysteresis which lead to limited accuracy for long-term measurements [99]. Furthermore, temperature variations cause thermal drift, which causes the nonlinear behavior of ISFET device [100]. In samples with low analyte concentrations, achieving a good signal-to-noise ratio can be challenging. Also, the complexity of fabrication, compared to conventional and solid-state ISEs could be a problem.
4 Conclusion
CSs are compounds widely used for different purposes. Apart from their desirable properties, they exhibit toxicity. Therefore, their determination in order to control the quality of commercial products, industrial processes, and their penetration into the environment is very important. Potentiometry and potentiometric sensors are among the most commonly used methods for this purpose due to their accuracy, reliability, simplicity, low cost, fast analysis time, eco-friendliness, and suitability for routine in situ analyses. From the period when the first potentiometric sensor for CSs determination was developed until today, significant progress has been made. It was achieved by a deeper understanding of the chemical and physical processes that affect ISE response and by improving the analytical performance of ISEs. Using new membrane materials and ionophores resulted in a lowering of the LOD values, a widening of the measuring range, and an improvement in the selectivity of ISEs. Introducing nanomaterials into the membrane of ISEs led to reduced signal drift, noise, and electrical resistance, and also improved the dynamic response of the ISEs. The elimination of an inner electrolyte in conventional ISEs and the development of solid-contact ISEs contributed to the significant improvement in the durability of electrodes. Further, electroactive materials characterized by electronic and ionic conductivity, that were used as ion-to-electron transducers between the membrane and electronic conductor, significantly improved the potential stability of ISEs. Additional improvement of all-solid-state electrodes led to the development of miniaturized electrodes and screen-printed electrodes that could be used in flow analysis and are more convenient for in situ analysis. This review summarizes potentiometric sensors developed for CSs determination, their advantages, and their drawbacks. Due to their simplicity and good analytical performance, solid-state electrodes demonstrated themselves to be the most appropriate and, consequently, the most frequently used type of sensor for determining CSs. Despite extensive research in this field, future progress can be expected. The combination of potentiometric sensors and nanotechnology represents a promising area for research. Additional efforts should be made to achieve calibration-free sensing and real-time CSs monitoring in order to develop sensors for in situ analysis with no maintenance. These sensors should be miniaturized solid-state electrodes with no drift of the electromotive force (EMF) response, which probably could be achieved using suitable materials as solid contacts. Also, the drift of the reference electrode should be considered. Furthermore, such electrodes should resist the adsorption of contaminants and biomolecules of the sample, which is still a great challenge. Some improvements could probably be achieved using suitable coatings [101]. Considering that self-calibrating and calibration-free sensors developed for continuous real-time monitoring have already been developed for some analytes [102,103,104], it can be expected that in the near future, such sensors will also be developed for CSs determination. Moreover, multi-analyte determination and achieving ISEs for trace-level analysis with enhanced selectivity, durability, and sensitivity remain challenges.
-
Funding information: Authors state no funding involved.
-
Author contributions: Mateja Budetić: conceptualization, investigation, writing – original draft, visualization, supervision, and writing – review and editing; Marija Jozanović: investigation and visualization; Iva Pukleš: investigation and visualization; Mirela Samardžić: conceptualization, investigation, writing – original draft, visualization, supervision, and writing – review and editing.
-
Conflict of interest: Authors state no conflict of interest.
-
Data availability statement: Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
References
[1] Myers D. Surfactant science and technology. 3rd edn. Hoboken: John Wiley & sons, Inc; 2006.Search in Google Scholar
[2] Surfactants Market Size | Global Industry Forecast https://www.marketsandmarkets.com/Market-Reports/biosurfactants-market-493.html (accessed nov 14, 2023).Search in Google Scholar
[3] Gonçalves RA, Holmberg K, Lindman B. Cationic surfactants: A review. J Mol Liq. 2023;375:121335. 10.1016/j.molliq.2023.121335.Search in Google Scholar
[4] Zakharova LY, Pashirova TN, Doktorovova S, Fernandes AR, Sanchez-Lopez E, Silva AM, et al. Cationic surfactants: Self-assembly, structure-activity correlation and their biological applications. Int J Mol Sci. 2019;20(22):5534. 10.3390/ijms20225534.Search in Google Scholar PubMed PubMed Central
[5] Obłąk E, Piecuch A, Rewak-Soroczyńska J, Paluch E. Activity of gemini quaternary ammonium salts against microorganisms. Appl Microbiol Biotechnol. 2019;103(2):625–32. 10.1007/s00253-018-9523-2.Search in Google Scholar PubMed
[6] Migahed MA, Negm NA, Shaban MM, Ali TA, Fadda AA. Synthesis, characterization, surface and biological activity of diquaternary cationic surfactants containing ester linkage. J Surfactants Deterg. 2016;19(1):119–28. 10.1007/s11743-015-1749-8.Search in Google Scholar
[7] Karamov EV, Larichev VF, Kornilaeva GV, Fedyakina IT, Turgiev AS, Shibaev AV, et al. Cationic surfactants as disinfectants against SARS-CoV-2. Int J Mol Sci. 2022;23(12):6645. 10.3390/ijms23126645.Search in Google Scholar PubMed PubMed Central
[8] Baker N, Williams AJ, Tropsha A, Ekins S. Repurposing quaternary ammonium compounds as potential treatments for COVID-19. Pharm Res. 2020;37(6):1–4. 10.1007/s11095-020-02842-8.Search in Google Scholar PubMed PubMed Central
[9] Hoque J, Akkapeddi P, Yarlagadda V, Uppu DSSM, Kumar P, Haldar J. Cleavable cationic antibacterial amphiphiles: Synthesis, mechanism of action, and cytotoxicities. 2012;28(33):12225–34. 10.1021/la302303d.Search in Google Scholar PubMed
[10] Tehrani-Bagha A, Holmberg K. Cleavable surfactants. Curr Opin Colloid Interface Sci. 2007;12(2):81–91. 10.1016/j.cocis.2007.05.006.Search in Google Scholar
[11] Damen M, Groenen AJJ, Van Dongen SFM, Nolte RJM, Scholte BJ, Feiters MC. Transfection by cationic gemini lipids and surfactants. Medchemcomm. 2018;9(9):1404–25. 10.1039/C8MD00249E.Search in Google Scholar PubMed PubMed Central
[12] Zhang XX, McIntosh TJ, Grinstaff MW. Functional lipids and lipoplexes for improved gene delivery. Biochimie. gener 2012;42–58. 10.1016/j.biochi.2011.05.005. NIH Public Access.Search in Google Scholar PubMed PubMed Central
[13] Wu SH, Lin HP. Synthesis of mesoporous silica nanoparticles. Chem Soc Rev. 2013;42(9):3862–75. 10.1039/c3cs35405a.Search in Google Scholar PubMed
[14] Lémery E, Briançon S, Chevalier Y, Bordes C, Oddos T, Gohier A, et al. Skin toxicity of surfactants: Structure/toxicity relationships. Colloids Surf A Physicochem Eng Asp. 2015;469:166–79. 10.1016/j.colsurfa.2015.01.019.Search in Google Scholar
[15] Passos MLC, Saraiva MLMFS, Lima JLFC. Application of sequential injection analysis to the determination of cationic surfactants based on the sensitized molybdenum-bromopyrogallol red reaction. Anal Sci. 2005;21(12):1509–14. 10.2116/analsci.21.1509.Search in Google Scholar PubMed
[16] Scott MJ, Jones MN. The biodegradation of surfactants in the environment. Biochim Biophys Acta - Biomembr. 2000;1508(1–2):235–51. 10.1016/S0304-4157(00)00013-7.Search in Google Scholar
[17] García MT, Ribosa I, Guindulain T, Sánchez-Leal J, Vives-Rego J. Fate and effect of monoalkyl quaternary ammonium surfactants in the aquatic environment. Env Pollut. 2001;111(1):169–75. 10.1016/S0269-7491(99)00322-X.Search in Google Scholar
[18] Kaczerewska O, Martins R, Figueiredo J, Loureiro S, Tedim J. Environmental behaviour and ecotoxicity of cationic surfactants towards marine organisms. J Hazard Mater. 2020;392:122299. 10.1016/j.jhazmat.2020.122299.Search in Google Scholar PubMed
[19] International organization for standardization. ISO 2871-1:2010 - Surface active agents — Detergents — Determination of cationic-active matter content — Part 1: High-molecular-mass cationic-active matter. 2010.Search in Google Scholar
[20] Vakh C, Evdokimova E, Pochivalov A, Moskvin L, Bulatov A. Effervescence assisted dispersive liquid–liquid microextraction followed by microvolume UV-Vis spectrophotometric determination of surfactants in water. Toxicol Env Chem. 2017;99(4):613–23. 10.1080/02772248.2016.1205069.Search in Google Scholar
[21] Biswas S, Pal A. Spectrophotometric determination of cationic surfactants in aqueous media using chrome azurol S as colour forming agent and 1-butanol as extracting solvent. Talanta. 2020;206:120238. 10.1016/j.talanta.2019.120238.Search in Google Scholar PubMed
[22] Morosanova MA, Morosanova EI. Solid phase spectrophotometric determination of cationic surfactants using silica-titania xerogel–pyrocatechol violet dye system. J Anal Chem. 2021;76(1):73–9. 10.1134/S1061934821010081.Search in Google Scholar
[23] Paun I, Iancu VI, Cruceru L, Niculescu M, Chiriac FL. Simultaneous determination of anionic, amphoteric and cationic surfactants mixtures in surface water. Rev Chim. 2018;69(1):27–30. 10.37358/rc.18.1.6038.Search in Google Scholar
[24] Wiest L, Giroud B, Assoumani A, Lestremau F, Vulliet E. A multi-family offline SPE LC-MS/MS analytical method for anionic, cationic and non-ionic surfactants quantification in surface water. Talanta. 2021;232:122441. 10.1016/j.talanta.2021.122441.Search in Google Scholar PubMed
[25] Sparham C, Ledbetter M, Cubberley R, Gore D, Sheffield D, Teixeira A, et al. Method validation and environmental monitoring of triethanolamine ester quaternary ammonium compounds. Chemosphere. 2024;346:140529. 10.1016/j.chemosphere.2023.140529.Search in Google Scholar PubMed
[26] Borse MS, Devi S. Importance of head group polarity in controlling aggregation properties of cationic gemini surfactants. Adv Colloid Interface Sci. 2006;123–126(Spec. Iss.):387–99. 10.1016/j.cis.2006.05.017.Search in Google Scholar PubMed
[27] Ahmady AR, Hosseinzadeh P, Solouk A, Akbari S, Szulc AM, Brycki BE. Cationic gemini surfactant properties, its potential as a promising bioapplication candidate, and strategies for improving its biocompatibility: A review. Adv Colloid Interface Sci. 2022;299:102581. 10.1016/j.cis.2021.102581.Search in Google Scholar PubMed
[28] Jesus CF, Alves AAS, Fiuza SM, Murtinho D, Antunes FE. Mini-review: Synthetic methods for the production of cationic sugar-based surfactants. J Mol Liq. 2021;342:117389. 10.1016/j.molliq.2021.117389.Search in Google Scholar
[29] Atkin R, Craig VSJ, Wanless EJ, Biggs S. Mechanism of cationic surfactant adsorption at the solid-aqueous interface. Adv Colloid Interface Sci. 2003;103(3):219–304. 10.1016/S0001-8686(03)00002-2.Search in Google Scholar PubMed
[30] Para G, Jarek E, Warszynski P. The Hofmeister series effect in adsorption of cationic surfactants-theoretical description and experimental results. Adv Colloid Interface Sci. 2006;122(1–3):39–55. 10.1016/j.cis.2006.06.017.Search in Google Scholar PubMed
[31] Warszyński P, Szyk-Warszyńska L, Wilk KA, Lamch Ł. Adsorption of cationic multicharged surfactants at liquid–gas interface. Curr Opin Colloid Interface Sci. 2022;59:101577. 10.1016/j.cocis.2022.101577.Search in Google Scholar
[32] Zdrachek E, Bakker E. Describing ion exchange at membrane electrodes for ions of different charge. Electroanalysis. 2018;30(4):633–40. 10.1002/elan.201700700.Search in Google Scholar
[33] Ivanova AD, Koltashova ES, Solovyeva EV, Peshkova MA, Mikhelson KN. Impact of the electrolyte co-extraction to the response of the ionophore-based ion-selective electrodes. Electrochim Acta. 2016;213:439–46. 10.1016/j.electacta.2016.07.142.Search in Google Scholar
[34] Masadome T, Yang JG, Imato T. Effect of plasticizer on the performance of the surfactant-selective electrode based on a poly(vinyl chloride) membrane with no added ion-exchanger. Microchim Acta. 2004;144(4):217–20. 10.1007/s00604-003-0112-3.Search in Google Scholar
[35] Sakač N, Madunić-Čačić D, Karnaš M, Đurin B, Kovač I, Jozanović M. The influence of plasticizers on the response characteristics of the surfactant sensor for cationic surfactant determination in disinfectants and antiseptics. Sensors. 2021;21(10):3535. 10.3390/s21103535.Search in Google Scholar PubMed PubMed Central
[36] Frag EY, Elashery SA, Ali AMA. A simple carbon electrode for the potentiometric determination of N,N,N-trimethyl-1-nonanammonium bromide as a cationic surfactant in formation water samples. J Anal Chem. 2023;78(1):105–12. 10.1134/S1061934823010045.Search in Google Scholar
[37] Gavach C, Seta P. Dosage potentiometrique des ions alkyl-trimethyl-ammonium a longue chaine et tetrabutyl-ammonium. Anal Chim Acta. 1970;50(3):407–12. 10.1016/0003-2670(70)80037-X.Search in Google Scholar
[38] Moody GJ, Oke RB, Thomas JDR. A calcium-sensitive electrode based on a liquid ion exchanger in a poly(vinyl chloride) matrix. Analyst. 1970;95(1136):910–8. 10.1039/an9709500910.Search in Google Scholar
[39] Cutler SG, Meares P, Hall DG. Surfactant-sensitive polymeric membrane electrodes. J Electroanal Chem. 1977;85(1):145–61. 10.1016/S0022-0728(77)80160-5.Search in Google Scholar
[40] Khalil MM, Angnel DF, Luca C. Poly (vinyl chloride) containing dibenzo-18-crown-6 as an ion-selective membrane for hyamine 1622. Anal Lett. 1986;19(7–8):807–24. 10.1080/00032718608066264.Search in Google Scholar
[41] Shoukry AF. Use of two ion-selective electrode-containing cells for potentiometric titration of hexadecylpyridinium bromide. Analyst. 1988;113(8):1305–8. 10.1039/AN9881301305.Search in Google Scholar
[42] Baillarger C, Mayaffre A, Turmine M, Letellier P, Suquet H. Clay membrane electrodes specific to cationic surfactants-applications. Electrochim Acta. 1994;39(6):813–6. 10.1016/0013-4686(93)E0010-J.Search in Google Scholar
[43] Gerlache M, Sentürk Z, Viré JC, Kauffmann JM. Potentiometric analysis of ionic surfactants by a new type of ion-selective electrode. Anal Chim Acta. 1997;349(1–3):59–65. 10.1016/S0003-2670(97)00277-8.Search in Google Scholar
[44] Matysik S, Matysik F-M, Mattusch J, Einicke W-D. Application of zeolite-polydimethylsiloxane electrodes to potentiometric studies of cationic species. Electroanalysis. 1998;10(2):98–102. 10.1002/(SICI)1521-4109(199802)10:2<98:AID-ELAN98>3.0.CO;2-N.Search in Google Scholar
[45] Saleh MB. Cationic surfactant ion-selective PVC membrane electrode containing macrocyclic diimine crown ether. Anal Lett. 1999;32(11):2201–15. 10.1080/00032719908542964.Search in Google Scholar
[46] Abbas MN, Mostafa GAE, Homoda AMA. Cetylpyridinium-iodomercurate PVC membrane ion selective electrode for the determination of cetylpyridinium cation in Ezafluor mouth wash and as a detector for some potentiometric titrations. Talanta. 2000;53(2):425–32. 10.1016/S0039-9140(00)00496-3.Search in Google Scholar
[47] Mostafa GAE. PVC matrix membrane sensor for potentiometric determination of cetylpyridinium chloride. Anal Sci. 2001;17(9):1043–7. 10.2116/analsci.17.1043.Search in Google Scholar
[48] Mahajan RK, Kaur I, Bakshi MS. Cationic surfactant-selective potentiometric sensors based on neutral ion-pair carrier complexes. J Surfactants Deterg. 2004;7(2):131–4. 10.1007/s11743-004-0295-9.Search in Google Scholar
[49] Mostafa GAE. s-Benzylthiuronium PVC matrix membrane sensor for potentiometric determination of cationic surfactants in some pharmaceutical formulation. J Pharm Biomed Anal. 2006;41(4):1110–5. 10.1016/j.jpba.2006.02.013.Search in Google Scholar
[50] Madunić-Čačić D, Sak-Bosnar M, Galović O, Sakač N, Matešić-Puač R. Determination of cationic surfactants in pharmaceutical disinfectants using a new sensitive potentiometric sensor. Talanta. 2008;76(2):259–64. 10.1016/j.talanta.2008.02.023.Search in Google Scholar
[51] Samardžić M, Sak-Bosnar M, Madunić-Čačić D. Simultaneous potentiometric determination of cationic and ethoxylated nonionic surfactants in liquid cleaners and disinfectants. Talanta. 2011;83(3):789–94. 10.1016/j.talanta.2010.10.046.Search in Google Scholar
[52] Najafi M, Maleki L, Rafati AA. Novel surfactant selective electrochemical sensors based on single walled carbon nanotubes. J Mol Liq. 2011;159(3):226–9. 10.1016/j.molliq.2011.01.013.Search in Google Scholar
[53] Devi S, Chattopadhyaya MC. A new PVC membrane ion selective electrode for determination of cationic surfactant in mouthwash. J Surfactants Deterg. 2012;15(3):387–91. 10.1007/s11743-011-1305-0.Search in Google Scholar
[54] Zorin I, Scherbinina T, Fetin P, Makarov I, Bilibin A. Novel surfactant-selective membrane electrode based on polyelectrolyte- surfactant complex. Talanta. 2014;130:177–81. 10.1016/j.talanta.2014.06.061.Search in Google Scholar PubMed
[55] Sakač N, Marković D, Šarkanj B, Madunić-Čačić D, Hajdek K, Smoljan B, et al. Direct potentiometric study of cationic and nonionic surfactants in disinfectants and personal care products by new surfactant sensor based on 1,3-dihexadecyl−1H-benzo[d]imidazol−3-ium. Molecules. 2021;26(5):1366. 10.3390/molecules26051366.Search in Google Scholar PubMed PubMed Central
[56] Sakač N, Madunić-Čačić D, Marković D, Ventura BD, Velotta R, Ptiček Siročić A, et al. The 1,3-dioctadecyl-1H-imidazol-3-ium based potentiometric surfactant sensor for detecting cationic surfactants in commercial products. 2022;22(23):9141. 10.3390/s22239141.Search in Google Scholar PubMed PubMed Central
[57] Sakač N, Madunić-Čačić D, Marković D, Jozanović M. Study of cationic surfactants raw materials for COVID-19 disinfecting formulations by potentiometric surfactant sensor. Sensors. 2023;23(4):2126. 10.3390/s23042126.Search in Google Scholar PubMed PubMed Central
[58] Fogg AG, Pathan AS, Burns DT. A silicone-rubber surfactant electrode. Anal Chim Acta. 1974;69(1):238–42. 10.1016/0003-2670(74)80034-6.Search in Google Scholar
[59] Freiser H. Coated wire ion-selective electrodes. Principles and practice. J Chem Soc Faraday Trans 1 Phys Chem Condens Phases. 1986;82(4):1217–21. 10.1039/F19868201217.Search in Google Scholar
[60] Cattrall RW, Freiser H, Cattrall RW. Coated wire ion selective electrodes. Anal Chem. 1971;43(13):1905–6. 10.1021/ac60307a032.Search in Google Scholar
[61] Vytřas K, Dajková M, Mach V. Coated-wire organic ion-selective electrodes in titrations based on ion-pair formation. Part 2. Determination of ionic surfactants. Anal Chim Acta. 1981;127(C):165–72. 10.1016/S0003-2670(01)83972-6.Search in Google Scholar
[62] Vytřas K, Kalous J, Symerský J. Determination of some ampholytic and cationic surfactants by potentiometric titrations based on ion-pair formation. Anal Chim Acta. 1985;177(C):219–23. 10.1016/S0003-2670(00)82955-4.Search in Google Scholar
[63] Pinzauti S, Papeschi G, La Porta E. Potentiometric titration of thiols, cationic surfactants and halides using a solid-state silver-silver sulphide electrode. J Pharm Biomed Anal. 1983;1(1):47–53. 10.1016/0731-7085(83)80007-7.Search in Google Scholar PubMed
[64] Dowle CJ, Cooksey BG, Ottaway JM, Campbell WC. Development of ion-selective electrodes for use in the titration of ionic surfactants in mixed solvent systems. Analyst. 1987;112(9):1299–302. 10.1039/AN9871201299.Search in Google Scholar
[65] Dowle CJ, Cooksey BG, Ottaway JM, Campbell WC. Determination of ionic surfactants by flow injection pseudotitration. Analyst. 1988;113(1):117–9. 10.1039/AN9881300117.Search in Google Scholar
[66] Matešić-Puač R, Stojanović M, Sak-Bosnar M, Hasenay D, Šeruga M. Cationic-surfactant response of ion-selective, N,N,N′,N′-tetracyclohexyl-3-oxapentanediamide-based PVC membrane electrode. Tenside Surfactants Deterg. 2000;37(4):222–8. 10.1515/tsd-2000-370403.Search in Google Scholar
[67] Matešic‐Puac R, Sak‐Bosnar M, Bilic M, Grabaric BS. New ion‐pair based all‐solid‐state surfactant sensitive sensor for potentiometric determination of cationic surfactants. Electroanalysis. 2004;16(10):843–51. 10.1002/elan.200302889.Search in Google Scholar
[68] Kulapina EG, Ovchinskii VA. New modified electrodes for the separate determination of anionic surfactants. J Anal Chem. 2000;55(2):169–74. 10.1007/BF02757745.Search in Google Scholar
[69] Walcarius A. Zeolite-modified electrodes in electroanalytical chemistry. Anal Chim Acta. 1999;384:1–16. Elsevier. 10.1016/S0003-2670(98)00849-6.Search in Google Scholar
[70] Kulapin AI, Arinushkina TV. Solid-contact potentiometric sensors based on ion pairs of cetylpyridinium with dodecylsulfate and tetraphenylborate for various surfactants. J Anal Chem. 2000;55(11):1096–101. 10.1007/BF02757340.Search in Google Scholar
[71] Kulapin AI, Chernova RK, Nikol’skaya EB, Kulapina EG. Modified potentiometric sensors for the separate determination of cationic surfactants. J Anal Chem. 2003;58(3):282–6. 10.1023/A:1022602907924.Search in Google Scholar
[72] Kulapin AI, Chernova RK, Kulapina EG, Mikhaleva NM. Separate detection of homologous surfactants by means of solid-contact unmodified and modified with molecular sieves potentiometric sensors. Talanta. 2005;66(3):619–26. 10.1016/j.talanta.2004.12.017.Search in Google Scholar PubMed
[73] Matysik S, Matysik FM, Einicke WD. A disposable electrode based on zeolite-polymer membranes for potentiometric titrations of ionic surfactants. Sens Actuators B Chem. 2002;85(1–2):104–8. 10.1016/S0925-4005(02)00060-6.Search in Google Scholar
[74] Plesha MA, Van Wie BJ, Mullin JM, Kidwell DA. Measuring quaternary ammonium cleaning agents with ion selective electrodes. Anal Chim Acta. 2006;570(2):186–94. 10.1016/j.aca.2006.04.024.Search in Google Scholar PubMed
[75] Renedo OD, Alonso-Lomillo MA, Martínez MJA. Recent developments in the field of screen-printed electrodes and their related applications. Talanta. 2007;73(2):202–19. 10.1016/j.talanta.2007.03.050.Search in Google Scholar PubMed
[76] Khaled E, Mohamed GG, Awad T. Disposal screen-printed carbon paste electrodes for the potentiometric titration of surfactants. Sens Actuators B Chem. 2008;135(1):74–80. 10.1016/j.snb.2008.07.027.Search in Google Scholar
[77] Mohamed GG, Ali TA, El‐Shahat MF, Al‐Sabagh AM, Migahed MA. New screen‐printed ion‐selective electrodes for potentiometric titration of cetyltrimethylammonium bromide in different civilic media. Electroanalysis. 2010;22(21):2587–99. 10.1002/elan.201000096.Search in Google Scholar
[78] Mohamed GG, Ali TA, El-Shahat MF, Al-Sabagh AM, Migahed MA, Khaled E. Potentiometric determination of cetylpyridinium chloride using a new type of screen-printed ion selective electrodes. Anal Chim Acta. 2010;673(1):79–87. 10.1016/j.aca.2010.05.016.Search in Google Scholar PubMed
[79] Mohamed GG, El-Shahat MF, Al-Sabagh AM, Migahed MA, Ali TA. Septonex-tetraphenylborate screen-printed ion selective electrode for the potentiometric determination of Septonex in pharmaceutical preparations. Analyst. 2011;136(7):1488–95. 10.1039/c0an00662a.Search in Google Scholar PubMed
[80] Ali TA, Eldidamony AM, Mohamed GG, Abdel-Rahman MA. A novel potentiometric sensor for selective determination of dodecyl(2-hydroxyethyl)-dimethylammonium bromide surfactant in environmental polluted samples. Int J Electrochem Sci. 2014;9(8):4158–71. 10.1016/s1452-3981(23)08082-3.Search in Google Scholar
[81] Ali TA, Mohamed GG, El-Dessouky MM, Ragheb RM. Highly selective potentiometric determination of 1-dodecyl-5-methyl-1H-benzo[d][1,2,3]triazol-1-ium bromide surfactant in polluted water samples using 1,4-bis-(8-Mercaptooctyloxy)-benzene ionophore. Int J Electrochem Sci. 2015;10(6):4820–31. 10.1016/s1452-3981(23)06668-3.Search in Google Scholar
[82] Khaled E, Hassan HNA, Abdelaziz MA, El-Attar RO. Novel enzymatic potentiometric approaches for surfactant analysis. Electroanalysis. 2017;29(3):716–21. 10.1002/elan.201600565.Search in Google Scholar
[83] Price R, Wan P. Determination of quaternary ammonium compounds by potentiometric titration with an ionic surfactant electrode: single-laboratory validation. J AOAC Int. 2010;93(5):1542–52. 10.1093/jaoac/93.5.1542.Search in Google Scholar
[84] Gaber M, Abu Shawish HM, Khedr AM, Abed-Almonem KI. Determination of benzalkonium chloride preservative in pharmaceutical formulation of eye and ear drops using new potentiometric sensors. Mater Sci Eng C. 2012;32(8):2299–305. 10.1016/j.msec.2012.06.018.Search in Google Scholar
[85] Issa YM, Mohamed SH, Baset MAEl. Chemically modified carbon paste and membrane sensors for the determination of benzethonium chloride and some anionic surfactants (SLES, SDS, and LABSA): Characterization using SEM and AFM. Talanta. 2016;155:158–67. 10.1016/j.talanta.2016.04.043.Search in Google Scholar PubMed
[86] Crespo GA, Macho S, Rius FX. Ion-selective electrodes using carbon nanotubes as ion-to-electron transducers. Anal Chem. 2008;80(4):1316–22. 10.1021/ac071156l.Search in Google Scholar PubMed
[87] Yin T, Qin W. Applications of nanomaterials in potentiometric sensors. TrAC - Trends Anal Chem. 2013;51:79–86. Elsevier B.V. 10.1016/j.trac.2013.06.009.Search in Google Scholar
[88] Düzgün A, Zelada-Guillén GA, Crespo GA, MacHo S, Riu J, Rius FX. Nanostructured materials in potentiometry. Anal Bioanal Chem. 2011;399(1):171–81. 10.1007/s00216-010-3974-3.Search in Google Scholar PubMed
[89] Hajduković M, Samardžić M, Galović O, Széchenyi A, Sak-Bosnar M. A functionalized nanomaterial based, new, solid state cationic-surfactant-selective sensor with fast response and low noise. Sens Actuators B Chem. 2017;251:795–803. 10.1016/j.snb.2017.05.067.Search in Google Scholar
[90] Rovira M, Lafaye C, Wang S, Fernandez-Sanchez C, Saubade M, Liu SC, et al. Analytical assessment of sodium ISFET based sensors for sweat analysis. Sens Actuators B Chem. 2023;393:134135. 10.1016/j.snb.2023.134135.Search in Google Scholar
[91] Zhang J, Rupakula M, Bellando F, Garcia Cordero E, Longo J, Wildhaber F, et al. Sweat biomarker sensor incorporating picowatt, three-dimensional extended metal gate ion sensitive field effect transistors. ACS Sens. 2019;4(8):2039–47. 10.1021/acssensors.9b00597.Search in Google Scholar PubMed
[92] Alegret S, Bartrolí J, Jimenez-Jorquera C, del Valle M, Dominguez C, Esteve J, et al. Flow-through pH-ISFET + reference-ISE as integrated detector in automated FIA determinations. Sens Actuators B Chem. 1992;7(1–3):555–60. 10.1016/0925-4005(92)80363-3.Search in Google Scholar
[93] Birrell SJ, Hummel JW. Real-time multi ISFET/FIA soil analysis system with automatic sample extraction. Comput Electron Agric. 2001;32(1):45–67. 10.1016/S0168-1699(01)00159-4.Search in Google Scholar
[94] Ipatov A, Abramova N, Bratov A. Autocalibration technique based on SIA and integrated multisensor chip. Talanta. 2008;77(2):581–6. 10.1016/j.talanta.2008.04.013.Search in Google Scholar
[95] Jimenez-Jorquera C, Orozco J, Baldi A. ISFET based microsensors for environmental monitoring. 2010;10(1):61–83. 10.3390/s100100061.Search in Google Scholar PubMed PubMed Central
[96] Campanella L, Aiello L, Colapicchioni C, Tomassetti M. Analysis of cationic surfactants in environmental aqueous matrices by new ISFET devices. Anal Lett. 1997;30(9):1611–29. 10.1080/00032719708001681.Search in Google Scholar
[97] Masadome T, Wakida SI, Kawabata Y, Imato T, Ishibashi N. Contribution of plasticizer to response of surfactant-selective plasticized poly(vinyl chloride) membrane electrode by using ion-sensitive field-effect transistor. Anal Sci. 1992;8(1):89–91. 10.2116/analsci.8.89.Search in Google Scholar
[98] Masadome T, Yada K, Wakida SI. Microfluidic polymer chip integrated with an ISFET detector for cationic surfactant assay in dental rinses. Anal Sci. 2006;22(8):1065–9. 10.2116/analsci.22.1065.Search in Google Scholar PubMed
[99] Casans S, Muñoz DR, Navarro AE, Salazar A. ISFET drawbacks minimization using a novel electronic compensation. Sens Actuators B Chem. 2004;99(1):42–9. 10.1016/j.snb.2003.09.015.Search in Google Scholar
[100] Archbold G, Parra C, Carrillo H, Mouazen AM. Towards the implementation of ISFET sensors for in-situ and real-time chemical analyses in soils: A practical review. Computers Electron Agric. 2023;209:107828. Elsevier B.V. 10.1016/j.compag.2023.107828.Search in Google Scholar
[101] Rousseau CR, Bühlmann P. Calibration-free potentiometric sensing with solid-contact ion-selective electrodes. TrAC - Trends Anal Chem. 2021;140:116277. Elsevier B.V. 10.1016/j.trac.2021.116277.Search in Google Scholar
[102] Cuartero M, Crespo G, Cherubini T, Pankratova N, Confalonieri F, Massa F, et al. In situ detection of macronutrients and chloride in seawater by submersible electrochemical sensors. Anal Chem. 2018;90(7):4702–10. 10.1021/acs.analchem.7b05299.Search in Google Scholar PubMed
[103] Parrilla M, Cuartero M, Crespo GA. Wearable potentiometric ion sensors. TrAC - Trends Anal Chem. 2019;110:303–20. 10.1016/j.trac.2018.11.024.Search in Google Scholar
[104] Zhang Z, Boselli E, Papautsky I. Potentiometric sensor system with self-calibration for long-term, in situ measurements. Chemosensors. 2023;11(1):48. 10.3390/chemosensors11010048.Search in Google Scholar
© 2024 the author(s), published by De Gruyter
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Research Articles
- Microfabricated potentiometric sensor based on a carbon nanotube transducer layer for selective Bosentan determination
- A novel Six Sigma approach and eco-friendly RP-HPLC technique for determination of pimavanserin and its degraded products: Application of Box–Behnken design
- Enantiomeric separation of four pairs of alkaloids by using a C18 column tandem polysaccharide-based chiral column
- Sustainable HPLC technique for measurement of antidiabetic drugs: Appraisal of green and white metrics, content uniformity, and in vitro dissolution
- Quantitative analysis of mixed lipid nanostructures in rat skin by HPLC-MS
- Review Articles
- Recent advance in electrochemical immunosensors for lung cancer biomarkers sensing
- Latest trends in honey contaminant analysis, challenges, and opportunities for green chemistry development
- Quantitative methods in the analysis of clozapine in human matrices – A scoping review
- Review of potentiometric determination of cationic surfactants
- Surface-enhanced Raman spectroscopy in forensic analysis
- Special Issue: Greener Analytical Methods - Part II
- Greenness of dispersive microextraction using molecularly imprinted polymers
- KPI-based standards benchmarking for the preference of different analytical approaches developed for simultaneous determination of ciprofloxacin and hydrocortisone: A SWOT case study
Articles in the same Issue
- Research Articles
- Microfabricated potentiometric sensor based on a carbon nanotube transducer layer for selective Bosentan determination
- A novel Six Sigma approach and eco-friendly RP-HPLC technique for determination of pimavanserin and its degraded products: Application of Box–Behnken design
- Enantiomeric separation of four pairs of alkaloids by using a C18 column tandem polysaccharide-based chiral column
- Sustainable HPLC technique for measurement of antidiabetic drugs: Appraisal of green and white metrics, content uniformity, and in vitro dissolution
- Quantitative analysis of mixed lipid nanostructures in rat skin by HPLC-MS
- Review Articles
- Recent advance in electrochemical immunosensors for lung cancer biomarkers sensing
- Latest trends in honey contaminant analysis, challenges, and opportunities for green chemistry development
- Quantitative methods in the analysis of clozapine in human matrices – A scoping review
- Review of potentiometric determination of cationic surfactants
- Surface-enhanced Raman spectroscopy in forensic analysis
- Special Issue: Greener Analytical Methods - Part II
- Greenness of dispersive microextraction using molecularly imprinted polymers
- KPI-based standards benchmarking for the preference of different analytical approaches developed for simultaneous determination of ciprofloxacin and hydrocortisone: A SWOT case study

