Abstract
Regenerative rehabilitation is a novel and rapidly developing multidisciplinary field that converges regenerative medicine and rehabilitation science, aiming to maximize the functions of disabled patients and their independence. While regenerative medicine provides state-of-the-art technologies that shed light on difficult-to-treated diseases, regenerative rehabilitation offers rehabilitation interventions to improve the positive effects of regenerative medicine. However, regenerative scientists and rehabilitation professionals focus on their aspects without enough exposure to advances in each other’s field. This disconnect has impeded the development of this field. Therefore, this review first introduces cutting-edge technologies such as stem cell technology, tissue engineering, biomaterial science, gene editing, and computer sciences that promote the progress pace of regenerative medicine, followed by a summary of preclinical studies and examples of clinical investigations that integrate rehabilitative methodologies into regenerative medicine. Then, challenges in this field are discussed, and possible solutions are provided for future directions. We aim to provide a platform for regenerative and rehabilitative professionals and clinicians in other areas to better understand the progress of regenerative rehabilitation, thus contributing to the clinical translation and management of innovative and reliable therapies.
Introduction
Regenerative rehabilitation is a burgeoning interdisciplinary field that converges regenerative medicine and rehabilitation sciences [1]. This convergence aims to optimize functional recovery and patient outcomes by integrating biological technologies focusing on tissue repair or replacement and rehabilitative methods such as mechanical, electrical, and thermal stimuli [2]. The goal is to restore the functions of disabled patients through tissue regeneration and repair, thereby improving their ability to daily living and quality of life (QoL). The first person who introduced regenerative medicine into the Department of Rehabilitation Medicine was Dr. Joel Stein at Columbia University in New York [3]. Later, the University of Washington and the University of Pittsburgh in the United States also integrated regenerative medicine technology into rehabilitation medicine [3]. Currently, most traditional rehabilitation professionals focus on the body’s response to physical means and often ignore cellular and molecular changes [1, 2]. In contrast, specialists in regenerative medicine usually pay more attention to the use of advanced technologies and changes occurring at the molecular, cell, and tissue levels [1, 2]. It is easy for regenerative scientists to ignore the clinical translation and effects of clinical methods. At the same time, scientists in the rehabilitation field are unaware of the latest advances in regenerative medicine, and vice versa. Therefore, a better understanding of innovative and advanced technologies in regenerative medicine and their combination with rehabilitative methodologies will speed up the progress pace of this novel field, contributing to clinical translation and providing reliable therapies for patients [1, 2].
To meet this requirement, our review first focuses on cutting-edge regenerative technologies and their applications in regenerative medicine. These technologies involve stem cell technology, tissue engineering, biomaterial science, gene editing, and computer sciences. Then, we introduce the integration of rehabilitation sciences into regenerative medicine and summarize up-to-date preclinical and clinical trials in regenerative medicine and rehabilitative approaches. Since regenerative medicine is mainly based on cell and tissue biology to restore tissue structure and function, we focus on stem cell therapy as an approach to regenerative medicine in this review. In the end, we conclude challenges and future directions in this field. This review addresses the need to integrate regenerative medicine and rehabilitative sciences, promote their convergence, and help the clinical translation of scientific discoveries in regenerative rehabilitation.
State-of-the-art technologies in regenerative medicine
As our population ages, the incidence of challenging diseases such as Alzheimer’s disease, Parkinson’s disease, heart failure (HF), diabetes, cancer, immune disorders, and age-related macular degeneration (AMD) is projected to increase. This aging demographic presents a growing challenge for conventional surgical techniques and pharmaceutical interventions. Consequently, regenerative medicine has garnered attention, as it encompasses the body’s natural processes of repairing and replacing damaged or lost tissues and organs to restore their structure and function. Regenerative medicine, as a medical discipline, aims to develop techniques for replacing, repairing, or regenerating impaired cells, tissues, or organs to address and potentially prevent a range of illnesses [1, 4]. It integrates cutting-edge bioengineering technologies, primarily centered around stem cell therapy, materials science, tissue and organ engineering, genetic technology, and computer science [1, 4, 5] (Figure 1).
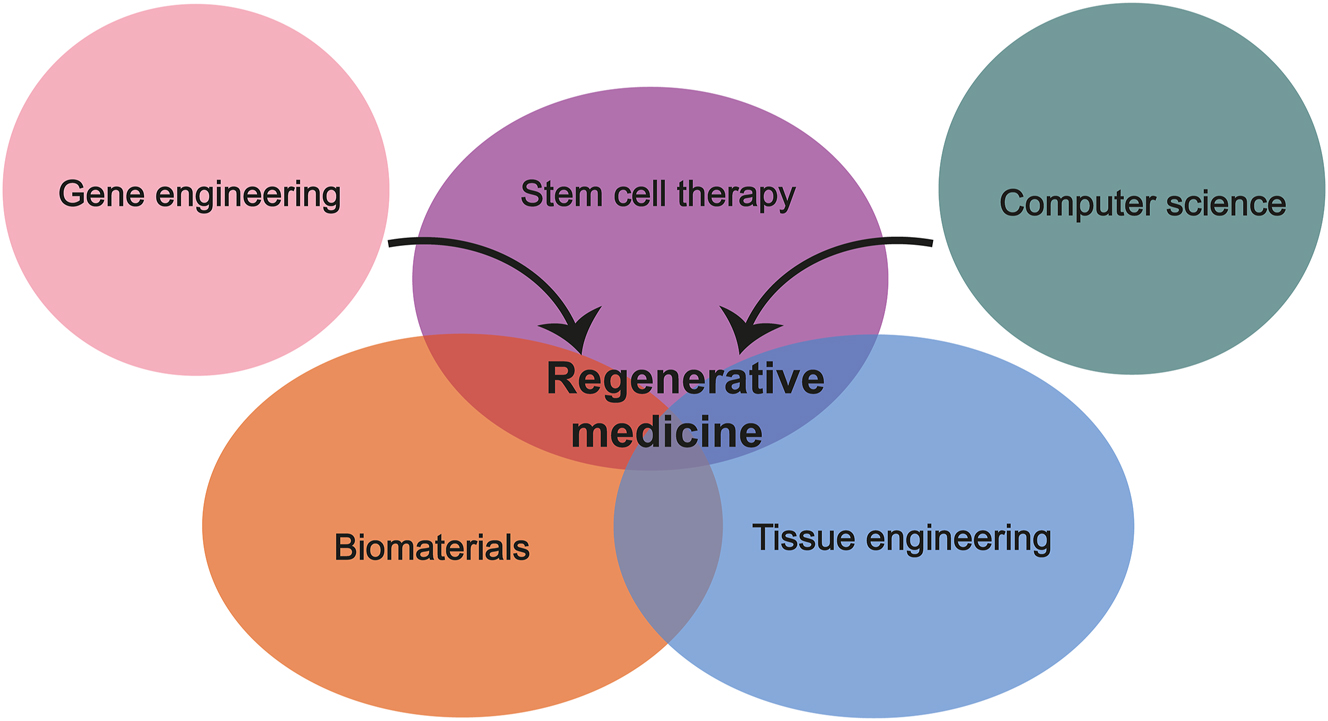
State-of-the-art technologies in regenerative medicine. Regenerative medicine combines modern bioengineering technologies, mainly involving stem cell therapy, biomaterial science, tissue engineering, genetic technology, and computer science.
Stem cell therapy
Regenerative medicine is mainly based on cell and tissue biology to restore tissue structure and function. Stem cells are undifferentiated cells that have the potential to renew themselves, develop into other cell types, and grow into tissue/organs [6]. Stem cell-based therapy aims to replace dysfunctional or injured tissue with new, healthy, functional cells or their derivatives to restore corresponding cell functions [7]. Mechanisms underlying regeneration involve inflammation inhibition [8], reduction in apoptosis [9], angiogenesis stimulation [10], and cell recruitment [11]. More than 6,000 clinical trials of stem cells worldwide are expected to solve major medical problems by repairing impaired cellular functions or pathological tissues. Preclinical and clinical investigations showed that stem cell therapy is effective in both acute and chronic diseases [7, 12, 13]. For example, transplantation of hematopoietic stem cells (HSCs) has shown positive therapeutic effects on immunodeficiency disorders [14], autoimmune neurological disorders [15], [16], [17], and blood system diseases such as multiple myeloma [18, 19] and thalassemia central [20, 21]. Application of induced pluripotent stem cells (iPSCs) has sound effects on AMD [22], [23], [24], Parkinson’s syndrome [25, 26], and other diseases [27], [28], [29]. Mesenchymal stem cells (MSCs) have regenerative properties and immunomodulatory and anti-inflammatory functions [30]. They have been used to treat immune diseases, such as rheumatoid arthritis, osteoarthritis (OA), and graft-versus-host disease, which showed promising results [30, 31]. Many companies and scientific research institutions worldwide have joined the drug development of stem cells. Several drugs have been successfully brought to market, including AstroStem, an autologous MSC drug used to treat Alzheimer’s disease [32], and Strimvelis, an autologous HSC drug indicated for the treatment of severe combined immunodeficiency caused by adenosine deaminase deficiency (ADA-SCID) [32].
Common challenges in cellular therapies involve the proliferation and survival of stem cells after transplantation, cell migration to the targeted sites, and successful cell differentiation into the targeted tissues [33]. Various factors such as diabetes [34, 35], age [36], and gender [35, 37] are important factors that affect the regeneration potential of donor stem cells. For example, a study conducted by Deasy et al. found that the myogenic capability of muscle-derived stem cells (MDSCs) from male mice is much lower than that of those from female counterparts [38]. This difference between genders could be due to lower stress tolerance of cells from males than those from females. Increasing cellular resistance to stress could improve the transplantation efficiency of donor cells [38]. Another study showed that transplantation of MDSCs from mice had better improvement of cardiac function than myoblasts in a mouse model of acute myocardial infarction (MI) [39]. The higher regeneration ability of MDSCs is related to their higher levels of antioxidants including glutathione (GSH) and superoxide dismutase [39]. Transplantation of hypoxia-conditioned bone marrow-derived MSCs could also promote the formation of new vessels and increase cell engraftment [40]. In addition, endothelial progenitor cells with diabetes exhibit significantly reduced angiogenesis than individuals with diabetes [34]. In particular, the host microenvironment or niche has been identified as a critical factor. It has been shown that implanted embryonic stem cells (ESCs) exhibit an aged phenotype after implantation into an aged environment [34, 41, 42]. Conversely, niche modification of the aged muscle environment through exposure to a young systemic environment rejuvenates the capacity for regenerative growth in ancient cells [42, 43]. Although many researchers are adopting a cell transplantation strategy for tissue repair, an alternative approach is to manipulate the stem cell microenvironment, or niche, to simplify repair by endogenous stem cells [44]. The microenvironment is highly active, with multiple opportunities for intervention, which include the administration of small molecules, biologics, gene therapy, and growth factors or biomaterials that target specific aspects of the niche, such as cell-cell and cell-extracellular matrix (ECM) interactions, to stimulate the survival and differentiation of stem cells, or to cause the reversion of differentiated cells to stem cells. Nevertheless, there are several challenges in targeting the niche therapeutically. These methods have limitations in terms of cost, safety, and long-term quality of repair, and avoiding treatment-related adverse effects such as carcinogenesis may restrict their use in medical applications [45]. Noninvasive, low-cost techniques to improve the stem cell microenvironment are clinically significant.
Tissue engineering and biomaterials
However, to completely restore functions of missing or impaired tissues to the pre-injury or diseased state is the ultimate goal of regenerative medicine, which indicates that only the regeneration or replacement of muscle, bone, nerve, vasculature, or skin is not enough, but correct and functional integration of these tissues is required [46, 47]. Tissue/organ engineering and biomedical materials are extensively studied with stem cell technologies to gain intact and functional tissues or organs. Stem cell technology provides the content, biomaterials serve as the scaffold, and tissue/organ engineering covers the content and scaffold to establish implantable and synthetic tissue substitutes. A scaffold is an artificial three-dimensional (3D) structure that provides a platform for living tissue cells and growth factors and can be implanted into an organism [48]. Scaffold materials must have good tissue compatibility and can be gradually degraded and absorbed by the human body [49]. They should also be suitable for cells’ adhesion, proliferation, differentiation, and organization [50]. Since the innovative development of the first bioprinter in the early 21st century, 3D bioprinting has been applied to realize tissue engineering via layer-by-layer manufacturing process of biomaterials, ECM, growth factors, and live cells to form complex biological structures in the body [51]. Bioprinting technology to create tissue-like materials is called bio-fabrication [52]. It can achieve high resolution, complexity, high yield rate, and personalization [53]. Combining cell technology, biomaterials, and 3D bioprinting provides a promising possibility for tissue defects and organ supply. For example, researchers at Northwestern University have 3D-printed hydrogel scaffolds that can sustain the survival of ovarian follicles by adjusting the scaffold-follicle interaction [54]. After being transplanted into sterilized mice, this bioprosthetic ovary became vascularized and restored ovarian function in these mice [54]. A group in the United States developed a tough hydrogel for bioprinting vascular conduits to create veinous and arterial vessel-like structures harboring corresponding functions [55]. It is worth noting that these studies are still proof-of-concept studies and are a long way from human clinical trials. 3D bioprinting technology is a rapidly developing field, and this type of research aims to create correctly structured, functional, survivable, and implantable organs and tissues for use in regenerative medicine.
However, there are challenges in applying tissue engineering in regenerative medicine. These include proper biomaterials, angiogenesis of transplanted tissues in the host, cell expansion and differentiation, and so on [56, 57]. For example, biomaterials are required to mimic the ECM of tissues of interest, exhibit proper mechanical properties, and support cellular growth, proliferation, and differentiation [57, 58]. These materials should also be biocompatible and biodegradable [57, 58]. Additionally, developing strategies to promote the vascularization of implanted tissues within the host poses significant challenges. For this purpose, drug delivery systems should be developed to precisely control the dosage, location, and duration of factor release required for angiogenesis [57, 58]. Thirdly, it is a demanding task to employ techniques to differentiate cells into heterogeneous cell populations while preventing unwanted cells [59]. Fourthly, methodologies for accurately organizing structures that possess mechanical properties and functions resembling those of native complex tissues remain formidable challenges [60].
Genetic engineering
For genetic diseases, stem cell therapy is supposed to repair cells or tissues influenced by the genetic mutation. Due to congenital abnormalities in patients’ cells, simple replacement of autologous stem cells can not significantly improve symptoms in some diseases. Allogeneic stem cells with normal function have the possibility of improving symptoms. However, a lack of matched donors and a risk of graft vs. host diseases are common problems for allogeneic stem cell therapy [61]. Therefore, genetic engineering that creates deletions, insertions, or replacements of desired sequences on the genome provides a powerful technology to solve problems of autologous or allogeneic stem cell therapy. CRISPR/Cas9-mediated gene editing technology has apparent advantages over first-generation zinc-finger nuclease (ZFN) and second-generation transcription activator-like effector nuclease (TALEN) editing technologies [62]. CRISPR/Cas9 plays an essential role in stem cell research, such as disease modeling to explore new therapeutic tools and developing stem cell drugs edited by CRISPR/Cas9 [63]. Multiple clinical trials have used CRISPR/Cas9 to gene edit HSCs to treat blood or immune system diseases, including sickle cell disease, β-thalassemia, myeloid malignancies, and acute myeloid leukemia [61, 64], [65], [66], [67], [68]. Compared with mature cells, iPSCs are more accessible to genetically modify and have substantial expansion and multi-lineage differentiation capabilities. These properties facilitate the development of relatively uniform, universal, off-the-shelf cellular drugs. Regarding drug quality control and development costs, using gene-edited iPSCs for cell drug development will become a general trend. Fate Therapeutics’ cell drug FT819 is a revolutionary therapy developed via inserting a novel 1XX CAR targeting CD19 into the T cell receptor alpha constant locus of a master iPSC line, which is then differentiated to produce universal CAR-T cells that are not subject to patient restrictions [69]. It is also the first off-the-shelf, iPSC-derived CAR-T cell drug [69]. In addition to HSC and iPSC therapy, CRISPR/Cas9 is widely used in other stem cell therapies, such as epidermal stem cells [70, 71] and MSCs [72, 73]. The application of CRISPR/Cas9 in stem cell therapy can potentially improve the therapeutic effect, increase the scope of treatable diseases, and positively impact safety and security.
While genetic editing holds significant promise for enhancing tissue repair through precise modifications of cellular genetics, several challenges must be addressed. Firstly, genetic engineering techniques such as CRISPR/Cas9 are plagued by off-target effects, wherein genetic modifications may occur at unintended genetic sites resembling the targeted sites [74]. Although methods for assessing off-target effects of CRISPR/Cas9 have advanced rapidly in the past decade, limitations persist in balancing the accuracy and sensitivity of these techniques. Direct assessment of off-target effects in vivo and in patients poses challenges. Overcoming these challenges would pave the way for the development of next-generation genome editing tools that expedite the advancement of gene therapy [74]. Secondly, achieving effective and targeted delivery of genetic editing tools to the desired cells or tissues is paramount. Developing techniques to facilitate the penetration of these tools through membranes or the blood-brain barrier (BBB) remains a formidable challenge [75]. Thirdly, it is imperative to ensure the long-term stability and safety of genetically edited cells and tissues for the success of regenerative medicine approaches [76, 77]. This entails vigilant monitoring of the persistence of edited cells, thorough assessment of potential genotoxicity or oncogenic risks, and diligent addressing of concerns related to insertional mutagenesis, all of which are crucial considerations for clinical applications.
Computer sciences
Although stem cell therapy seems simple in theory, describing all cell products in depth is difficult because cells are unstable [78]. Along with these obstacles, artificial intelligence (AI) such as data mining, machine learning, and convolutional neural networks enter stem cell research, which is studied to achieve automated and dynamic adjustment of the stem cell differentiation process, maintain the stability of pluripotent stem cell products between different batches, and predict prognosis of stem cell therapies [79], [80], [81], [82]. For instance, The Nanba group in Japan has released a deep learning-based automated cell tracking (DeepACT) strategy that can be used to identify cultured human keratinocyte stem cells and monitor individual keratinocytes, which enables non-invasive quality control of stem cells and reduce variability in pluripotent stem cell differentiation system [83]. Deep learning has been applied to predict the differentiation fate of NSCs at the early stage [84]. If the differentiation type of cells could be expected from the initial conditions, it would be possible to increase the yield of produced cells via controlling initial conditions to accelerate the process of stem cell application. Recent studies show that computer science is changing the future of stem cell research and applications. This advanced technology facilitates the detection of a reliable framework for iPSC colony classification, identification of cell morphology and undescribed morphological features in cancer stem cells, and accurate prediction of drug effects [78]. Nevertheless, AI is still in a relatively primitive stage nowadays. More investigations are needed in the future to summarize the detailed benefits and risks of specific diseases related to AI and stem cell treatments.
Regenerative medicine encompasses a wide array of datasets, ranging from genomic information and cellular behavior to tissue characteristics and patient clinical data. However, integrating and standardizing these vast and diverse datasets to facilitate analysis and interpretation presents significant challenges [85]. Moreover, the development of sophisticated computational models and simulation techniques to predict complex biological systems is hindered by the intricate nature of tissue regeneration, disease progression, and the multitude of cellular and molecular pathways involved [85]. In the realm of regenerative medicine, visualizing tissue structure, function, and dynamics is essential [86]. Medical image analysis is pivotal in the field of regenerative medicine. However, progress in this area is impeded by various challenges. These include adapting analysis technologies to suit clinical tasks, developing algorithms capable of handling heterogeneous images, extracting relevant features, and mitigating issues such as image noise, artifacts, and variability [86].
Progress of rehabilitative strategies in regenerative medicine
Stem cell therapy and rehabilitation: emerging partnerships
Rehabilitation is recommended for many conditions, targeting to achieve two primary aims: promoting tissue healing and enhancing the overall function of the surrounding tissues as the injured area undergoes repair. Furthermore, rehabilitation modalities are commonly prescribed for various conditions after diagnosis, suggesting that after any potential regenerative medicine intervention, rehabilitation is likely to be a fundamental part of the treatment continuum [87]. Rehabilitation-based programs are firmly established as effective therapeutic interventions with extensive applications across numerous medical fields. These rehabilitation interventions can range from standard exercise to directed and supervised mechanotherapy, offering external stimulation such as electrical stimulation, low-intensity pulsed ultrasound, and pulsed electromagnetic field therapy [88–91] (Figure 2). The benefits are likely to enhance functional outcomes and biomechanics following the application of regenerative medicine, reduce patients’ disabilities, and improve QoL, combining with the application of regenerative medicine. For instance, exercise has been revealed to significantly reduce the risk of many chronic diseases, including diabetes, Alzheimer’s disease, cardiovascular diseases, various cancers, osteoporosis, arthritis, and many other disorders [92]. The typical approach to stem cell transplantation has been introducing the cells into the host and hoping for the best outcomes. However, effective stem cell therapy depends on cell replacement and proper functional integration, connection, and differentiation into the host environment. Recent research shows that rehabilitation interventions can have direct regenerative benefits, such as the direction of stem cell differentiation, mobilization of stem cells into circulation, and enhancement of secreted regenerative factors [93]. Also, many of the characteristics controlling the functioning of cells, such as cellular tolerance to physiological stress [94, 95], division [96], survival [97], and the microenvironment, are flexible through the use of targeted loading approaches. Mechanical stimulation includes stretching, compression, and fluid shear stress that modifies the cellular microenvironment [98], and the application of forces can help donor cells integrate into the body beneficially and effectively [99]. Using target mechanical stimuli offers a method for inaugurating communication with transplanted cells. This methodology enables researchers to guide these cells to function in vivo as initially intended. It is noteworthy that stem cells exhibit responsiveness to external mechanical forces both in controlled in vitro environments and within the complex milieu of in vivo settings [100, 101]. Animals housed in an Enriched Environment (EE) (i.e., increased sensory input and social interaction) exhibited augmented survival of newborn hippocampal neurons over time [102]. In addition, voluntary exercise resulted in a temporally noteworthy increase in the generation of new hippocampal neurons and enhanced their overall survival [103]. Moreover, exercise and EE have been well-studied in the context of stroke [104]. When compared to standard housing rats, EE strategies result in increased neuronal survival in the striatum and cortex, improved cerebral blood flow, increased angiogenesis, increased endogenous progenitor cell proliferation, increased endogenous neuronal differentiation in the ischemic regions, increased axonal guidance proteins, and reduced ischemic BBB capillary damage [105]. Many molecular mechanisms induced by EE supported these incredible findings. Angiogenesis and the rescue of the neurovascular unit were portrayed through signaling pathways such as phosphorylated PI3K, AKT, and GSK-3 but reduced phosphorylated β-catenin [105]. The positive outcomes of both EE and stem cell transplantation arise from the training of the stem cell graft to seamlessly merge with the host brain. Stem cell transplantation offers many therapeutic advantages, such as regulating the immune system, rescuing dying cells, stimulating neurogenesis, and enhancing angiogenesis. These advantages are combined with the positive effects of environmental enrichment and regular exercise [106, 107]. Rehabilitation represents a natural synergy with the capacity to exert a significant clinical impact. Rehabilitation and target-directed activity-based efforts post-cell transplantation increase and facilitate proper connectivity/integration of regenerative approaches. Combining rehabilitation approaches with stem cell therapy provides promising therapeutic strategies for hard-to-treated diseases. The standard procedure shown in Figure 3 involves subjecting stem cells to various stimulation techniques, such as mechanical, electrical, and thermal, within a laboratory setting to enhance their viability, growth, and differentiation. After the transplantation of the cells, patients are also subjected to similar stimulation techniques. Additionally, physical activities, assistive technologies, and EE are designed to help individuals increase their activity levels independently and improve their overall QoL. However, exploring the mechanism of combining rehabilitation approaches with stem cell therapy in various diseases is crucial for a deeper understanding of how this synergy works and how it can be optimized to maximize patient outcomes. To date, many preclinical studies have combined stem cell therapy with rehabilitation. Some examples of how incorporating rehabilitation may benefit developing regenerative medicine strategies will be addressed.
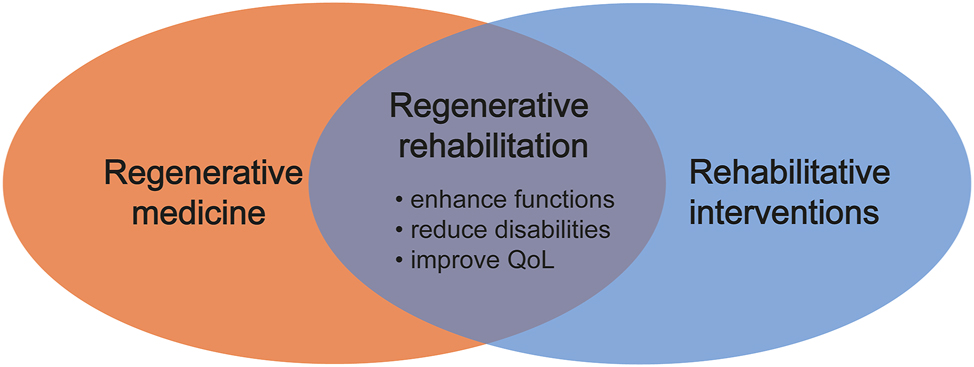
Converging regenerative medicine and rehabilitation. Rehabilitation interventions can range from standard exercise to mechanotherapy, offering external stimulation such as electrical stimulation, low-intensity pulsed ultrasound, and pulsed electromagnetic field therapy. Combining rehabilitative interventions with regenerative medicine will likely enhance functional outcomes, reduce patients’ disabilities, and improve QoL. QoL, quality of life.
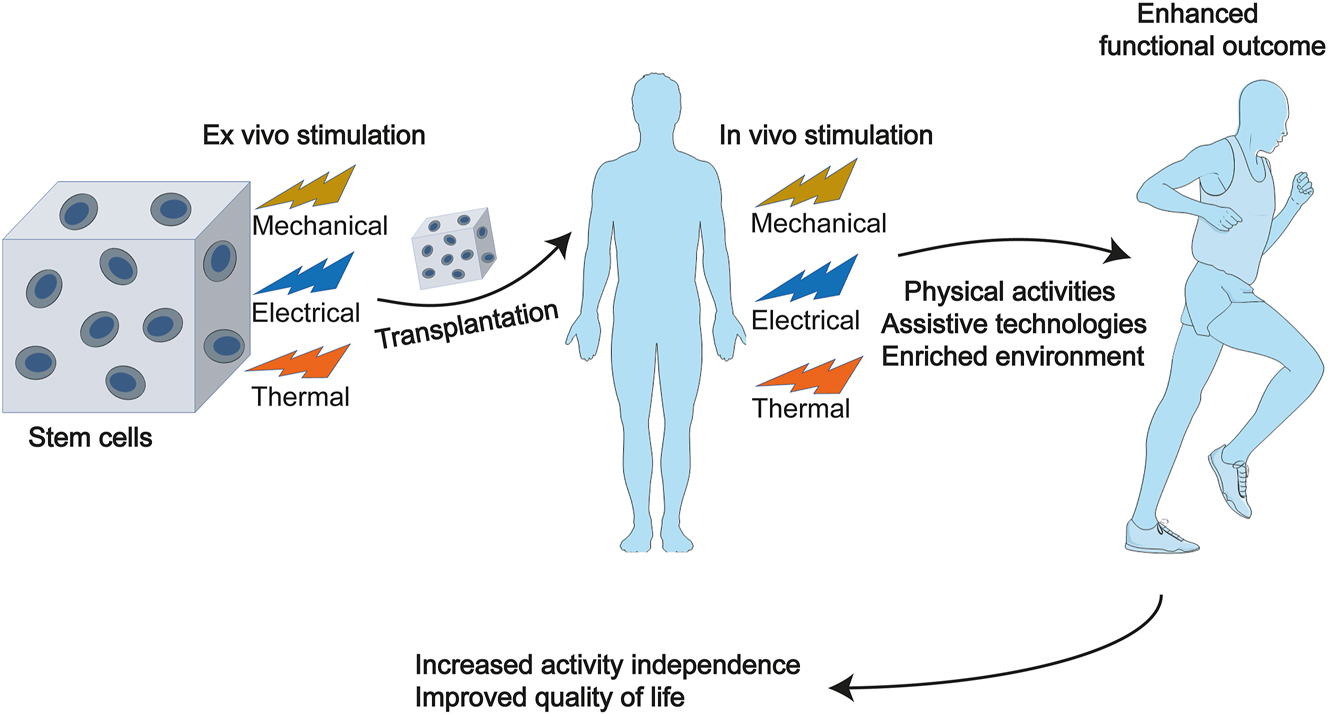
Stem cell therapy and rehabilitation. Combining rehabilitation approaches with stem cell therapy offers promising therapeutic strategies to increase activity independence and improve patients’ quality of life. Ex vivo stimulation to stem cells involves mechanical, electrical, thermal stimulations, and so on. After cell transplantation, in addition to these stimuli, physical activities, assistive technologies, and an enriched environment need to be designed to help enhance functional outcomes. Parts of the figure were drawn by using pictures from Servier Medical Art. Servier Medical Art by Servier is licensed under a Creative Commons Attribution 3.0 Unported License (https://creativecommons.org/licenses/by/3.0/).
Regenerative rehabilitation for stroke recovery
Stroke is a cerebrovascular disease that occurs when the brain’s blood flow is disrupted or when bleeding occurs in brain tissue, resulting in loss of brain function [108, 109]. Stroke is a life-threatening condition that occurs every 2 s, with individuals dying every 6 s and 15 million occurring each year. After a stroke, about 40 % of patients have a functional disability, and 15 %–30 % have severe motor, sensory, cognitive, perceptual, and/or language impairments [108, 109]. There is currently a lack of treatment options for stroke or traumatic brain injury that can restore cognitive and motor function.
Regenerative and rehabilitation medicine have accompanied promising new methods for mediating recovery in the central nervous system (CNS), the most salient of which are rehabilitation and stem cell therapies that, when combined, result in more obvious recovery than one approach alone [110, 111]. The benefits of combining rehabilitation and regeneration medicine have been shown in Figure 4. Regenerative medicine, notably MSC therapy, promotes cell regeneration, the recruitment of endogenous neural progenitors, anti-inflammatory characteristics, and circuitry repair. Several studies investigated the effects of combining intravenous cell transplantation with rehabilitation. For example, in a rat model of Middle Cerebral Artery Occlusion (MCAO), it was shown that a combination of MSC transplantation and treadmill exercise had neuroprotective effects, evidenced by the decreased number of apoptotic cells, increased motor function in the dual-treated group than in the single-treated groups [112]. Furthermore, treadmill exercise significantly increased the quantity of engrafted MSCs and inhibited cell death in both host and donor cells in the combination group. This anti-apoptotic effect is thought to be caused by the upregulation of two cell death regulators, survivin and bcl-2 [112]. In a similar trial, Sasaki et al. found that combination therapy had a synergistic impact, significantly decreasing infarct volume, increasing corpus callosum thickness, and triggering synaptogenesis, resulting in improved motor function recovery [113]. Further study revealed that combining mild therapeutic hypothermia (33 °C) with rat adipose-derived stem cells (rADSCs) enhanced functional recovery additively or synergistically by reducing neuronal apoptosis and gliosis, promoting angiogenesis, and decreasing innate immune cell infiltration in MCAO model rats [114]. Several researches investigated the effects of combining local cell transplantation into the cerebral parenchyma with treadmill exercise or EE treatments. Seo et al. evaluated the impact of combination treatment transplantation of human adipose stem cells hASCs to the striatum and EE in hypoxic-ischemic brain injury model mice [115]. Their study showed that combining therapies promoted functional recovery by enhancing engrafting and neural differentiation of transplanted hASCs, inducing intrinsic neurogenesis, and activating astrocytes through fibroblast growth factor-2 (FGF-2). A more detailed study of the same group found that therapy combining hASC transplantation and EE promoted functional recovery through synergistic upregulation of angiogenic factors such as FGF-2, vascular cell adhesion protein-1 (VCAM-1), matrix metalloproteinase-2 (MMP-2) released from activated astrocytes, and concurrent intrinsic angiogenesis [116]. Another study evaluated the effects of EE in combination therapy using animal models of cerebral infarction and transplantable cells. In MCAO model rats, mouse subventricular zone (SVZ)-derived stem cells were implanted into the sensory cortex and striatum [117]. Although EE dramatically improved the distance of stem cell migration towards the infarct site, leading to earlier motor function recovery, the study did not include a treatment group that received EE alone. Therefore, it remains unclear whether the combination of cell transplantation and EE has a synergistic effect. While animal studies on the combination of rehabilitation and stem cell therapy look promising, this combination is relatively unexplored. Verifying the effects of combining two treatments is only possible if the therapeutic effects of each are independently assured. Further comprehensive investigation and research are necessary to deepen our understanding.
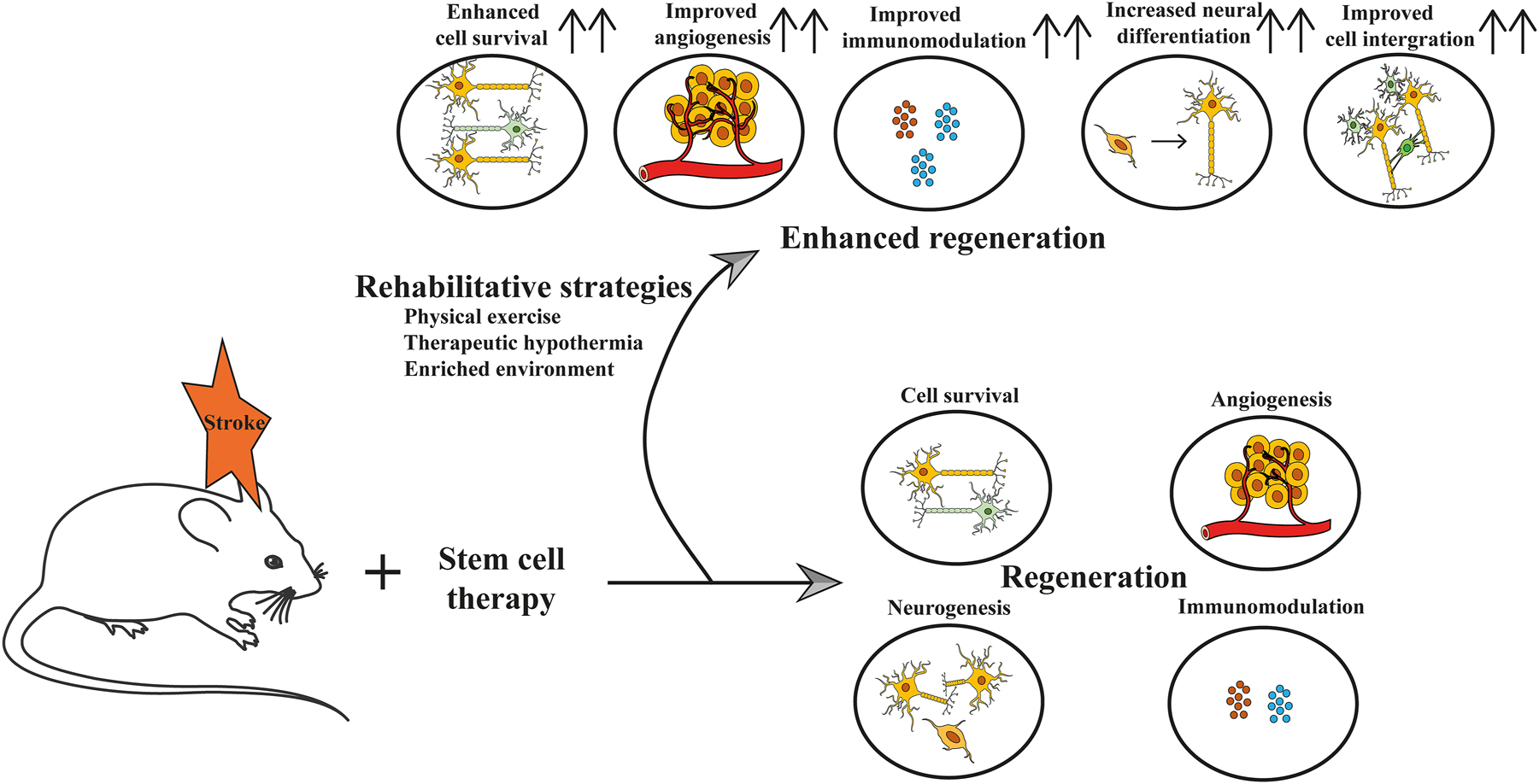
Regenerative rehabilitation for stroke. In studies using rodents to model stroke, stem cell therapy has been shown to promote cell survival, neurogenesis, angiogenesis, and modulate immune response. There are also rehabilitative strategies such as physical exercise, therapeutic hypothermia, and enriched environments that can improve these effects, and increase neural differentiation and cell integration into the local environment.
Regenerative rehabilitation for spinal cord injury
Spinal cord injury (SCI) is a serious condition with several sequelae indicated by motor, sensory, and autonomic disorders that have a significant impact on patients’ psychosocial lives and quality of life [118]. Stem cell-based regenerative therapy has opened an opportunity for functional recovery of patients with SCI. Significant therapeutic results with different cell sources, including neural stem/progenitor cells (NS/PCs), MSCs, and olfactory ensheathing cells (OECs), have been described clinically and preclinically in the acute and subacute phases [119]. Various therapeutic mechanisms, such as transplanted cell-mediated neuronal replacement, remyelination, and trophic support, have been determined by researchers, which further induce tissue protection and enhance neural plasticity [120, 121]. Regenerative rehabilitation has attracted wide attention because of its effects, feasibility, and non-invasiveness. Rehabilitation science and medicine include treatments incorporating mechanical stimuli, including rehabilitative training, joint mobilization, tissue loading, stretching, and traction, and physical stimuli, including ultrasound stimulation, electrical stimulation, magnetic stimulation, and temperature gradients [93]. Notably, the status of regenerative rehabilitation differs between clinical and preclinical studies in numerous respects. First, individuals with SCI habitually undergo rehabilitation in clinical settings and frequently continue rehabilitative training to preserve their mobility, joint range of motion, muscle strength, and activities of daily living. Second, rehabilitation is possible in many clinical settings and is combined with stem cell therapies in clinical trials. Third, it is relatively easy to plan appropriate training for patients according to their impairments because there is much knowledge of SCI rehabilitation, and rehabilitation therapists and doctors can receive patient feedback. On the other hand, in preclinical studies, rehabilitation training is less feasible, very time-consuming, and expensive [122], and laboratories investigating regenerative treatment often lack the capabilities to achieve rehabilitative techniques [123]. Likewise, the appropriate training and its load have not been structurally validated or standardized among research groups. Converging stem cell therapy and rehabilitation medicine have shown benefits for SCI through various biological processes (Figure 5). The following rehabilitative training approaches have been applied in combination with stem cell therapies to animal models of SCI regardless of chronicity: bipedal treadmill training with body-weight support. The study focused on mouse thoracic chronic SCI and the results found that NS/PC transplantation alone led to limited improvements. Notably, significant locomotor recovery was observed only in the group that received both NS/PC transplantation and treadmill training. Further investigation showed that NS/PC transplantation improved spinal conductivity and central pattern generator activity, while treadmill training improved inhibitory motor control. This combined therapy synergistically improved these aspects and promoted neuronal differentiation of transplanted cells, suggesting that rehabilitative treatment can enhance locomotor recovery even in chronic SCI cases [124]. Another recent study examined the effect of treadmill training on transplanted NPCs following SCI injury in rats. The findings indicate that combining stem cell therapy with treadmill training enhances NPC survival, differentiation into neurons and oligodendrocytes, myelination, and regeneration. This combination approach increases functional recovery, emphasizing the significance of synergistic treatments for SCI [125]. Active quadrupedal training, the study investigated the combined effects of NPC transplants and intensive locomotor training (ILT) on neuropathic pain in rats with SCI. They found that both NPC and ILT transplants alone could alleviate pain. the combination of NPC and ILT significantly improved pain reduction, including allodynia and hyperalgesia. This combined approach also decreases pro-inflammatory markers and spinal damage, with GABAergic neuronal density nearly restored to normal levels. The results propose that combining ILT with NPC transplants may offer a more efficient way to reduce neuropathic pain and promote spinal cord recovery after SCI [126]. On the other hand, the following physical medicine modalities have been applied using apparatus resembling epidural electrical stimulation in clinical use. Thornton et al. examined the effects of combining olfactory ensheathing cell (OEC) transplantation and epidural electrical stimulation during a motor task for SCI in rats. The OEC-treated group showed increased axon crossing at the injury site and enhanced connectivity, indicating potential axon regeneration and circuit sparing. However, there were no observed behavioral variations in this small-scale study. In summary, combining OEC transplantation, stimulation, and motor activity improved axonal growth and connectivity across the injury site but did not lead to direct behavioral improvements [127]. Further study demonstrated that intermittent repetitive transcranial magnetic stimulation (rTMS) combined with human umbilical cord blood mesenchymal stem cell (hUCB-MSC) transplantation could promote motor functions in a rat model of SCI, which was supported by the Basso, Beattie, and Bresnahan (BBB) locomotor assessments, motor evoked potentials (MEPs) and reduced neuron apoptosis [128]. When hUCB-MSC transplantation and rTMS were combined, there was a significant increase in positive cells for neural stem cell markers, such as nestin, BrdU, and Tuj1, and a decrease in markers, such as Ng2+ and GFAP. Furthermore, the levels of growth factors bFGF and EGF were upregulated. Overall, the combination of rTMS and hUCB-MSC transplantation effectively reduced SCI-induced neural stem cell apoptosis and enhanced motor function in rats [128]. In addition, when hADSC therapy was combined with low-level laser, the motor function was significantly improved, and allodynia and hyperalgesia were ameliorated in rats with SCI [129]. These combined treatments also increased the expression levels of GDNF mRNA, and the number of axons near the site of SCI [129]. In conclusion, existing evidence has demonstrated the advantageous synergy between regenerative rehabilitation and stem cell therapy in treating SCI. However, further investigation is warranted to fully explore the possibilities of integrating stem cell therapy with regenerative rehabilitation.
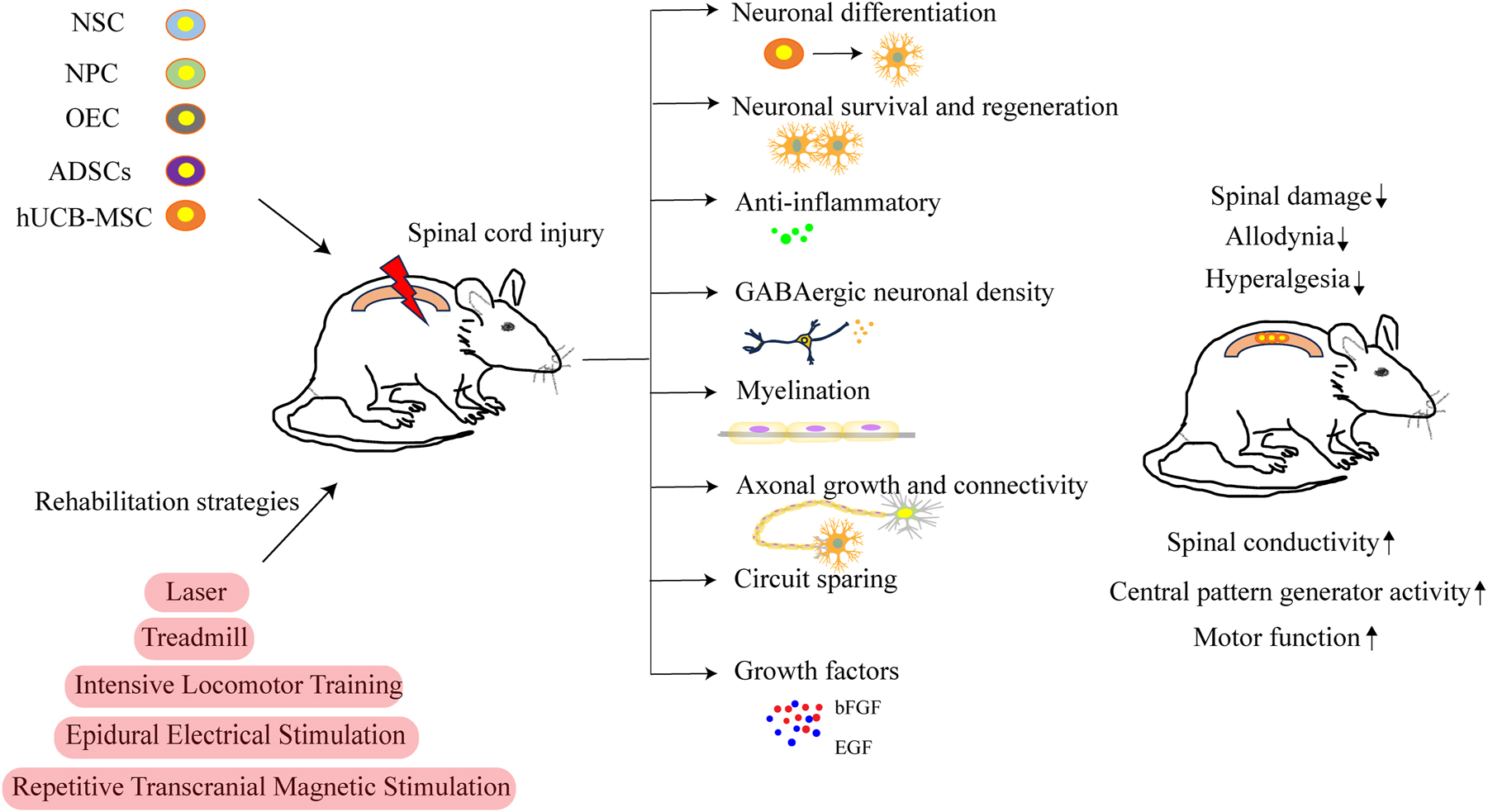
Regenerative rehabilitation for spinal cord injury. Cell therapy using NSC, NPC, OEC, ADSCs or hUCB-MSC, when combined with rehabilitative strategies, has been studied in rodent models of spinal cord injury. The rehabilitative interventions include techniques such as laser therapy, treadmilling, intensive locomotor training, epidural electrical stimulation, and repetitive transcranial magnetic stimulation. This combination has the potential to influence neural differentiation, neuronal survival and regeneration, anti-inflammatory abilities, GABAergic neuronal density, myelination, axonal growth and connectivity, circuit sparing, as well as growth factors, thus contributing towards the decreased spinal damage, reduced allodynia and hyperalgesia, increased spinal conductivity, enhanced central pattern generator activity, and improved motor functions. OEC, olfactory ensheathing cells; ADSC, adipose-derived stem cells; hUCB-MSC, human umbilical cord blood mesenchymal stem cell.
Regenerative rehabilitation for knee osteoarthritis
OA, a degenerative condition, affects approximately 15 % of the adult population and ranks as the second leading cause of disability globally [130]. The knee joint is predominantly affected, characterized by progressive degradation of articular cartilage, loss of ECM, and ongoing inflammation. OA significantly impacts both the individual quality of life and healthcare system expenditures [131]. Traditional OA management primarily focuses on symptom alleviation, particularly pain relief, rather than facilitating cartilage regeneration or mitigating joint inflammation. MSCs have emerged as a promising candidate for cell-based repair of articular cartilage in patients with knee OA [132]. Given the limited inherent repair capacity of articular cartilage, intra-articular ligaments, and menisci, MSCs have garnered significant attention in the development of novel therapeutic approaches for OA treatment. Their ability to differentiate into chondrocytes and modulate immune responses makes MSCs a key target for innovative cell-based OA therapies [133]. Mechanical stimulation is vital for bone health as it affects bone shape based on force direction, with bone mass increasing in high-stress areas and decreasing in low-stress areas. The bone may adjust to the external mechanical loading [134]. Mechanical stimulation can be sensed by a variety of bone cells, including bone marrow MSCs and osteocytes. These cells have different roles in a variety of physiological processes and respond to external mechanical stimuli. Recent research shows that external mechanical stimulation directs BMSCs into an osteogenic lineage that is independent of osteocyte regulation [135]. Some studies on the molecular mechanisms of MSCs’ responses to different mechanical stimuli have been addressed here (Figure 6). Tensile strain stimulation augments MSC osteogenesis differentiation while inhibiting adipogenesis differentiation, mostly via the Smad signaling pathway [136]. Chen et al. suggested that optimum levels of mechanical stretching enhanced the osteogenesis of BMSCs decreased ROS levels in BMSCs and elicited antioxidant responses by activating the AMPK-SIRT1 pathway [137]. MSC differentiation and mineralization toward osteogenesis and chondrogenesis can be promoted through dynamic compression [138]. Another study revealed that compression-induced MSC differentiation toward chondrogenic or osteogenic lineages was dependent on ERK1/2 pathway activation, while under normal settings, dynamic compression produces chondrogenic differentiation of MSCs and osteogenesis differentiation when the ERK1/2 pathway is inhibited [139]. However, the mechanism underlying the effect of compression stimulus on MSCs and the means of controlling the differentiation of MSCs has not yet been fully investigated.
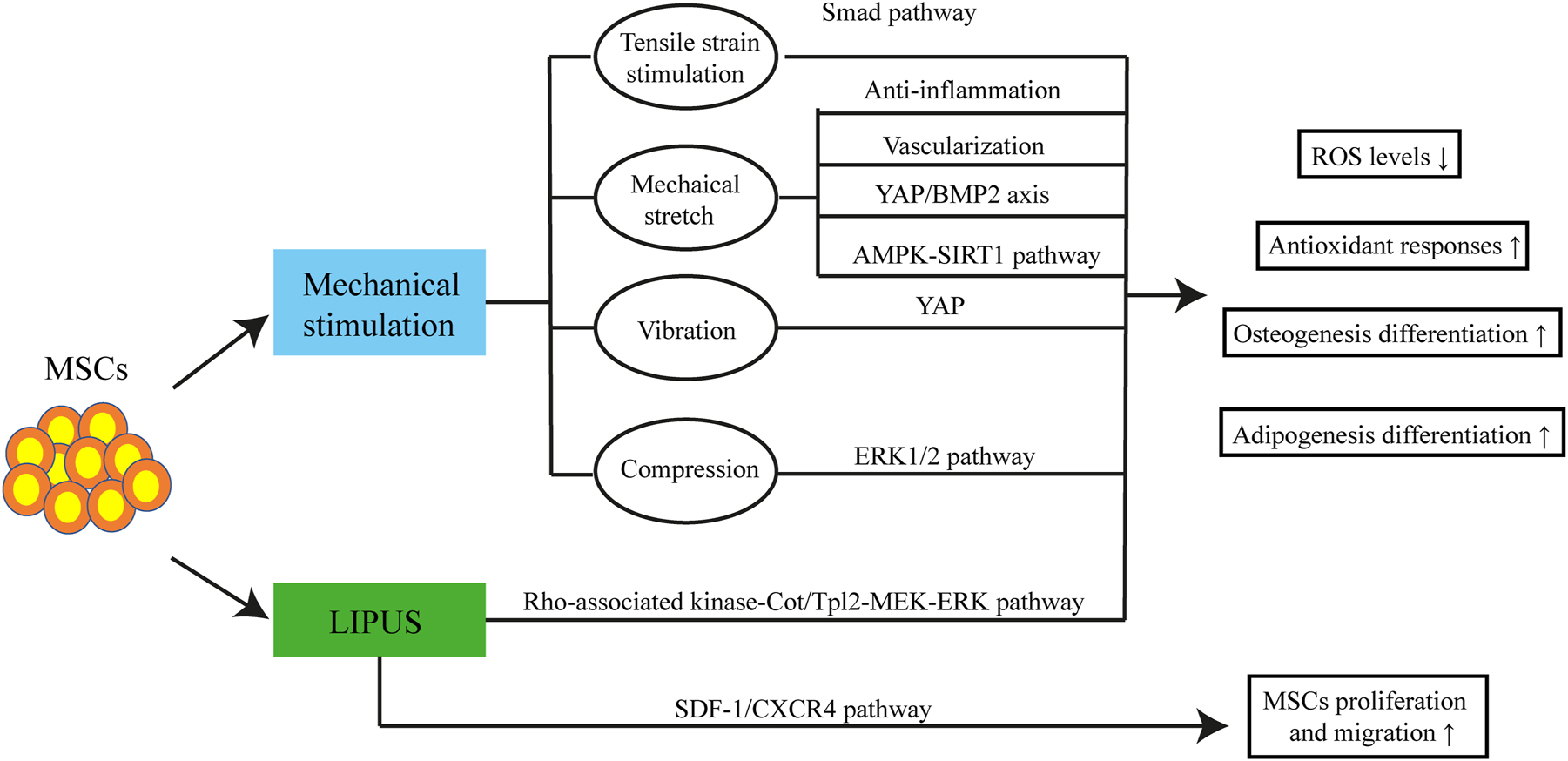
Regenerative rehabilitation for knee osteoarthritis. In animal models of knee osteoarthritis, therapies of MSCs combined with mechanical stimulation involving tensile strain stimulation, mechanical stretch, vibration, compression, and LIPUS reduce ROS levels while increasing antioxidant responses, osteogenesis differentiation, and adipogenesis differentiation. These effects are mediated via Smad pathway, anti-inflammatory effects, vascularization, YAP/BMP2 axis, AMPK-SIRT1 pathway, YAP, ERK 1/2 pathway, and Rho-associated kinase-Cot/Tpl2-MERK-ERK pathway. In addition, LIPUS coupled with MSC therapy promotes the proliferation and migration of MSCs via the SDF-1/CXCR4 pathway. MSCs, mesenchymal stem cells; LIPUS, low-intensity pulsed ultrasound; YAP, yes-associated protein.
Furthermore, appropriate amplitude and frequency vibrations can induce anabolic reactions in the bones. Recent research demonstrated the effect of vibration on the Yes-associated protein (YAP), a transcription factor significant in MSC osteogenesis. Thompson et al. [140] revealed that vibration boosted YAP nuclear shuttling and restored baseline nuclear levels of YAP, resulting in MSC osteogenesis. Low-Intensity Pulsed Ultrasound (LIPUS) is also found to be a type of force to promotes bone formation. LIPUS increased all MSC-type’s proliferation. ERK1/2 was activated in dental pulp stem cells (DPSCs). Besides proliferation, LIPUS also promotes the MSCs migration in bone healing possibly through activating the SDF-1/CXCR4 signaling [141]. Several studies illustrated that LIPUS led to better osteointegration [142]. The possible osteogenic differentiation mechanism is the actuation of the Rho-associated kinase-Cot/Tpl2-MEK-ERK signaling pathway [143]. MCS niches create a microenvironment vital for MSC self-renewal differentiation, and bone regeneration. The process involves inflammatory responses, blood vessel formation of endothelial cells, and the osteogenic process of MSCs. Effective intercellular communication within the niche, including crosstalk with macrophages, vascular endothelial cells, and osteocytes, is critical for bone regeneration. Additionally, mechanical stimulation regulates key aspects, such as inflammation and blood vessel formation, and mechanical stimulation modulates the surrounding microenvironment of MCS in bone regeneration. In this context, we state some studies that link to the regulation of microenvironments surrounding MCS through mechanical stimuli. Most studies have focused on MSCs’ responses to mechanical stimulation during osteogenesis. A recent study revealed that macrophages, under specific stretch conditions, effectively enhance osteogenic differentiation in MSCs. This involves an interaction between M2 polarization and the YAP/BMP2 axis. Cyclic stretch stimulates macrophages, leading to M2 polarization, which produces anti-inflammatory cytokines. Moreover, mechanical stretch triggers YAP activation, regulating BMP2 expression and enhancing MSCs’ osteogenesis [144]. Mechanical stimulation is one of the biophysical factors that stimulate osteogenesis and is important in the interaction of MSCs and vascular endothelial growth factors (VECs). The significance of VEGF, a recognized signaling protein that regulates osteogenesis and vascularization, in mechanical stimulation-induced osteogenesis has been found. Charoenpanich et al. [145] used microarray analysis to investigate the effect of stretching on human MSC gene expression. The results of cyclic tensile strain of magnitude not only enhanced osteogenesis in hMSC but also enhanced expression of angiogenic factors VEGF. Furthermore, Jiang’s team showed that stretching-stimulated VEGF secretion of MSCs not only increased tube formation but also induced VECs to release bone formation growth factors such as BMP-2 and IGF-1, which in turn regulated the osteogenesis process of MSCs [146]. Overall, MSCs respond to mechanical signals by orchestrating responses that include immunological regulation, angiogenesis, and osteogenesis. The exciting interaction of mechanical stimulation in the environment around MSCs calls for further research.
Regenerative rehabilitation for cardiovascular diseases
Cardiovascular diseases remain the foremost cause of death in the developed as well as in the developing, rendering cardiovascular diseases the main contributor to mortality worldwide [147]. Over the years, an increasing set of solid data has shown that aerobic physical exercise has beneficial effects on the prevention and reduction of cardiovascular risk, even if the underlying mechanisms have not been completely refined [148]. Aerobic or endurance training, such as running, jogging, slow swimming, and cycling, are particularly effective in initiating a series of mechanisms that allow the body, including the heart and vessels, to adapt and improve overall cardiovascular function [149, 150]. Research shows physical exercise training (ET) has positive implications for HF management, attenuates HF symptoms, and improves both systolic and diastolic function, affecting cellular contraction [151, 152] (Figure 7). Exercise can also initiate positive mechanisms that improve blood pressure, reduce resting vascular resistance, and improve cardiac function and structure (Figure 7). MSC therapy has been related to various beneficial effects in reducing myocardial infarct size (Figure 7). These benefits of therapy include a reduction in the size of the infarcted area, reduced thinning of the left ventricular (LV) wall, antiapoptotic properties, and the mitigation of ventricular remodeling via antifibrotic mechanisms. Likewise, MSCs have shown the capacity to enhance myocardial perfusion and preserve both systolic and diastolic performance, as well as cardiac electrical viability and impulse propagation [153–156]. At the same time, nitric oxide (NO) is increasingly recognized as a molecule of importance for the protection, growth, and differentiation of a variety of stem/progenitor cells [150] (Figure 7). This can be relevant because one of the problems associated with autologous and allogeneic stem cell therapies is the poor survival of transplanted cells. To enhance the post-stem cell transplantation survival and therapeutic potential through physical ET, by enhancing the bioavailability of NO and its downstream signaling, can protect stem cells in a hostile environment and enhance their activation, mobilization, and differentiation that, in turn, can translate to improved tissue repair. In an investigation involving rats with MI, the rats were divided into groups: sedentary without transplant cells, sedentary with cells, trained without cells, and trained with cells. The sedentary group exhibited a lower left ventricle ejection fraction (LVEF), whereas the trained group with cells demonstrated a higher LVEF and stabilized ventricular remodeling [157]. Besides in the trained groups, collagen reduction and decreased cardiac fibrosis were observed, indicating the potential benefits of combining cell treatment with physical exercise on ventricular function [157]. A more recent study investigated the effect of aerobic exercise and MSC therapy on MI in rats. Rats were divided into sedentary (SC) and exercised (EX) groups with or without MSC transplanted. The findings showed that rats in the combination group had higher cardiac function than those in the SC group and had lower collagen content than SC, while EX and EX-MSC had lower s α-actin expression than SC.
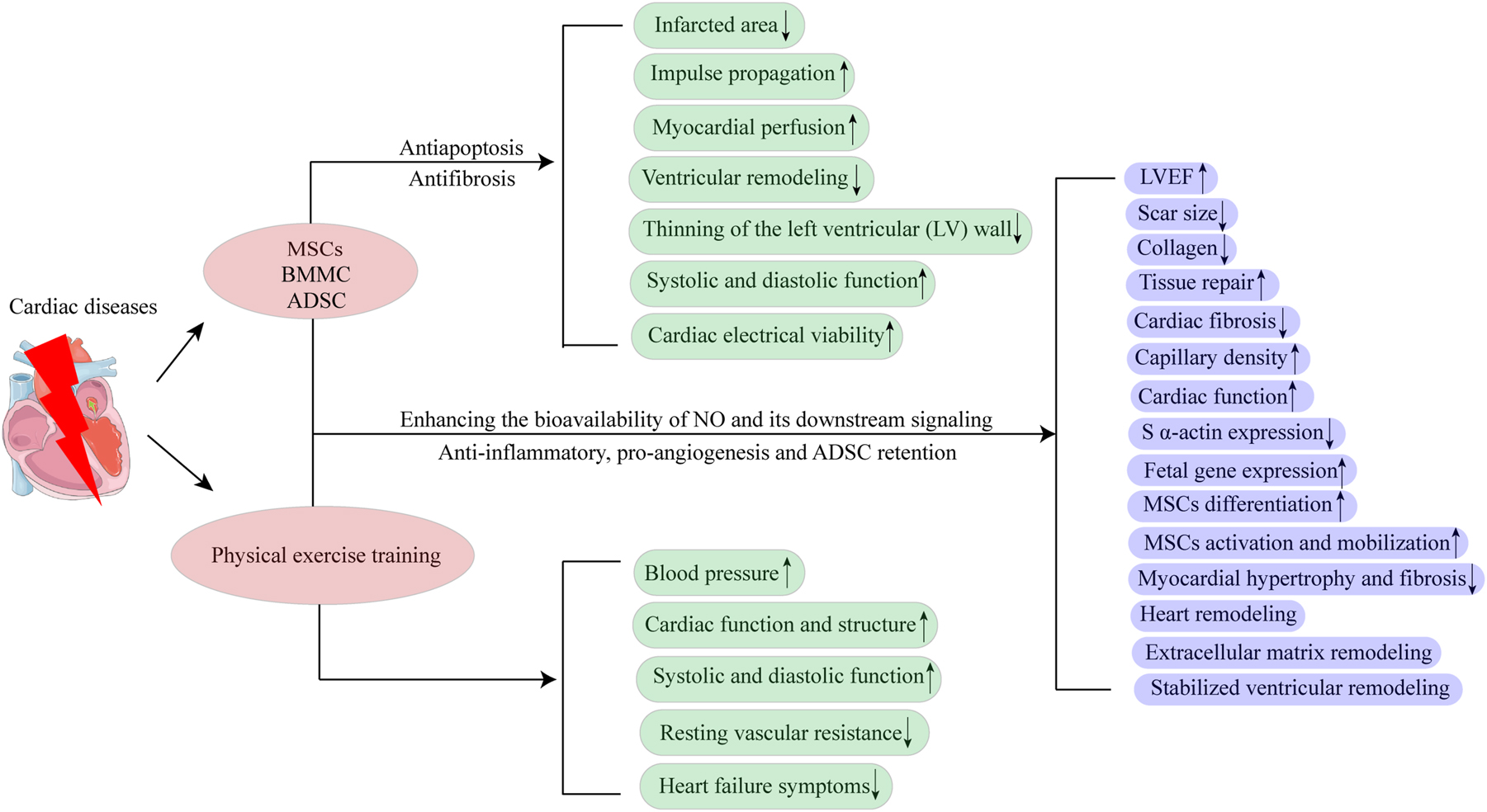
Regenerative rehabilitation for cardiovascular diseases. Stem cell therapy, which includes MSCs, BMMS, and ADSC, has been found to be effective in reducing the infarcted area, thinning of the LV wall, and ventricular remodeling in cardiovascular diseases. It also improves myocardial perfusion, impulse propagation, systolic and diastolic function, and cardiac electrical viability. These positive results are due to the anti-apoptosis and anti-fibrosis effects of stem cell therapies. Moreover, physical exercise training has been found to be helpful in increasing blood pressure, cardiac function and structure, and systolic and diastolic function, as well as reducing resting vascular resistance and heart failure symptoms. However, when stem cell therapy is combined with physical exercise training, the effects of these treatments can be enhanced, and more positive results can be achieved. This is because of the enhanced bioavailability of NO and its downstream signaling, anti-inflammatory abilities, pro-angiogenesis, and ADSC retention. MSCs, mesenchymal stem cells; ADSC, adipose-derived stem cells; LV, left ventricular; NO, nitric oxide.
Aerobic exercise seems to boost the beneficial effects of stem cell therapy on ECM remodeling and fetal gene expression in the left ventricle of rats with moderate infarction [158]. Furthermore, another study investigated how ET affects the cardioprotective impact of ADSC transplantation in rats following MI. Echocardiography revealed reduced scar size in both non-trained rats receiving ADSC transplantation (sADSC) and trained rats receiving ADSC transplantation (tADSC), However, fractional shortening was improved only in the tADSC group. In vitro myocardial performance, compared to sMI rats, the tADSC group exhibited higher expression of phosphoSer473Akt1 and VEGF, and histologic examination indicated enhanced capillary density in the remote and border zones of infarcted areas. In summary, exercise preconditioning creates a pro-angiogenic environment, which could boost the therapeutic effects of ADSCs on heart remodeling after MI [158] (Figure 7). Moreover, the convergence of aerobic exercise and stem cell therapy could prove to be a comprehensive approach to cardiovascular disease treatment providing significant improvements in the management of cardiovascular conditions particularly MI.
Another study explored the impact of ET on post-infarction retention of ADSCs and its combined effects on the inflammatory environment. ET was investigated for its impact on post-MI cardiac remodeling in rats following ADSC treatment. ET for nine weeks before MI and subsequent ADSC transplantation improved LV parameters inhibited myocardial hypertrophy and fibrosis, and decreased proinflammatory cytokines in rats compared to sedentary MI rats. Notably, ET increased the positive effects of ADSCs, attenuation of proinflammatory cytokines (such as interleukins 1β and 10, tumor necrosis factor α, and transforming growth factor β), and was correlated with higher ADSC retention in the myocardium. These results highlight the significance of exercise in improving ADSC retention and reducing cardiac remodeling following MI suggesting a positive effect on the myocardial microenvironment based on anti-inflammatory actions [158].
Regenerative rehabilitation for other diseases
In addition to stroke, SCI, OA, cardiovascular diseases, and other well-recognized conditions, regenerative rehabilitation has been investigated in other conditions such as skeletomuscular diseases, dry eyes, neurodegenerative diseases, and diabetic peripheral neuropathy [159–163]. For example, it has been shown that exercise combined with a 3D nano fibrillar scaffold can improve the abundance of neuromuscular junctions and increase the regenerating myofibers in a mouse model of volumetric muscle loss [160]. For corneal complications of dry eye disease, transplantation of limbal stem cells can restore limbal stem cell deficiency [161]. Additionally, regenerative rehabilitation based on exosomes provides approaches to promoting neural regeneration. It was shown that electrical stimulation combined with exosomes from BMSCs had positive effects on diabetic peripheral neuropathy [162]. Furthermore, emerging areas such as wound healing, immune modulation, and organ regeneration offer exciting opportunities for regenerative rehabilitation to optimize recovery and enhance the QoL [164–166].
Regenerative rehabilitation in clinical practice
Hematopoietic stem cell transplantation (HSCT) combined with rehabilitation therapy, is one of the most widely used treatments for various types of malignant tumors [167]. It has a vast amount of clinical experience. Every year, more than 50,000 HSCT treatments are performed globally, significantly improving the survival rate of patients with malignant tumors [167]. However, despite the effectiveness of HSCT, it is observed that the incidence of cardiovascular risk factors and cardiovascular diseases in these patients is higher than that of the general population [167]. The incidence rates of hypertension, dyslipidemia, diabetes, and obesity among HSCT survivors can reach up to 28 %–74 %, 33 %–58 %, 10 %–41 %, and 20 %–44 %, respectively [168–170]. The incidence of arrhythmias, HF, and MI ranges from 2 to 13 %, 1 %–9 %, and 1 %–6 %, respectively [171–173]. It is worth noting that these data imply that HSCT survivors have a 7- to 16-fold higher incidence of CVD risk factors and a 2- to 4-fold increase in CVD mortality compared with the general population [174–177]. The risk factors for such outcomes may be pre-existing cardiovascular disease risk factors and functional disability, prolonged bed rest, organ damage from chemotherapy/radiation therapy, and graft-versus-host disease (GVHD) [173]. HSCT patients typically have an average hospitalization period of ≥ 4 weeks, which may negatively affect their cardiorespiratory health due to long-term bed rest, such as decreased muscle mass and exercise capacity [173, 178, 179]. Therefore, improving the cardiopulmonary health of HSCT patients is of utmost importance. As recommended by the American College of Sports Medicine (ACSM), incorporate moderate-intensity aerobic training for at least 30 min at least 3 times per week for at least 8–12 weeks, and at least 2 times/week using at least 2 sets of 8–15 resistance training at a maximum of 60 % of 1 repetition should be prescribed for cancer patients [180, 181]. Many clinical studies have shown that physical activity can improve cardiovascular health and strength, reduce fatigue, and improve HSCT patients’ and survivors’ QoL. Some studies have shown that six-week treadmill walking can improve metabolism in patients with solid tumors and non-Hodgkin lymphoma undergoing HSCT [182]. Combining walking, stretching, and strength training improves the 6-min walk distance in patients with multiple myeloma undergoing HSCT. Exercise therapy can be safely initiated during or before transplant hospitalization, positively affecting limb strength and cardiorespiratory fitness [183]. Moreover, physical therapy can increase or decrease the engraftment rate of HSCs, shorten the duration of total parenteral nutrition, reduce the patient’s need for blood transfusions, and reduce the number of attempts required to harvest stem cells [183–186]. Physical therapy and exercise can also reduce fatigue and the emotional impact of HSCT on patients [184, 187]. Studies have shown that patient-reported physical function before HSCT predicts survival and that exercise initiated before HSCT significantly positively affects upper and lower limb strength.
Challenges and perspectives
Interdisciplinary collaborations
The intersection of rehabilitation science and technology with regenerative therapy is becoming increasingly crucial. Consequently, these two fields are mutually influencing each other to a greater extent. However, efforts to elucidate the precise biological mechanisms underlying regenerative rehabilitation strategies are still ongoing. Establishing a new field requires time, vision, and the commitment of key stakeholders. Federal government agencies have been allocating resources to support research and clinical care endeavors in regenerative medicine. Their collective aim is to define a vision for the field, identify crucial challenges that must be addressed, and discuss key initiatives to promote the growth of regenerative rehabilitation. Panelists unanimously concurred that the moment has arrived to transition regenerative rehabilitation research from isolated collaborative endeavors to the formal establishment of an interdisciplinary field [47].
Facilitating interdisciplinary interactions
Interdisciplinary research, where knowledge and approaches are combined, is often the impetus for a new field. Within this latter approach, researchers possess training and expertise that extend more than one discipline, allowing them to rapidly integrate fundamental principles across both fields and act as focal points to bridge collaborations. However, rehabilitation medicine has a long history of interdisciplinary approaches following prevalent conditions such as spinal injury, stroke, joint replacement, etc. Regenerative rehabilitation is poised to transition from multidisciplinary collaborations to a new interdisciplinary field. The question remains as to how to orchestrate this transition with the most effectiveness and efficiency. Experts in regenerative medicine and rehabilitation can benefit greatly from working together in a multidisciplinary group (Figure 8). Such a group would comprise regenerative medicine experts, rehabilitation doctors, and rehabilitation therapists. Through collaboration, they can deepen their understanding and cooperation to develop innovative therapeutic methods that can improve the functions and activities of daily living of disabled patients [188]. Additionally, in rehabilitation medicine centers, a similar multidisciplinary working group including experts from the rehabilitation field and regenerative medicine can be established to explore projects that carry out regenerative rehabilitation in scientific research activities. To support this, cell biologists, molecular biologists, material scientists, bioinformatics scientists, and physicians are required to help translate innovative therapeutic methods from laboratories to the bedsides of patients [2] (Figure 8). The University of Washington has established a steering committee and developed a set of guidelines to guide regenerative rehabilitation activities. The aim is to establish an intersection of research, education, and clinical application to optimize the precision and effects of regenerative medicine [188]. Since stem cells are susceptible to mechanics and mechanical stimulation can enhance their functions [189–191], it is important to determine the optimal time to start rehabilitation protocols to maximize the benefits of regenerative medicine.
Interdisciplinary collaboration of regenerative rehabilitation. Experts in multidisciplinary areas must work together and have close communications, aiming to establish an intersection of research, education, and clinical application to optimize the precision and effects of regenerative medicine.
Developing regenerative rehabilitation at the institutional level
The growth of an interdisciplinary field of research at the institutional level will demand engagement with researchers and clinicians who are actively treating patients (Figure 8). First, researchers, clinicians, and educators should engage in education of regenerative rehabilitation, and its effects and potential benefits in various disease conditions. New educational curricula that encompass regenerative medicine and rehabilitative strategies need to be developed. It is necessary to introduce advanced biological technologies such as stem cell therapies, tissue engineering, gene editing, and computer sciences to offer valuable insights and a deep understanding of this integrated new field. Furthermore, there is a pressing need to establish training programs that bridge these disciplines, offering certificates, degrees, and fellowships in novel multidisciplinary areas. Leveraging new tools and technologies, including online platforms such as Massive Open Online Courses (MOOCs), can facilitate this educational evolution and enable broader access to interdisciplinary training opportunities.
Increasing the visibility of regenerative rehabilitation
With the formation of any new field, promotion, and visibility are needed for the area to engage established researchers and attract new trainees. The area will benefit from stakeholders, ICRR members, and regenerative rehabilitation researchers taking an influential role in promoting success, engaging at both the pre-clinical and clinical levels with outreach to current fields. Notably, regeneration rehabilitation can grow by highlighting its solutions and successes to funding partners, particularly regenerative rehabilitation (NIH and VA). A further step in adopting leading-edge technologies in regenerative rehabilitation includes implementing new regenerative technologies in the clinic and new regenerative therapies, which can significantly improve the translation of regenerative medicines and ultimately optimize patient outcomes. The emergence and growth of the field of regenerative rehabilitation have great potential to improve clinical outcomes for patients with disabilities. However, this field is in its infancy, and perfectly combining regenerative and rehabilitation medicine technology is an issue that needs to be continuously investigated.
Ethical issues
As regenerative rehabilitation is rapidly evolving, an exciting promise is to provide a comprehensive solution for patients on the waiting lists for organ transplantation. By merging organ bioengineering, transplantation, and rehabilitation, patients can receive a more effective treatment plan [5] (Figure 9). However, ethical issues are an important factor affecting the progress of stem cell therapy. First, the fundamental issue of applying human embryonic stem cells (hESCs) is to destroy early human embryos. Faced with this issue, different countries have introduced different policies [192]. For example, in the UK, the use of hESCs for research is allowed, but nuclear transfer for reproductive or therapeutic purposes is illegal [192]. In Italy, all hESC research is illegal. The United States prohibits the destruction of embryos to produce hESCs lines but allows the use of hESCs lines generated before August 9, 2001 [192]. Therefore, most current research on hESCs focuses on animal models [193]. Later, the emergence of iPSCs solved this ethical problem because the production of iPSC lines does not require the destruction of embryos. Patient-specific iPSCs can be used for drug screening, establishing in vitro models of human diseases, and creating new reproductive technologies. Since iPSC-derived cells are produced from somatic cells obtained from the patient, there is no risk of immune rejection after iPSC transplantation [194]. Developments in reproductive technologies have enabled the production of sperm and eggs from human iPSCs [195]. This technology may be useful in treating infertility, but the use of iPSC-derived gametes has created additional ethical issues, such as concerning the potential utilization of created embryos, human NT, the possibility of isosexual and asexually derived gametes [195]. MSCs are considered safe to date, but continued monitoring and long-term follow-up should be performed to avoid the possibility of tumor formation. However, it is worth noting that this is also a challenging issue when clinics offer unproven stem cell treatments. Therefore, researchers and clinicians are ethically obligated to adhere to ethical guidelines.
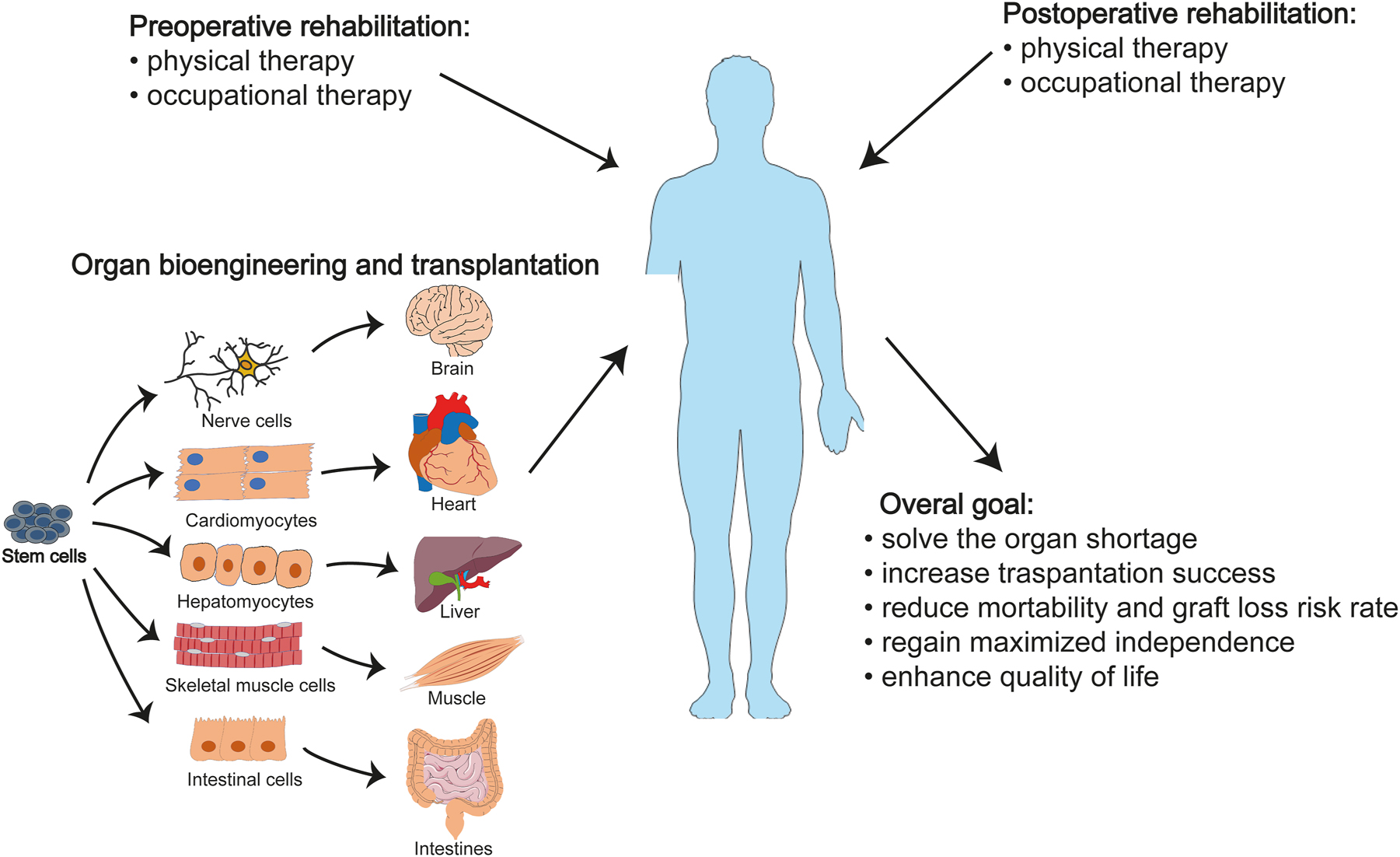
Organ bioengineering, transplantation, and rehabilitation. Regenerative rehabilitation is an exciting field that is rapidly evolving. One of its most promising applications is providing a comprehensive solution for patients who are on organ transplantation waiting lists. By combining organ bioengineering, transplantation, and rehabilitation, patients can receive a more effective treatment plan. Organoids generated from patient-derived iPSCs can be used to address the issues of organ shortages and immune rejection. Additionally, incorporating rehabilitation strategies such as physical and occupational therapies before and after transplantation can significantly improve the success rate of transplantations, reduce the risk of graft loss and mortality, and increase independence and quality of life for patients. It is important to note, however, that the development of organ bioengineering is still ongoing and requires further research and development. iPSCs, induced pluripotent stem cells. Parts of the figure were drawn by using pictures from Servier Medical Art. Servier Medical Art by Servier is licensed under a Creative Commons Attribution 3.0 Unported License (https://creativecommons.org/licenses/by/3.0/).
Regulatory and safety concerns
To ensure patients’ safety in the field of regenerative rehabilitation, stem cell therapy poses several regulatory and safety issues. First, before and after stem cell transplantation, their behavior and efficacy need to be tightly controlled. The microenvironment that affects stem cell proliferation and survival, migration to the site of interest, and the ability to differentiate into specific cells should also be modeled to ensure success for the host treatment [13, 33]. Secondly, we need to further research and apply new technologies, such as 3D bioprinting and suitable biomaterials, to overcome the challenges of tissue heterogeneity and vascularization and achieve structurally and functionally correct tissue or organ survival after transplantation. Third, an ideal immune system in the host can optimize the outcome of regenerative therapies, so a better understanding of immune responses and appropriate techniques to adapt stem cells to their environment will address this issue. Stem cell treatments, which are subject to scientific, ethical, and legal controversies, need to be regulated, and major countries such as the United States, Japan, and China have currently issued regulatory guidelines to ensure the safety and effectiveness of these treatments [196–199]. With the rapid development of this multidisciplinary field, more guidelines should be developed to ensure the safety and effectiveness of new treatment technologies and protocols in clinical applications.
Safety concerns for unproven stem cell therapies
All treatment options have associated benefits and risks, and stem cell treatments that have not been proven safe and effective can be extremely dangerous for clinical patients. The risks include: first, after injecting stem cells, the local reaction is unclear; second, the injected stem cells may transfer from the injection site to other places, and then develop into unfavorable cell types in the microenvironment of other places, or possessing other unpredictable abilities; third, cells may not develop according to the ideal state, but develop into tumors due to their ability to proliferate indefinitely; fourth, stem cell treatments that have not been verified and standardized may be a risk of contamination. Although autologous stem cells are currently considered safer than allogeneic stem cells, the risks mentioned above still exist. For example, patients with systemic lupus erythematosus developed renal failure due to the infusion of autologous HSCs into the kidneys and developed hemangioma and proliferative lesions [200]. In the end, their kidneys had to be removed [200]. Intravitreal injection of stem cells derived from autologous adipose tissue has also caused vision deterioration and even loss of light perception in patients with macular degeneration [201]. Therefore, if unapproved stem cells are being used, or if stem cell products are processed in a way that goes beyond minimal manipulation, there must be relevant administrative and judicial measures to curb these practices.
Some therapies exhibit a favorable risk-benefit balance, while others lack proven efficacy, and their associated adverse effects often go unreported. The extent to which the clinical benefits of these therapies outweigh potential risks remains uncertain. Strengthening oversight of clinical applications and stem cell clinics is crucial to mitigate potential risks. To address the benefits and risks associated with stem cell therapy, the FDA published an article in the New England Journal of Medicine (NEJM) in March 2017 [196]. This article emphasized the FDA’s role in facilitating the development and approval of new stem cell therapies [196]. However, it underscores the importance of providing scientific evidence to support the safety and efficacy of these products. Based on evidence-based standards, unsafe or ineffective treatments will be excluded from routine use [196]. Therefore, stem cell treatments require controlled clinical studies to determine that the therapy is safe and effective for its intended use.
Accelerated approval for breakthrough therapies
Due to the complexity of mammalian cells, the behavior of stem cells when introduced into new environments is difficult to predict, so the safety of such treatments requires documented empirical data. The FDA said that for treatments that can provide relatively impressive benefits to patients, only large-scale studies are needed to prove that the benefits outweigh the risks. For treatments with fairly significant benefits, regulators can approve modest trial sizes, such as Randomized trials with at least 42 participating patients redetected statistically significant 100 % improvements in outcome measures with α=0.05 and β=0.1 [196]. Additionally, the FDA believes that for life-threatening serious illnesses, expedited pathways are readily available when current medical needs are not sufficient to treat the disease, especially in the field of oncology, to develop creative treatments that will meet the needs of patients and physicians and give them an interest in commercially available treatments-drugs and biologics with confidence. For applications in non-life-threatening situations but with relatively large patient numbers, moderately sized trials are likely to show a favorable benefit-risk profile, and such trials will facilitate the development of truly effective cell preparations and delivery methods. Stem cell therapy product developers need to conduct a systematic understanding of the available pathways to develop cell therapy products, collect necessary clinical data, and establish innovative methods for evidence production to facilitate the development and eventual licensing of safe, effective stem cell therapies. Therefore, to promote and ensure the safe and effective application of stem cell therapy in rehabilitation, drug regulatory authorities/stem cell therapy product researchers and sponsors must work together to solve existing problems and difficulties with stem cells.
Funding source: National Natural Science Foundation of China
Award Identifier / Grant number: 82171422
Funding source: Key Research and Development Project of Hubei Province of China
Award Identifier / Grant number: 2022BCA028
Acknowledgments
We thank our colleagues for their support in writing this review.
-
Research ethics: This article is not associated with ethics.
-
Informed consent: This article contains no studies with human participants or animals performed by authors.
-
Author contributions: H. Chen and C. Deng conceived and designed the manuscript. C. Deng and F. Aldali prepared the draft of the manuscript and figures. H. Luo assisted in the preparation of the figures. H. Chen read and revised it. All authors have accepted responsibility for the entire content of this manuscript and approved its submission.
-
Competing interests: The authors state no conflict of interest.
-
Research funding: This work was funded by grants from the National Natural Science Foundation of China (82171422) and Key Research and Development Project of Hubei Province of China (2022BCA028).
-
Data availability: All datasets are available in the main text or the Supplementary Materials and will be available from the corresponding author upon request.
References
1. Willett, NJ, Boninger, ML, Miller, LJ, Alvarez, L, Aoyama, T, Bedoni, M, et al.. Taking the next steps in regenerative rehabilitation: establishment of a new interdisciplinary field. Arch Phys Med Rehabil 2020;101:917–23. https://doi.org/10.1016/j.apmr.2020.01.007.Suche in Google Scholar PubMed PubMed Central
2. Cheuy, V, Picciolini, S, Bedoni, M. Progressing the field of Regenerative Rehabilitation through novel interdisciplinary interaction. NPJ Regen Med 2020;5:16. https://doi.org/10.1038/s41536-020-00102-2.Suche in Google Scholar PubMed PubMed Central
3. Perez-Terzic, C, Childers, MK. Regenerative rehabilitation: a new future? Am J Phys Med Rehabil 2014;93:S73–8. https://doi.org/10.1097/phm.0000000000000211.Suche in Google Scholar
4. Daar, AS, Greenwood, HL. A proposed definition of regenerative medicine. J Tissue Eng Regen Med 2007;1:179–84. https://doi.org/10.1002/term.20.Suche in Google Scholar PubMed
5. Edgar, L, Pu, T, Porter, B, Aziz, JM, La Pointe, C, Asthana, A, et al.. Regenerative medicine, organ bioengineering and transplantation. Br J Surg 2020;107:793–800. https://doi.org/10.1002/bjs.11686.Suche in Google Scholar PubMed
6. He, S, Nakada, D, Morrison, SJ. Mechanisms of stem cell self-renewal. Annu Rev Cell Dev Biol 2009;25:377–406. https://doi.org/10.1146/annurev.cellbio.042308.113248.Suche in Google Scholar PubMed
7. Hoang, DM, Pham, PT, Bach, TQ, Ngo, ATL, Nguyen, QT, Phan, TTK, et al.. Stem cell-based therapy for human diseases. Signal Transduct Targeted Ther 2022;7:272. https://doi.org/10.1038/s41392-022-01134-4.Suche in Google Scholar PubMed PubMed Central
8. Salari, V, Mengoni, F, Del Gallo, F, Bertini, G, Fabene, PF. The anti-inflammatory properties of mesenchymal stem cells in epilepsy: possible treatments and future perspectives. Int J Mol Sci 2020;21:9683. https://doi.org/10.3390/ijms21249683.Suche in Google Scholar PubMed PubMed Central
9. He, A, Jiang, Y, Gui, C, Sun, Y, Li, J, Wang, JA. The antiapoptotic effect of mesenchymal stem cell transplantation on ischemic myocardium is enhanced by anoxic preconditioning. Can J Cardiol 2009;25:353–8. https://doi.org/10.1016/s0828-282x(09)70094-7.Suche in Google Scholar PubMed PubMed Central
10. Jahani, M, Rezazadeh, D, Mohammadi, P, Abdolmaleki, A, Norooznezhad, A, Mansouri, K. Regenerative medicine and angiogenesis; challenges and opportunities. Adv Pharmaceut Bull 2020;10:490–501. https://doi.org/10.34172/apb.2020.061.Suche in Google Scholar PubMed PubMed Central
11. Schwarz, S, Huss, R, Schulz-Siegmund, M, Vogel, B, Brandau, S, Lang, S, et al.. Bone marrow-derived mesenchymal stem cells migrate to healthy and damaged salivary glands following stem cell infusion. Int J Oral Sci 2014;6:154–61. https://doi.org/10.1038/ijos.2014.23.Suche in Google Scholar PubMed PubMed Central
12. Aly, RM. Current state of stem cell-based therapies: an overview. Stem Cell Invest 2020;7:8. https://doi.org/10.21037/sci-2020-001.Suche in Google Scholar PubMed PubMed Central
13. Mao, AS, Mooney, DJ. Regenerative medicine: current therapies and future directions. Proc Natl Acad Sci U S A 2015;112:14452–9. https://doi.org/10.1073/pnas.1508520112.Suche in Google Scholar PubMed PubMed Central
14. Bakhtiar, S, Fekadu, J, Seidel, MG, Gambineri, E. Allogeneic hematopoietic stem cell transplantation for congenital immune dysregulatory disorders. Front Pediatr 2019;7:461. https://doi.org/10.3389/fped.2019.00461.Suche in Google Scholar PubMed PubMed Central
15. Das, J, Sharrack, B, Snowden, JA. Autologous hematopoietic stem-cell transplantation in neurological disorders: current approach and future directions. Expert Rev Neurother 2020;20:1299–313. https://doi.org/10.1080/14737175.2020.1820325.Suche in Google Scholar PubMed
16. Mariottini, A, Bulgarini, G, Cornacchini, S, Damato, V, Saccardi, R, Massacesi, L. Hematopoietic stem cell transplantation for the treatment of autoimmune neurological diseases: an update. Bioengineering 2023;10:176. https://doi.org/10.3390/bioengineering10020176.Suche in Google Scholar PubMed PubMed Central
17. Muraro, PA, Martin, R, Mancardi, GL, Nicholas, R, Sormani, MP, Saccardi, R. Autologous haematopoietic stem cell transplantation for treatment of multiple sclerosis. Nat Rev Neurol 2017;13:391–405. https://doi.org/10.1038/nrneurol.2017.81.Suche in Google Scholar PubMed
18. Cavo, M, Gay, F, Beksac, M, Pantani, L, Petrucci, MT, Dimopoulos, MA, et al.. Autologous haematopoietic stem-cell transplantation versus bortezomib-melphalan-prednisone, with or without bortezomib-lenalidomide-dexamethasone consolidation therapy, and lenalidomide maintenance for newly diagnosed multiple myeloma (EMN02/HO95): a multicentre, randomised, open-label, phase 3 study. Lancet Haematol 2020;7:e456–8. https://doi.org/10.1016/s2352-3026(20)30099-5.Suche in Google Scholar PubMed
19. Gonsalves, WI, Buadi, FK, Ailawadhi, S, Bergsagel, PL, Chanan Khan, AA, Dingli, D, et al.. Utilization of hematopoietic stem cell transplantation for the treatment of multiple myeloma: a Mayo Stratification of Myeloma and Risk-Adapted Therapy (mSMART) consensus statement. Bone Marrow Transplant 2019;54:353–67. https://doi.org/10.1038/s41409-018-0264-8.Suche in Google Scholar PubMed PubMed Central
20. Strocchio, L, Locatelli, F. Hematopoietic stem cell transplantation in thalassemia. Hematol Oncol Clin N Am 2018;32:317–28. https://doi.org/10.1016/j.hoc.2017.11.011.Suche in Google Scholar PubMed
21. Yesilipek, MA, Uygun, V, Kupesiz, A, Karasu, G, Ozturk, G, Ertem, M, et al.. Thalassemia-free and graft-versus-host-free survival: outcomes of hematopoietic stem cell transplantation for thalassemia major, Turkish experience. Bone Marrow Transplant 2022;57:760–7. https://doi.org/10.1038/s41409-022-01613-w.Suche in Google Scholar PubMed
22. Mandai, M, Watanabe, A, Kurimoto, Y, Hirami, Y, Morinaga, C, Daimon, T, et al.. Autologous induced stem-cell-derived retinal cells for macular degeneration. N Engl J Med 2017;376:1038–46. https://doi.org/10.1056/nejmoa1608368.Suche in Google Scholar PubMed
23. Scudellari, M. How iPS cells changed the world. Nature 2016;534:310–2. https://doi.org/10.1038/534310a.Suche in Google Scholar PubMed
24. Apatoff, MBL, Sengillo, JD, White, EC, Bakhoum, MF, Bassuk, AG, Mahajan, VB, et al.. Autologous stem cell therapy for inherited and acquired retinal disease. Regen Med 2018;13:89–96. https://doi.org/10.2217/rme-2017-0089.Suche in Google Scholar PubMed PubMed Central
25. Wernig, M, Zhao, JP, Pruszak, J, Hedlund, E, Fu, D, Soldner, F, et al.. Neurons derived from reprogrammed fibroblasts functionally integrate into the fetal brain and improve symptoms of rats with Parkinson’s disease. Proc Natl Acad Sci USA 2008;105:5856–61. https://doi.org/10.1073/pnas.0801677105.Suche in Google Scholar PubMed PubMed Central
26. Kikuchi, T, Morizane, A, Doi, D, Magotani, H, Onoe, H, Hayashi, T, et al.. Human iPS cell-derived dopaminergic neurons function in a primate Parkinson’s disease model. Nature 2017;548:592–6. https://doi.org/10.1038/nature23664.Suche in Google Scholar PubMed
27. Ortuño-Costela, MDC, Cerrada, V, García-López, M, Gallardo, ME. The challenge of bringing iPSCs to the patient. Int J Mol Sci 2019;20. https://doi.org/10.3390/ijms20246305.Suche in Google Scholar PubMed PubMed Central
28. Aboul-Soud, MAM, Alzahrani, AJ, Mahmoud, A. Induced pluripotent stem cells (iPSCs)-Roles in regenerative therapies, disease modelling and drug screening. Cells 2021;10. https://doi.org/10.3390/cells10092319.Suche in Google Scholar PubMed PubMed Central
29. Deinsberger, J, Reisinger, D, Weber, B. Global trends in clinical trials involving pluripotent stem cells: a systematic multi-database analysis. NPJ Regen Med 2020;5:15. https://doi.org/10.1038/s41536-020-00100-4.Suche in Google Scholar PubMed PubMed Central
30. Hwang, JJ, Rim, YA, Nam, Y, Ju, JH. Recent developments in clinical applications of mesenchymal stem cells in the treatment of rheumatoid arthritis and osteoarthritis. Front Immunol 2021;12:631291. https://doi.org/10.3389/fimmu.2021.631291.Suche in Google Scholar PubMed PubMed Central
31. Squillaro, T, Peluso, G, Galderisi, U. Clinical trials with mesenchymal stem cells: an update. Cell Transplant 2016;25:829–48. https://doi.org/10.3727/096368915x689622.Suche in Google Scholar
32. Mendell, JR, Al-Zaidy, SA, Rodino-Klapac, LR, Goodspeed, K, Gray, SJ, Kay, CN, et al.. Current clinical applications of in vivo gene therapy with AAVs. Mol Ther 2021;29:464–88. https://doi.org/10.1016/j.ymthe.2020.12.007.Suche in Google Scholar PubMed PubMed Central
33. Bongso, A, Fong, CY. The therapeutic potential, challenges and future clinical directions of stem cells from the Wharton’s jelly of the human umbilical cord. Stem Cell Rev Rep 2013;9:226–40. https://doi.org/10.1007/s12015-012-9418-z.Suche in Google Scholar PubMed
34. Kränkel, N, Adams, V, Linke, A, Gielen, S, Erbs, S, Lenk, K, et al.. Hyperglycemia reduces survival and impairs function of circulating blood-derived progenitor cells. Arterioscler Thromb Vasc Biol 2005;25:698–703. https://doi.org/10.1161/01.atv.0000156401.04325.8f.Suche in Google Scholar
35. Capla, JM, Grogan, RH, Callaghan, MJ, Galiano, RD, Tepper, OM, Ceradini, DJ, et al.. Diabetes impairs endothelial progenitor cell–mediated blood vessel formation in response to hypoxia. Plast Reconstr Surg 2007;119:59–70. https://doi.org/10.1097/01.prs.0000244830.16906.3f.Suche in Google Scholar PubMed
36. Biressi, S, Miyabara, EH, Gopinath, SD, Carlig, PM, Rando, TA. A Wnt-TGFβ2 axis induces a fibrogenic program in muscle stem cells from dystrophic mice. Sci Transl Med 2014;6:267ra176. https://doi.org/10.1126/scitranslmed.3008411.Suche in Google Scholar PubMed PubMed Central
37. Neal, A, Boldrin, L, Morgan, JE. The satellite cell in male and female, developing and adult mouse muscle: distinct stem cells for growth and regeneration. PLoS One 2012;7:e37950. https://doi.org/10.1371/journal.pone.0037950.Suche in Google Scholar PubMed PubMed Central
38. Deasy, BM, Lu, A, Tebbets, JC, Feduska, JM, Schugar, RC, Pollett, JB, et al.. A role for cell sex in stem cell–mediated skeletal muscle regeneration: female cells have higher muscle regeneration efficiency. J Cell Biol 2007;177:73–86. https://doi.org/10.1083/jcb.200612094.Suche in Google Scholar PubMed PubMed Central
39. Drowley, L, Okada, M, Beckman, S, Vella, J, Keller, B, Tobita, K, et al.. Cellular antioxidant levels influence muscle stem cell therapy. Mol Ther 2010;18:1865–73. https://doi.org/10.1038/mt.2010.160.Suche in Google Scholar PubMed PubMed Central
40. Archacka, K, Grabowska, I, Mierzejewski, B, Graffstein, J, Górzyńska, A, Krawczyk, M, et al.. Hypoxia preconditioned bone marrow-derived mesenchymal stromal/stem cells enhance myoblast fusion and skeletal muscle regeneration. Stem Cell Res Ther 2021;12:1–18. https://doi.org/10.1186/s13287-021-02530-3.Suche in Google Scholar PubMed PubMed Central
41. Conboy, IM, Rando, TA. Aging, stem cells and tissue regeneration: lessons from muscle. Cell Cycle 2005;4:407–10. https://doi.org/10.4161/cc.4.3.1518.Suche in Google Scholar PubMed
42. Carlson, ME, Conboy, IM. Loss of stem cell regenerative capacity within aged niches. Aging Cell 2007;6:371–82. https://doi.org/10.1111/j.1474-9726.2007.00286.x.Suche in Google Scholar PubMed PubMed Central
43. Carlson, B, Faulkner, J. Muscle transplantation between young and old rats: age of host determines recovery. Am J Physiol 1989;256:C1262–6. https://doi.org/10.1152/ajpcell.1989.256.6.c1262.Suche in Google Scholar
44. Xia, H, Li, X, Gao, W, Fu, X, Fang, RH, Zhang, L, et al.. Tissue repair and regeneration with endogenous stem cells. Nat Rev Mater 2018;3:174–93. https://doi.org/10.1038/s41578-018-0027-6.Suche in Google Scholar
45. Lane, SW, Williams, DA, Watt, FM. Modulating the stem cell niche for tissue regeneration. Nat Biotechnoll 2014;32:795–803. https://doi.org/10.1038/nbt.2978.Suche in Google Scholar PubMed PubMed Central
46. Vacanti, JP, Otte, JB, Wertheim, JA. Chapter 1 - introduction: regenerative medicine and solid organ transplantation from a historical perspective. In: Orlando, G, Lerut, J, Soker, S, Stratta, RJ, editors. Regen med appl organ transplant. Boston: Academic Press; 2014:1–15 pp.10.1016/B978-0-12-398523-1.00001-XSuche in Google Scholar
47. Rose, LF, Wolf, EJ, Brindle, T, Cernich, A, Dean, WK, Dearth, CL, et al.. The convergence of regenerative medicine and rehabilitation: federal perspectives. NPJ Regen Med 2018;3:19. https://doi.org/10.1038/s41536-018-0056-1.Suche in Google Scholar PubMed PubMed Central
48. Roseti, L, Parisi, V, Petretta, M, Cavallo, C, Desando, G, Bartolotti, I, et al.. Scaffolds for bone tissue engineering: state of the art and new perspectives. Mater Sci Eng C 2017;78:1246–62. https://doi.org/10.1016/j.msec.2017.05.017.Suche in Google Scholar PubMed
49. Wei, S, Ma, JX, Xu, L, Gu, XS, Ma, XL. Biodegradable materials for bone defect repair. Mil Med Res 2020;7:54. https://doi.org/10.1186/s40779-020-00280-6.Suche in Google Scholar PubMed PubMed Central
50. Jang, JW, Min, KE, Kim, C, Shin, J, Lee, J, Yi, S. Review: scaffold characteristics, fabrication methods, and biomaterials for the bone tissue engineering. Int J Precis Eng Manuf 2023;24:511–29. https://doi.org/10.1007/s12541-022-00755-7.Suche in Google Scholar
51. Ke, D, Niu, C, Yang, X. Evolution of 3D bioprinting-from the perspectives of bioprinting companies. Bioprinting 2022;25:e00193. https://doi.org/10.1016/j.bprint.2022.e00193.Suche in Google Scholar
52. Moroni, L, Burdick, JA, Highley, C, Lee, SJ, Morimoto, Y, Takeuchi, S, et al.. Biofabrication strategies for 3D in vitro models and regenerative medicine. Nat Rev Mater 2018;3:21–37. https://doi.org/10.1038/s41578-018-0006-y.Suche in Google Scholar PubMed PubMed Central
53. Ji, S, Guvendiren, M. Complex 3D bioprinting methods. APL Bioeng 2021;5:011508. https://doi.org/10.1063/5.0034901.Suche in Google Scholar PubMed PubMed Central
54. Laronda, MM, Rutz, AL, Xiao, S, Whelan, KA, Duncan, FE, Roth, EW, et al.. A bioprosthetic ovary created using 3D printed microporous scaffolds restores ovarian function in sterilized mice. Nat Commun 2017;8:15261. https://doi.org/10.1038/ncomms15261.Suche in Google Scholar PubMed PubMed Central
55. Wang, D, Maharjan, S, Kuang, X, Wang, Z, Mille, LS, Tao, M, et al.. Microfluidic bioprinting of tough hydrogel-based vascular conduits for functional blood vessels. Sci Adv 2022;8:eabq6900. https://doi.org/10.1126/sciadv.abq6900.Suche in Google Scholar PubMed PubMed Central
56. Grounds, MD. Obstacles and challenges for tissue engineering and regenerative medicine: Australian nuances. Clin Exp Pharmacol Physiol 2018;45:390–400. https://doi.org/10.1111/1440-1681.12899.Suche in Google Scholar PubMed
57. Naderi, H, Matin, MM, Bahrami, AR. Review paper: critical issues in tissue engineering: biomaterials, cell sources, angiogenesis, and drug delivery systems. J Biomater Appl 2011;26:383–417. https://doi.org/10.1177/0885328211408946.Suche in Google Scholar PubMed
58. Younes, HM, Kadavil, H, Ismail, HM, Adib, SA, Zamani, S, Alany, RG, et al.. Overview of tissue engineering and drug delivery applications of reactive electrospinning and crosslinking techniques of polymeric nanofibers with highlights on their biocompatibility testing and regulatory aspects. Pharmaceutics 2023;16:32. https://doi.org/10.3390/pharmaceutics16010032.Suche in Google Scholar PubMed PubMed Central
59. Fowler, JL, Ang, LT, Loh, KM. A critical look: challenges in differentiating human pluripotent stem cells into desired cell types and organoids. Wiley Interdiscip Rev Dev Biol 2020;9:e368. https://doi.org/10.1002/wdev.368.Suche in Google Scholar PubMed PubMed Central
60. Guimarães, CF, Gasperini, L, Marques, AP, Reis, RL. The stiffness of living tissues and its implications for tissue engineering. Nat Rev Mater 2020;5:351–70. https://doi.org/10.1038/s41578-019-0169-1.Suche in Google Scholar
61. Worthington, AK, Forsberg, EC. A CRISPR view of hematopoietic stem cells: moving innovative bioengineering into the clinic. Am J Hematol 2022;97:1226–35. https://doi.org/10.1002/ajh.26588.Suche in Google Scholar PubMed PubMed Central
62. Ayanoğlu, FB, Elçin, AE, Elçin, YM. Bioethical issues in genome editing by CRISPR-Cas9 technology. Turk J Biol 2020;44:110–20. https://doi.org/10.3906/biy-1912-52.Suche in Google Scholar PubMed PubMed Central
63. Valenti, MT, Serena, M, Carbonare, LD, Zipeto, D. CRISPR/Cas system: an emerging technology in stem cell research. World J Stem Cell 2019;11:937–56. https://doi.org/10.4252/wjsc.v11.i11.937.Suche in Google Scholar PubMed PubMed Central
64. Sheridan, C. CRISPR therapies march into clinic, but genotoxicity concerns linger. Nat Biotechnol 2021;39:897–9. https://doi.org/10.1038/d41587-021-00017-3.Suche in Google Scholar PubMed
65. Borot, F, Wang, H, Ma, Y, Jafarov, T, Raza, A, Ali, AM, et al.. Gene-edited stem cells enable CD33-directed immune therapy for myeloid malignancies. Proc Natl Acad Sci USA 2019;116:11978–87. https://doi.org/10.1073/pnas.1819992116.Suche in Google Scholar PubMed PubMed Central
66. Frangoul, H, Altshuler, D, Cappellini, MD, Chen, YS, Domm, J, Eustace, BK, et al.. CRISPR-Cas9 gene editing for sickle cell disease and β-thalassemia. N Engl J Med 2021;384:252–60. https://doi.org/10.1530/ey.18.14.4.Suche in Google Scholar
67. Kim, MY, Yu, KR, Kenderian, SS, Ruella, M, Chen, S, Shin, TH, et al.. Genetic inactivation of CD33 in hematopoietic stem cells to enable CAR T cell immunotherapy for acute myeloid leukemia. Cell 2018;173:1439–53.e19. https://doi.org/10.1016/j.cell.2018.05.013.Suche in Google Scholar PubMed PubMed Central
68. Xu, L, Wang, J, Liu, Y, Xie, L, Su, B, Mou, D, et al.. CRISPR-edited stem cells in a patient with HIV and acute lymphocytic leukemia. N Engl J Med 2019;381:1240–7. https://doi.org/10.1056/nejmoa1817426.Suche in Google Scholar PubMed
69. From Pluripotent Stem to CAR T Cells. From pluripotent stem to CAR T cells. Cancer Discov 2018;8:Of5. https://doi.org/10.1158/2159-8290.CD-NB2018-057.Suche in Google Scholar PubMed
70. Kong, Q, Li, Y, Yue, J, Wu, X, Xu, M. Reducing alcohol and/or cocaine-induced reward and toxicity via an epidermal stem cell-based gene delivery platform. Mol Psychiatr 2021;26:5266–76. https://doi.org/10.1038/s41380-021-01043-y.Suche in Google Scholar PubMed PubMed Central
71. Witkowski, TA, Li, B, Andersen, JG, Kumar, B, Mroz, EA, Rocco, JW. Y-27632 acts beyond ROCK inhibition to maintain epidermal stem-like cells in culture. J Cell Sci 2023;136:jcs260990. https://doi.org/10.1242/jcs.260990.Suche in Google Scholar PubMed PubMed Central
72. Hazrati, A, Malekpour, K, Soudi, S, Hashemi, SM. CRISPR/Cas9-engineered mesenchymal stromal/stem cells and their extracellular vesicles: a new approach to overcoming cell therapy limitations. Biomed Pharmacother 2022;156:113943. https://doi.org/10.1016/j.biopha.2022.113943.Suche in Google Scholar PubMed
73. Lee, J, Bayarsaikhan, D, Arivazhagan, R, Park, H, Lim, B, Gwak, P, et al.. CRISPR/Cas9 edited sRAGE-MSCs protect neuronal death in Parkinson’s disease model. Int J Stem Cells 2019;12:114–24. https://doi.org/10.15283/ijsc18110.Suche in Google Scholar PubMed PubMed Central
74. Guo, C, Ma, X, Gao, F, Guo, Y. Off-target effects in CRISPR/Cas9 gene editing. Front Bioeng Biotechnol 2023;11:1143157. https://doi.org/10.3389/fbioe.2023.1143157.Suche in Google Scholar PubMed PubMed Central
75. Yin, H, Kauffman, KJ, Anderson, DG. Delivery technologies for genome editing. Nat Rev Drug Discov 2017;16:387–99. https://doi.org/10.1038/nrd.2016.280.Suche in Google Scholar PubMed
76. Allen, D, Rosenberg, M, Hendel, A. Using synthetically engineered guide RNAs to enhance CRISPR genome editing systems in mammalian cells. Front Genome 2020;2:617910. https://doi.org/10.3389/fgeed.2020.617910.Suche in Google Scholar PubMed PubMed Central
77. Deng, HX, Zhai, H, Shi, Y, Liu, G, Lowry, J, Liu, B, et al.. Efficacy and long-term safety of CRISPR/Cas9 genome editing in the SOD1-linked mouse models of ALS. Commun Biol 2021;4:396. https://doi.org/10.1038/s42003-021-01942-4.Suche in Google Scholar PubMed PubMed Central
78. Srinivasan, M, Thangaraj, SR, Ramasubramanian, K, Thangaraj, PP, Ramasubramanian, KV. Exploring the current trends of artificial intelligence in stem cell therapy: a systematic review. Cureus 2021;13:e20083. https://doi.org/10.7759/cureus.20083.Suche in Google Scholar PubMed PubMed Central
79. Schaub, NJ, Hotaling, NA, Manescu, P, Padi, S, Wan, Q, Sharma, R, et al.. Deep learning predicts function of live retinal pigment epithelium from quantitative microscopy. J Clin Invest 2020;130:1010–23. https://doi.org/10.1172/jci131187.Suche in Google Scholar PubMed PubMed Central
80. Fan, K, Zhang, S, Zhang, Y, Lu, J, Holcombe, M, Zhang, X. A machine learning assisted, label-free, non-invasive approach for somatic reprogramming in induced pluripotent stem cell colony formation detection and prediction. Sci Rep 2017;7:13496. https://doi.org/10.1038/s41598-017-13680-x.Suche in Google Scholar PubMed PubMed Central
81. Shouval, R, Labopin, M, Bondi, O, Mishan-Shamay, H, Shimoni, A, Ciceri, F, et al.. Prediction of allogeneic hematopoietic stem-cell transplantation mortality 100 Days after transplantation using a machine learning algorithm: a European group for blood and marrow transplantation acute leukemia working party retrospective data mining study. J Clin Oncol 2015;33:3144–51. https://doi.org/10.1200/jco.2014.59.1339.Suche in Google Scholar PubMed
82. Juhola, M, Penttinen, K, Joutsijoki, H, Aalto-Setälä, K. Analysis of drug effects on iPSC cardiomyocytes with machine learning. Ann Biomed Eng 2021;49:129–38. https://doi.org/10.1007/s10439-020-02521-0.Suche in Google Scholar PubMed PubMed Central
83. Hirose, T, Kotoku, J, Toki, F, Nishimura, EK, Nanba, D. Label-free quality control and identification of human keratinocyte stem cells by deep learning-based automated cell tracking. Stem Cell 2021;39:1091–100. https://doi.org/10.1002/stem.3371.Suche in Google Scholar PubMed PubMed Central
84. Zhu, Y, Huang, R, Wu, Z, Song, S, Cheng, L, Zhu, R. Deep learning-based predictive identification of neural stem cell differentiation. Nat Commun 2021;12:2614. https://doi.org/10.1038/s41467-021-22758-0.Suche in Google Scholar PubMed PubMed Central
85. Nosrati, H, Nosrati, M. Artificial intelligence in regenerative medicine: applications and implications. Biomimetics 2023;8:442. https://doi.org/10.3390/biomimetics8050442.Suche in Google Scholar PubMed PubMed Central
86. Weese, J, Lorenz, C. Four challenges in medical image analysis from an industrial perspective. Med Image Anal 2016;33:44–9. https://doi.org/10.1016/j.media.2016.06.023.Suche in Google Scholar PubMed
87. Naci, H, Ioannidis, JP. Comparative effectiveness of exercise and drug interventions on mortality outcomes: metaepidemiological study. BMJ 2013;347:f5577. https://doi.org/10.1136/bmj.f5577.Suche in Google Scholar PubMed PubMed Central
88. Molloy, CD, Long, L, Mordi, IR, Bridges, C, Sagar, VA, Davies, EJ, et al.. Exercise-based cardiac rehabilitation for adults with heart failure – 2023 Cochrane systematic review and meta-analysis. Eur J Heart Fail 2023;25:2263–73.10.1002/ejhf.3046Suche in Google Scholar PubMed
89. Troosters, T, Janssens, W, Demeyer, H, Rabinovich, RA. Pulmonary rehabilitation and physical interventions. Eur Respir Rev 2023;32. https://doi.org/10.1183/16000617.0222-2022.Suche in Google Scholar PubMed PubMed Central
90. Weber, C, Thai, V, Neuheuser, K, Groover, K, Christ, O. Efficacy of physical therapy for the treatment of lateral epicondylitis: a meta-analysis. BMC Musculoskelet Disord 2015;16:223.10.1186/s12891-015-0665-4Suche in Google Scholar PubMed PubMed Central
91. Hannemann, PF, Mommers, EH, Schots, JP, Brink, PR, Poeze, M. The effects of low-intensity pulsed ultrasound and pulsed electromagnetic fields bone growth stimulation in acute fractures: a systematic review and meta-analysis of randomized controlled trials. Arch Orthop Trauma Surg 2014;134:1093–106.10.1007/s00402-014-2014-8Suche in Google Scholar PubMed
92. Supriya, R, Singh, KP, Gao, Y, Gu, Y, Baker, JS. Effect of exercise on secondary sarcopenia: a comprehensive literature review. Biology 2021;11:51. https://doi.org/10.3390/biology11010051.Suche in Google Scholar PubMed PubMed Central
93. Rando, TA, Ambrosio, F. Regenerative rehabilitation: applied biophysics meets stem cell therapeutics. Cell Stem Cell 2018;22:306–9. https://doi.org/10.1016/j.stem.2018.02.003.Suche in Google Scholar PubMed PubMed Central
94. Ambrosio, F, Ferrari, RJ, Distefano, G, Plassmeyer, JM, Carvell, GE, Deasy, BM, et al.. The synergistic effect of treadmill running on stem-cell transplantation to heal injured skeletal muscle. Tissue Eng 2010;16:839–49. https://doi.org/10.1089/ten.tea.2009.0113.Suche in Google Scholar
95. Hwang, DH, Shin, HY, Kwon, MJ, Choi, JY, Ryu, BY, Kim, BG. Survival of neural stem cell grafts in the lesioned spinal cord is enhanced by a combination of treadmill locomotor training via insulin-like growth factor-1 signaling. J Neurosci 2014;34:12788–800. https://doi.org/10.1523/jneurosci.5359-13.2014.Suche in Google Scholar
96. De Lisio, M, Parise, G. Characterization of the effects of exercise training on hematopoietic stem cell quantity and function. J Appl Physiol 2012;113:1576–84. https://doi.org/10.1152/japplphysiol.00717.2012.Suche in Google Scholar PubMed PubMed Central
97. Hwang, DH, Park, HH, Shin, HY, Cui, Y, Kim, BG. Insulin-like growth factor-1 receptor dictates beneficial effects of treadmill training by regulating survival and migration of neural stem cell grafts in the injured spinal cord. Exp Neurobiol 2018;27:489. https://doi.org/10.5607/en.2018.27.6.489.Suche in Google Scholar PubMed PubMed Central
98. Oh, B, Wu, YW, Swaminathan, V, Lam, V, Ding, J, George, PM. Modulating the electrical and mechanical microenvironment to guide neuronal stem cell differentiation. Adv Sci 2021;8:2002112. https://doi.org/10.1002/advs.202002112.Suche in Google Scholar PubMed PubMed Central
99. Zhang, P, Zhang, C, Li, J, Han, J, Liu, X, Yang, H. The physical microenvironment of hematopoietic stem cells and its emerging roles in engineering applications. Stem Cell Res Ther 2019;10:1–13. https://doi.org/10.1186/s13287-019-1422-7.Suche in Google Scholar PubMed PubMed Central
100. Bouchentouf, M, Benabdallah, BF, Mills, P, Tremblay, JP. Exercise improves the success of myoblast transplantation in mdx mice. Neuromuscul Disord 2006;16:518–29. https://doi.org/10.1016/j.nmd.2006.06.003.Suche in Google Scholar PubMed
101. Sun, Y, Chen, CS, Fu, J. Forcing stem cells to behave: a biophysical perspective of the cellular microenvironment. Annu Rev Biophys 2012;41:519–42. https://doi.org/10.1146/annurev-biophys-042910-155306.Suche in Google Scholar PubMed PubMed Central
102. Grigoryan, G. Molecular-Cellular mechanisms of plastic restructuring produced by an enriched environment. Effects on learning and memory. Neurochem J 2021;15:226–39. https://doi.org/10.1134/s1819712421030041.Suche in Google Scholar
103. Karelina, K, Schneiderman, K, Shah, S, Fitzgerald, J, Cruz, RV, Oliverio, R, et al.. Moderate intensity treadmill exercise increases survival of newborn hippocampal neurons and improves neurobehavioral outcomes after traumatic brain injury. J Neurotrauma 2021;38:1858–69. https://doi.org/10.1089/neu.2020.7389.Suche in Google Scholar PubMed PubMed Central
104. Hannan, A. Environmental enrichment and brain repair: harnessing the therapeutic effects of cognitive stimulation and physical activity to enhance experience‐dependent plasticity. Neuropathol Appl Neurobiol 2014;40:13–25. https://doi.org/10.1111/nan.12102.Suche in Google Scholar PubMed
105. Zhan, Y, Li, MZ, Yang, L, Feng, XF, Lei, JF, Zhang, N, et al.. The three-phase enriched environment paradigm promotes neurovascular restorative and prevents learning impairment after ischemic stroke in rats. Neurobiol Dis 2020;146:105091. https://doi.org/10.1016/j.nbd.2020.105091.Suche in Google Scholar PubMed
106. Dunnett, SB. Neural tissue transplantation, repair, and rehabilitation. Handb Clin Neurol 2013;110:43–59. https://doi.org/10.1016/b978-0-444-52901-5.00004-6.Suche in Google Scholar
107. Clinch, S, Busse, M, Döbrössy, MD, Dunnett, SB. Rehabilitation training in neural restitution. Prog Brain Res 2017;230:305–29. https://doi.org/10.1016/bs.pbr.2016.12.006.Suche in Google Scholar PubMed
108. Duncan, PW, Horner, RD, Reker, DM, Samsa, GP, Hoenig, H, Hamilton, B, et al.. Adherence to postacute rehabilitation guidelines is associated with functional recovery in stroke. Stroke 2002;33:167–78. https://doi.org/10.1161/hs0102.101014.Suche in Google Scholar PubMed
109. Hochstenbach, J, Prigatano, G, Mulder, T. Patients’ and relatives’ reports of disturbances 9 months after stroke: subjective changes in physical functioning, cognition, emotion, and behavior. Arch Phys Med Rehabil 2005;86:1587–93. https://doi.org/10.1016/j.apmr.2004.11.050.Suche in Google Scholar PubMed
110. Aravamudhan, S, Bellamkonda, RV. Toward a convergence of regenerative medicine, rehabilitation, and neuroprosthetics. J Neurotrauma 2011;28:2329–47. https://doi.org/10.1089/neu.2010.1542.Suche in Google Scholar PubMed
111. Mu, J, Bakreen, A, Juntunen, M, Korhonen, P, Oinonen, E, Cui, L, et al.. Combined adipose tissue-derived mesenchymal stem cell therapy and rehabilitation in experimental stroke. Front Neurol 2019;10:235. https://doi.org/10.3389/fneur.2019.00235.Suche in Google Scholar PubMed PubMed Central
112. Zhang, YX, Yuan, MZ, Cheng, L, Lin, LZ, Du, HW, Chen, RH, et al.. Treadmill exercise enhances therapeutic potency of transplanted bone mesenchymal stem cells in cerebral ischemic rats via anti-apoptotic effects. BMC Neurosci 2015;16:1–8. https://doi.org/10.1186/s12868-015-0196-9.Suche in Google Scholar PubMed PubMed Central
113. Sasaki, Y, Sasaki, M, Kataoka-Sasaki, Y, Nakazaki, M, Nagahama, H, Suzuki, J, et al.. Synergic effects of rehabilitation and intravenous infusion of mesenchymal stem cells after stroke in rats. Phys Ther 2016;96:1791–8. https://doi.org/10.2522/ptj.20150504.Suche in Google Scholar PubMed
114. Ito, A, Kubo, N, Liang, N, Aoyama, T, Kuroki, H. Regenerative rehabilitation for stroke recovery by inducing synergistic effects of cell therapy and neurorehabilitation on motor function: a narrative review of pre-clinical studies. Int J Mol Sci 2020;21:3135. https://doi.org/10.3390/ijms21093135.Suche in Google Scholar PubMed PubMed Central
115. Seo, JH, Kim, H, Park, ES, Lee, JE, Kim, DW, Kim, HO, et al.. Environmental enrichment synergistically improves functional recovery by transplanted adipose stem cells in chronic hypoxic-ischemic brain injury. Cell Transplant 2013;22:1553–68. https://doi.org/10.3727/096368912x662390.Suche in Google Scholar
116. Cho, S-R, Suh, H, Yu, JH, Kim, H, Seo, JH, Seo, CH. Astroglial activation by an enriched environment after transplantation of mesenchymal stem cells enhances angiogenesis after hypoxic-ischemic brain injury. Int J Mol Sci 2016;17:1550. https://doi.org/10.3390/ijms17091550.Suche in Google Scholar PubMed PubMed Central
117. Hicks, A, Hewlett, K, Windle, V, Chernenko, G, Ploughman, M, Jolkkonen, J, et al.. Enriched environment enhances transplanted subventricular zone stem cell migration and functional recovery after stroke. Neuroscience 2007;146:31–40. https://doi.org/10.1016/j.neuroscience.2007.01.020.Suche in Google Scholar PubMed
118. Hou, S, Rabchevsky, AG. Autonomic consequences of spinal cord injury. Compr Physiol 2014;4:1419–53. https://doi.org/10.1002/cphy.c130045.Suche in Google Scholar PubMed
119. Kabu, S, Gao, Y, Kwon, BK, Labhasetwar, V. Drug delivery, cell-based therapies, and tissue engineering approaches for spinal cord injury. J Contr Release 2015;219:141–54. https://doi.org/10.1016/j.jconrel.2015.08.060.Suche in Google Scholar PubMed PubMed Central
120. Fan, X, Wang, JZ, Lin, XM, Zhang, L. Stem cell transplantation for spinal cord injury: a meta-analysis of treatment effectiveness and safety. Neural Regen Res 2017;12:815. https://doi.org/10.4103/1673-5374.206653.Suche in Google Scholar PubMed PubMed Central
121. Lu, P, Kadoya, K, Tuszynski, MH. Axonal growth and connectivity from neural stem cell grafts in models of spinal cord injury. Curr Opin Neurobiol 2014;27:103–9. https://doi.org/10.1016/j.conb.2014.03.010.Suche in Google Scholar PubMed
122. Fenrich, KK, Hallworth, BW, Vavrek, R, Raposo, PJ, Misiaszek, JE, Bennett, DJ, et al.. Self-directed rehabilitation training intensity thresholds for efficient recovery of skilled forelimb function in rats with cervical spinal cord injury. Exp Neurol 2021;339:113543. https://doi.org/10.1016/j.expneurol.2020.113543.Suche in Google Scholar PubMed
123. Tashiro, S, Nakamura, M, Okano, H. Regenerative rehabilitation and stem cell therapy targeting chronic spinal cord injury: a review of preclinical studies. Cells 2022;11:685. https://doi.org/10.3390/cells11040685.Suche in Google Scholar PubMed PubMed Central
124. Tashiro, S, Nishimura, S, Iwai, H, Sugai, K, Zhang, L, Shinozaki, M, et al.. Functional recovery from neural stem/progenitor cell transplantation combined with treadmill training in mice with chronic spinal cord injury. Sci Rep 2016;6:30898. https://doi.org/10.1038/srep30898.Suche in Google Scholar PubMed PubMed Central
125. Younsi, A, Zheng, G, Scherer, M, Riemann, L, Zhang, H, Tail, M, et al.. Treadmill training improves survival and differentiation of transplanted neural precursor cells after cervical spinal cord injury. Stem Cell Res 2020;45:101812. https://doi.org/10.1016/j.scr.2020.101812.Suche in Google Scholar PubMed
126. Dugan, EA, Jergova, S, Sagen, J. Mutually beneficial effects of intensive exercise and GABAergic neural progenitor cell transplants in reducing neuropathic pain and spinal pathology in rats with spinal cord injury. Exp Neurol 2020;327:113208. https://doi.org/10.1016/j.expneurol.2020.113208.Suche in Google Scholar PubMed
127. Thornton, MA, Mehta, MD, Morad, TT, Ingraham, KL, Khankan, RR, Griffis, KG, et al.. Evidence of axon connectivity across a spinal cord transection in rats treated with epidural stimulation and motor training combined with olfactory ensheathing cell transplantation. Exp Neurol 2018;309:119–33. https://doi.org/10.1016/j.expneurol.2018.07.015.Suche in Google Scholar PubMed PubMed Central
128. Guo, M, Wu, L, Song, Z, Yang, B. Enhancement of neural stem cell proliferation in rats with spinal cord injury by a combination of repetitive transcranial magnetic stimulation (rTMS) and human umbilical cord blood mesenchymal stem cells (hUCB-MSCs). Med Sci Mon Int Med J Exp Clin Res 2020;26:e924445-1. https://doi.org/10.12659/msm.924445.Suche in Google Scholar
129. Sarveazad, A, Janzadeh, A, Taheripak, G, Dameni, S, Yousefifard, M, Nasirinezhad, F. Co-administration of human adipose-derived stem cells and low-level laser to alleviate neuropathic pain after experimental spinal cord injury. Stem Cell Res Ther 2019;10:183. https://doi.org/10.1186/s13287-019-1269-y.Suche in Google Scholar PubMed PubMed Central
130. Lawrence, RC, Felson, DT, Helmick, CG, Arnold, LM, Choi, H, Deyo, RA, et al.. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States: Part II. Arthritis Rheum 2008;58:26–35. https://doi.org/10.1002/art.23176.Suche in Google Scholar PubMed PubMed Central
131. Litwic, A, Edwards, MH, Dennison, EM, Cooper, C. Epidemiology and burden of osteoarthritis. Br Med Bull 2013;105:185–99. https://doi.org/10.1093/bmb/lds038.Suche in Google Scholar PubMed PubMed Central
132. Kon, E, Filardo, G, Roffi, A, Andriolo, L, Marcacci, M. New trends for knee cartilage regeneration: from cell-free scaffolds to mesenchymal stem cells. Curr Rev Musculoskelet Med 2012;5:236–43. https://doi.org/10.1007/s12178-012-9135-x.Suche in Google Scholar PubMed PubMed Central
133. Gupta, PK, Das, AK, Chullikana, A, Majumdar, AS. Mesenchymal stem cells for cartilage repair in osteoarthritis. Stem Cell Res Ther 2012;3:1–9. https://doi.org/10.1186/scrt116.Suche in Google Scholar PubMed PubMed Central
134. Woo, SL, Kuei, SC, Amiel, D, Gomez, M, Hayes, W, White, F, et al.. The effect of prolonged physical training on the properties of long bone: a study of Wolff’s Law. J Bone Joint Surg Am 1981;63:780–7. https://doi.org/10.2106/00004623-198163050-00013.Suche in Google Scholar
135. Schreivogel, S, Kuchibhotla, V, Knaus, P, Duda, GN, Petersen, A. Load-induced osteogenic differentiation of mesenchymal stromal cells is caused by mechano-regulated autocrine signaling. J Tissue Eng Regen Med 2019;13:1992–2008. https://doi.org/10.1002/term.2948.Suche in Google Scholar PubMed
136. Grier, W, Moy, A, Harley, B. Cyclic tensile strain enhances human mesenchymal stem cell Smad 2/3 activation and tenogenic differentiation in anisotropic collagen-glycosaminoglycan scaffolds. Eur Cell Mater 2017;33:227. https://doi.org/10.22203/ecm.v033a17.Suche in Google Scholar
137. Chen, X, Yan, J, He, F, Zhong, D, Yang, H, Pei, M, et al.. Mechanical stretch induces antioxidant responses and osteogenic differentiation in human mesenchymal stem cells through activation of the AMPK-SIRT1 signaling pathway. Free Radic Biol Med 2018;126:187–201. https://doi.org/10.1016/j.freeradbiomed.2018.08.001.Suche in Google Scholar PubMed PubMed Central
138. Cao, W, Lin, W, Cai, H, Chen, Y, Man, Y, Liang, J, et al.. Dynamic mechanical loading facilitated chondrogenic differentiation of rabbit BMSCs in collagen scaffolds. Regen Biomater 2019;6:99–106. https://doi.org/10.1093/rb/rbz005.Suche in Google Scholar PubMed PubMed Central
139. Pelaez, D, Arita, N, Cheung, HS. Extracellular signal-regulated kinase (ERK) dictates osteogenic and/or chondrogenic lineage commitment of mesenchymal stem cells under dynamic compression. Biochem Biophys Res Commun 2012;417:1286–91. https://doi.org/10.1016/j.bbrc.2011.12.131.Suche in Google Scholar PubMed
140. Thompson, M, Woods, K, Newberg, J, Oxford, JT, Uzer, G. Low-intensity vibration restores nuclear YAP levels and acute YAP nuclear shuttling in mesenchymal stem cells subjected to simulated microgravity. NPJ Microgravity 2020;6:35. https://doi.org/10.1038/s41526-020-00125-5.Suche in Google Scholar PubMed PubMed Central
141. Wei, FY, Leung, KS, Li, G, Qin, J, Chow, SKH, Huang, S, et al.. Low intensity pulsed ultrasound enhanced mesenchymal stem cell recruitment through stromal derived factor-1 signaling in fracture healing. PLoS One 2014;9:e106722. https://doi.org/10.1371/journal.pone.0106722.Suche in Google Scholar PubMed PubMed Central
142. Hui, CFF, Chan, CW, Yeung, HY, Lee, KM, Qin, L, Li, G, et al.. Low-intensity pulsed ultrasound enhances posterior spinal fusion implanted with mesenchymal stem cells-calcium phosphate composite without bone grafting. Spine 2011;36:1010–6. https://doi.org/10.1097/brs.0b013e318205c5f5.Suche in Google Scholar
143. Kusuyama, J, Bandow, K, Shamoto, M, Kakimoto, K, Ohnishi, T, Matsuguchi, T. Low intensity pulsed ultrasound (LIPUS) influences the multilineage differentiation of mesenchymal stem and progenitor cell lines through ROCK-Cot/Tpl2-MEK-ERK signaling pathway. J Biol Chem 2014;289:10330–44. https://doi.org/10.1074/jbc.m113.546382.Suche in Google Scholar
144. Dong, L, Song, Y, Zhang, Y, Zhao, W, Wang, C, Lin, H, et al.. Mechanical stretch induces osteogenesis through the alternative activation of macrophages. J Cell Physiol 2021;236:6376–90. https://doi.org/10.1002/jcp.30312.Suche in Google Scholar PubMed
145. Charoenpanich, A, Wall, ME, Tucker, CJ, Andrews, DM, Lalush, DS, Dirschl, DR, et al.. Cyclic tensile strain enhances osteogenesis and angiogenesis in mesenchymal stem cells from osteoporotic donors. Tissue Eng 2014;20:67–78. https://doi.org/10.1089/ten.tea.2013.0006.Suche in Google Scholar
146. Jiang, YN, Zhao, J, Chu, FT, Jiang, YY, Tang, GH. Tension-loaded bone marrow stromal cells potentiate the paracrine osteogenic signaling of co-cultured vascular endothelial cells. Biol Open 2018;7:bio032482. https://doi.org/10.1242/bio.032482.Suche in Google Scholar PubMed PubMed Central
147. Labarthe, DR. Epidemiology and prevention of cardiovascular diseases: a global challenge. Massachusetts, US: Jones & Bartlett Learning; 2010.Suche in Google Scholar
148. Fiuza-Luces, C, Santos-Lozano, A, Joyner, M, Carrera-Bastos, P, Picazo, O, Zugaza, JL, et al.. Exercise benefits in cardiovascular disease: beyond attenuation of traditional risk factors. Nat Rev Cardiol 2018;15:731–43. https://doi.org/10.1038/s41569-018-0065-1.Suche in Google Scholar PubMed
149. Patel, H, Alkhawam, H, Madanieh, R, Shah, N, Kosmas, CE, Vittorio, TJ. Aerobic vs anaerobic exercise training effects on the cardiovascular system. World J Cardiol 2017;9:134–8. https://doi.org/10.4330/wjc.v9.i2.134.Suche in Google Scholar PubMed PubMed Central
150. Marino, F, Scalise, M, Cianflone, E, Salerno, L, Cappetta, D, Salerno, N, et al.. Physical exercise and cardiac repair: the potential role of nitric Oxide in boosting stem cell regenerative biology. Antioxidants 2021;10:1002. https://doi.org/10.3390/antiox10071002.Suche in Google Scholar PubMed PubMed Central
151. Xu, X, Wan, W, Powers, AS, Li, J, Ji, LL, Lao, S, et al.. Effects of exercise training on cardiac function and myocardial remodeling in post myocardial infarction rats. J Mol Cell Cardiol 2008;44:114–22. https://doi.org/10.1016/j.yjmcc.2007.10.004.Suche in Google Scholar PubMed PubMed Central
152. Bito, V, De Waard, MC, Biesmans, L, Lenaerts, I, Ozdemir, S, van Deel, E, et al.. Early exercise training after myocardial infarction prevents contractile but not electrical remodelling or hypertrophy. Cardiovasc Res 2010;86:72–81. https://doi.org/10.1093/cvr/cvp381.Suche in Google Scholar PubMed
153. DeSantiago, J, Bare, D, Semenov, I, Minshall, R, Geenen, D, Wolska, B, et al.. Excitation–contraction coupling in ventricular myocytes is enhanced by paracrine signaling from mesenchymal stem cells. J Mol Cell Cardiol 2012;52:1249–56. https://doi.org/10.1016/j.yjmcc.2012.03.008.Suche in Google Scholar PubMed PubMed Central
154. Shake, JG, Gruber, PJ, Baumgartner, WA, Senechal, G, Meyers, J, Redmond, JM, et al.. Mesenchymal stem cell implantation in a swine myocardial infarct model: engraftment and functional effects. Ann Thorac Surg 2002;73:1919–26. https://doi.org/10.1016/s0003-4975(02)03517-8.Suche in Google Scholar PubMed
155. Timmers, L, Lim, SK, Arslan, F, Armstrong, JS, Hoefer, IE, Doevendans, PA, et al.. Reduction of myocardial infarct size by human mesenchymal stem cell conditioned medium. Stem Cell Res 2008;1:129–37. https://doi.org/10.1016/j.scr.2008.02.002.Suche in Google Scholar PubMed
156. Mills, WR, Mal, N, Kiedrowski, MJ, Unger, R, Forudi, F, Popovic, ZB, et al.. Stem cell therapy enhances electrical viability in myocardial infarction. J Mol Cell Cardiol 2007;42:304–14. https://doi.org/10.1016/j.yjmcc.2006.09.011.Suche in Google Scholar PubMed
157. Cosmo, S, Francisco, JC, Cunha, RC, Macedo, RM, Faria-Neto, JR, Simeoni, R, et al.. Effect of exercise associated with stem cell transplantation on ventricular function in rats after acute myocardial infarction. Rev Bras Cir Cardiovasc 2012;27:542–51. https://doi.org/10.5935/1678-9741.20120096.Suche in Google Scholar PubMed
158. de Freitas, JS, Neves, CA, Del Carlo, RJ, Belfort, FG, Lavorato, VN, Silame-Gomes, LH, et al.. Effects of exercise training and stem cell therapy on the left ventricle of infarcted rats. Rev Port Cardiol 2019;38:649–56. https://doi.org/10.1016/j.repc.2019.02.013.Suche in Google Scholar PubMed
159. Ashraf, SS, Hosseinpour Sarmadi, V, Larijani, G, Naderi Garahgheshlagh, S, Ramezani, S, Moghadamifar, S, et al.. Regenerative medicine improve neurodegenerative diseases. Cell Tissue Bank 2023;24:639–50. https://doi.org/10.1007/s10561-022-10062-0.Suche in Google Scholar PubMed
160. Nakayama, KH, Alcazar, C, Yang, G, Quarta, M, Paine, P, Doan, L, et al.. Rehabilitative exercise and spatially patterned nanofibrillar scaffolds enhance vascularization and innervation following volumetric muscle loss. NPJ Regen Med 2018;3:16. https://doi.org/10.1038/s41536-018-0054-3.Suche in Google Scholar PubMed PubMed Central
161. Kruse, FE, Cursiefen, C. Surgery of the cornea: corneal, limbal stem cell and amniotic membrane transplantation. Dev Ophthalmol 2008;41:159–70. https://doi.org/10.1159/000131087.Suche in Google Scholar PubMed
162. Singh, A, Raghav, A, Shiekh, PA, Kumar, A. Transplantation of engineered exosomes derived from bone marrow mesenchymal stromal cells ameliorate diabetic peripheral neuropathy under electrical stimulation. Bioact Mater 2021;6:2231–49. https://doi.org/10.1016/j.bioactmat.2021.01.008.Suche in Google Scholar PubMed PubMed Central
163. Sivandzade, F, Cucullo, L. Regenerative stem cell therapy for neurodegenerative diseases: an overview. Int J Mol Sci 2021;22. https://doi.org/10.3390/ijms22042153.Suche in Google Scholar PubMed PubMed Central
164. Pang, C, Ibrahim, A, Bulstrode, NW, Ferretti, P. An overview of the therapeutic potential of regenerative medicine in cutaneous wound healing. Int Wound J 2017;14:450–9. https://doi.org/10.1111/iwj.12735.Suche in Google Scholar PubMed PubMed Central
165. Aurora, AB, Olson, EN. Immune modulation of stem cells and regeneration. Cell Stem Cell 2014;15:14–25. https://doi.org/10.1016/j.stem.2014.06.009.Suche in Google Scholar PubMed PubMed Central
166. Ajmal, L, Ajmal, S, Ajmal, M, Nawaz, G. Organ regeneration through stem cells and tissue engineering. Cureus 2023;15:e34336. https://doi.org/10.7759/cureus.34336.Suche in Google Scholar PubMed PubMed Central
167. Mohananey, D, Sarau, A, Kumar, R, Lewandowski, D, Abreu-Sosa, SM, Nathan, S, et al.. Role of physical activity and cardiac rehabilitation in patients undergoing hematopoietic stem cell transplantation. JACC CardioOncol 2021;3:17–34. https://doi.org/10.1016/j.jaccao.2021.01.008.Suche in Google Scholar PubMed PubMed Central
168. Armenian, SH, Sun, CL, Vase, T, Ness, KK, Blum, E, Francisco, L, et al.. Cardiovascular risk factors in hematopoietic cell transplantation survivors: role in development of subsequent cardiovascular disease. Blood 2012;120:4505–12. https://doi.org/10.1182/blood-2012-06-437178.Suche in Google Scholar PubMed PubMed Central
169. Armenian, SH, Sun, CL, Mills, G, Teh, JB, Francisco, L, Durand, JB, et al.. Predictors of late cardiovascular complications in survivors of hematopoietic cell transplantation. Biol Blood Marrow Transplant 2010;16:1138–44. https://doi.org/10.1016/j.bbmt.2010.02.021.Suche in Google Scholar PubMed PubMed Central
170. Chow, EJ, Wong, K, Lee, SJ, Cushing-Haugen, KL, Flowers, ME, Friedman, DL, et al.. Late cardiovascular complications after hematopoietic cell transplantation. Biol Blood Marrow Transplant 2014;20:794–800. https://doi.org/10.1016/j.bbmt.2014.02.012.Suche in Google Scholar PubMed PubMed Central
171. Chow, EJ, Baker, KS, Lee, SJ, Flowers, ME, Cushing-Haugen, KL, Inamoto, Y, et al.. Influence of conventional cardiovascular risk factors and lifestyle characteristics on cardiovascular disease after hematopoietic cell transplantation. J Clin Oncol 2014;32:191–8. https://doi.org/10.1200/jco.2013.52.6582.Suche in Google Scholar PubMed PubMed Central
172. Chow, EJ, Mueller, BA, Baker, KS, Cushing-Haugen, KL, Flowers, ME, Martin, PJ, et al.. Cardiovascular hospitalizations and mortality among recipients of hematopoietic stem cell transplantation. Ann Intern Med 2011;155:21–32. https://doi.org/10.7326/0003-4819-155-1-201107050-00004.Suche in Google Scholar
173. Scott, JM, Armenian, S, Giralt, S, Moslehi, J, Wang, T, Jones, LW. Cardiovascular disease following hematopoietic stem cell transplantation: pathogenesis, detection, and the cardioprotective role of aerobic training. Crit Rev Oncol Hematol 2016;98:222–34. https://doi.org/10.1016/j.critrevonc.2015.11.007.Suche in Google Scholar
174. Danaher, EH, Ferrans, C, Verlen, E, Ravandi, F, van Besien, K, Gelms, J, et al.. Fatigue and physical activity in patients undergoing hematopoietic stem cell transplant. Oncol Nurs Forum 2006;33:614–24. https://doi.org/10.1188/06.onf.614-624.Suche in Google Scholar
175. Hayes, S, Davies, PS, Parker, T, Bashford, J. Total energy expenditure and body composition changes following peripheral blood stem cell transplantation and participation in an exercise programme. Bone Marrow Transplant 2003;31:331–8. https://doi.org/10.1038/sj.bmt.1703867.Suche in Google Scholar
176. Baumann, FT, Zimmer, P, Finkenberg, K, Hallek, M, Bloch, W, Elter, T. Influence of endurance exercise on the risk of pneumonia and Fever in leukemia and lymphoma patients undergoing high dose chemotherapy. A pilot study. J Sports Sci Med 2012;11:638–42.Suche in Google Scholar
177. Hayes, S, Davies, PS, Parker, T, Bashford, J, Newman, B. Quality of life changes following peripheral blood stem cell transplantation and participation in a mixed-type, moderate-intensity, exercise program. Bone Marrow Transplant 2004;33:553–8. https://doi.org/10.1038/sj.bmt.1704378.Suche in Google Scholar
178. McGavock, JM, Hastings, JL, Snell, PG, McGuire, DK, Pacini, EL, Levine, BD, et al.. A forty-year follow-up of the Dallas Bed Rest and Training study: the effect of age on the cardiovascular response to exercise in men. J Gerontol A Biol Sci Med Sci 2009;64:293–9. https://doi.org/10.1093/gerona/gln025.Suche in Google Scholar
179. Saltin, B, Blomqvist, G, Mitchell, JH, Johnson, RLJr, Wildenthal, K, Chapman, CB, et al.. Response to exercise after bed rest and after training. Circulation 1968;38:Vii1–78. https://doi.org/10.1161/01.cir.38.5s7.vii-1.Suche in Google Scholar
180. Campbell, KL, Winters-Stone, KM, Wiskemann, J, May, AM, Schwartz, AL, Courneya, KS, et al.. Exercise guidelines for cancer survivors: consensus statement from international multidisciplinary roundtable. Med Sci Sports Exerc 2019;51:2375–90. https://doi.org/10.1249/mss.0000000000002116.Suche in Google Scholar
181. Schmitz, KH, Campbell, AM, Stuiver, MM, Pinto, BM, Schwartz, AL, Morris, GS, et al.. Exercise is medicine in oncology: engaging clinicians to help patients move through cancer. CA A Cancer J Clin 2019;69:468–84. https://doi.org/10.3322/caac.21579.Suche in Google Scholar
182. Dimeo, FC, Tilmann, MH, Bertz, H, Kanz, L, Mertelsmann, R, Keul, J. Aerobic exercise in the rehabilitation of cancer patients after high dose chemotherapy and autologous peripheral stem cell transplantation. Cancer 1997;79:1717–22. https://doi.org/10.1002/(sici)1097-0142(19970501)79:9<1717::aid-cncr12>3.3.co;2-2.10.1002/(SICI)1097-0142(19970501)79:9<1717::AID-CNCR12>3.0.CO;2-0Suche in Google Scholar
183. Coleman, EA, Coon, SK, Kennedy, RL, Lockhart, KD, Stewart, CB, Anaissie, EJ, et al.. Effects of exercise in combination with epoetin alfa during high-dose chemotherapy and autologous peripheral blood stem cell transplantation for multiple myeloma. Oncol Nurs Forum 2008;35:E53–61. https://doi.org/10.1188/08.onf.e53-e61.Suche in Google Scholar
184. Liang, Y, Zhou, M, Wang, F, Wu, Z. Exercise for physical fitness, fatigue and quality of life of patients undergoing hematopoietic stem cell transplantation: a meta-analysis of randomized controlled trials. Jpn J Clin Oncol 2018;48:1046–57. https://doi.org/10.1093/jjco/hyy144.Suche in Google Scholar PubMed
185. Jarden, M, Baadsgaard, MT, Hovgaard, DJ, Boesen, E, Adamsen, L. A randomized trial on the effect of a multimodal intervention on physical capacity, functional performance and quality of life in adult patients undergoing allogeneic SCT. Bone Marrow Transplant 2009;43:725–37. https://doi.org/10.1038/bmt.2009.27.Suche in Google Scholar PubMed
186. Morishita, S, Kaida, K, Setogawa, K, Kajihara, K, Ishii, S, Ikegame, K, et al.. Safety and feasibility of physical therapy in cytopenic patients during allogeneic haematopoietic stem cell transplantation. Eur J Cancer Care 2013;22:289–99. https://doi.org/10.1111/ecc.12027.Suche in Google Scholar PubMed
187. Persoon, S, Kersten, MJ, van der Weiden, K, Buffart, LM, Nollet, F, Brug, J, et al.. Effects of exercise in patients treated with stem cell transplantation for a hematologic malignancy: a systematic review and meta-analysis. Cancer Treat Rev 2013;39:682–90. https://doi.org/10.1016/j.ctrv.2013.01.001.Suche in Google Scholar PubMed
188. Ambrosio, F, Russell, A. Regenerative rehabilitation: a call to action. J Rehabil Res Dev 2010;47:xi–xv. https://doi.org/10.1682/jrrd.2010.03.0021.Suche in Google Scholar PubMed
189. Lim, R, Banerjee, A, Biswas, R, Chari, AN, Raghavan, S. Mechanotransduction through adhesion molecules: emerging roles in regulating the stem cell niche. Front Cell Dev Biol 2022;10:966662. https://doi.org/10.3389/fcell.2022.966662.Suche in Google Scholar PubMed PubMed Central
190. Engler, AJ, Sen, S, Sweeney, HL, Discher, DE. Matrix elasticity directs stem cell lineage specification. Cell 2006;126:677–89. https://doi.org/10.1016/j.cell.2006.06.044.Suche in Google Scholar PubMed
191. Qu, F, Guilak, F, Mauck, RL. Cell migration: implications for repair and regeneration in joint disease. Nat Rev Rheumatol 2019;15:167–79. https://doi.org/10.1038/s41584-018-0151-0.Suche in Google Scholar PubMed PubMed Central
192. De Trizio, E, Brennan, CS. The business of human embryonic stem cell research and an international analysis of relevant laws. J BioLaw Bus 2004;7:14–22.Suche in Google Scholar
193. Godfrey, KJ, Mathew, B, Bulman, JC, Shah, O, Clement, S, Gallicano, GI. Stem cell-based treatments for Type 1 diabetes mellitus: bone marrow, embryonic, hepatic, pancreatic and induced pluripotent stem cells. Diabet Med 2012;29:14–23. https://doi.org/10.1111/j.1464-5491.2011.03433.x.Suche in Google Scholar PubMed
194. Ghaedi, M, Niklason, LE. Human pluripotent stem cells (iPSC) generation, culture, and differentiation to lung progenitor cells. Methods Mol Biol 2019;1576:55–92. https://doi.org/10.1007/7651_2016_11.Suche in Google Scholar PubMed PubMed Central
195. Mathews, DJ, Donovan, PJ, Harris, J, Lovell-Badge, R, Savulescu, J, Faden, R. Pluripotent stem cell-derived gametes: truth and (potential) consequences. Cell Stem Cell 2009;5:11–4. https://doi.org/10.1016/j.stem.2009.06.005.Suche in Google Scholar PubMed PubMed Central
196. Marks, PW, Witten, CM, Califf, RM. Clarifying stem-cell therapy’s benefits and risks. N Engl J Med 2017;376:1007–9. https://doi.org/10.1056/nejmp1613723.Suche in Google Scholar
197. Cyranoski, D. The potent effects of Japan’s stem-cell policies. Nature 2019;573:482–5. https://doi.org/10.1038/d41586-019-02847-3.Suche in Google Scholar PubMed
198. Konomi, K, Tobita, M, Kimura, K, Sato, D. New Japanese initiatives on stem cell therapies. Cell Stem Cell 2015;16:350–2. https://doi.org/10.1016/j.stem.2015.03.012.Suche in Google Scholar PubMed
199. Gao, J, Gao, C. Development and regulation of stem cell-based therapies in China. Cell Prolif 2022;55:e13217. https://doi.org/10.1111/cpr.13217.Suche in Google Scholar PubMed PubMed Central
200. Thirabanjasak, D, Tantiwongse, K, Thorner, PS. Angiomyeloproliferative lesions following autologous stem cell therapy. J Am Soc Nephrol 2010;21:1218–22. https://doi.org/10.1681/asn.2009111156.Suche in Google Scholar PubMed PubMed Central
201. Kuriyan, AE, Albini, TA, Townsend, JH, Rodriguez, M, Pandya, HK, Leonard, RE, et al.. Vision loss after intravitreal injection of autologous “stem cells” for AMD. N Engl J Med 2017;376:1047–53. https://doi.org/10.1056/nejmoa1609583.Suche in Google Scholar PubMed PubMed Central
© 2024 the author(s), published by De Gruyter, Berlin/Boston
This work is licensed under the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
Artikel in diesem Heft
- Frontmatter
- Reviews
- Understanding autoimmune response after SARS-CoV-2 infection and the pathogenesis/mechanisms of long COVID
- Improving lung allograft function in the early post-operative period through the inhibition of pyroptosis
- The occurrence mechanism, assessment, and non-pharmacological treatment of dyspnea
- Regenerative rehabilitation: a novel multidisciplinary field to maximize patient outcomes
- Advances in the prerequisite and consequence of STING downstream signalosomes
- Complement factor H in molecular regulation of angiogenesis
Artikel in diesem Heft
- Frontmatter
- Reviews
- Understanding autoimmune response after SARS-CoV-2 infection and the pathogenesis/mechanisms of long COVID
- Improving lung allograft function in the early post-operative period through the inhibition of pyroptosis
- The occurrence mechanism, assessment, and non-pharmacological treatment of dyspnea
- Regenerative rehabilitation: a novel multidisciplinary field to maximize patient outcomes
- Advances in the prerequisite and consequence of STING downstream signalosomes
- Complement factor H in molecular regulation of angiogenesis

