Abstract
Lepidium meyenii (Walp.), commonly called maca, is an Andean crop belonging to the Brassicaceae family. Maca hypocotils are habitually consumed as customary food as well as traditional remedies for pathological conditions such as infertility. Moreover, the characterization of maca extracts revealed the presence of compounds that are able to modulate the nervous system. Aimed to evaluate the efficacy of L. meyenii in persistent pain, the present study analyzed the effects of a commercial root extract from maca in different animal models reproducing the most common causes of chronic painful pathologies. A qualitative characterization of this commercial extract by high performance liquid chromatography-mass spectrometry and tandem mass spectrometry analyses allowed us to confirm the presence of some macamides known as bioactive constituents of this root and the absence of the main aromatic glucosinolates. The acute oral administration of maca extract is able to reduce mechanical hypersensitivity and postural unbalance induced by the intra-articular injection of monoiodoacetate and the chronic-constriction injury of the sciatic nerve. Furthermore, L. meyenii extract reverts pain threshold alterations evoked by oxaliplatin and paclitaxel. A good safety profile in mice and rats was shown. In conclusion, the present maca extract could be considered as a therapeutic opportunity to relieve articular and neuropathic pain.
1 Introduction
Persistent pain is defined as pain lasting for a period of 6 months or more. Primary care settings in Asia, Africa, Europe, and in the Americas had patients reporting persistent pain in approximately a quarter of the population [1]. Arthritis-related diseases, nerve trauma or compression, and neuropathies induced by metabolic syndromes or neurotoxic drugs are among the most common causes of persistent pain. Clinical evidence of neuropathic or inflammatory pain are hyperalgesia, thermal allodynia and spontaneous pain [2]. The choice of therapy depends on the type of pain, its intensity, and how the patient responds to treatment. Drug therapy, however, continues to be inadequate to completely control persistent pain [3]. In this context, plant-derived products characterized by a safety profile (especially those used as food) emerge as interesting resources for individuating novel therapeutic possibilities to manage chronic pain.
Maca (Lepidium meyenii Walp.) is a traditional Andean crop belonging to the Brassicaceae family and it is traditionally used for its nutritional and medicinal properties [4]. Maca’s hypocotils are usually consumed fresh or dehydrated after boiling in water or milk, after being made into juices, maca coffee, or alcoholic beverages [5], [6]. The tubers of L. meyenii have been widely investigated during these last years, pointing out the copresence of two main groups of phytochemicals, the glucosinolates and the macamides and macaenes. The first group is typical of Brassicaceae; in maca, the main forms are aromatic glucosinolates [7], suggested by some authors as chemical markers to explain some biological effects of this plant [8] and denied by others. Over the last few years, different studies that have focused on the analysis and purification of macamides from this plant have been available in the literature. To this aim, lipophilic extracts of roots, such as those by petroleum ether [9] or by n-hexane [10], were selected. Nevertheless, Ganzera et al. [11] highlighted, using high performance liquid chromatography (HPLC), the presence of some macamides also in root powder methanol extract. Because the macamides have not been found in other Lepidium species, they have been proposed as phytochemical markers of maca [10]. Recently, it has been highlighted that some aromatic glucosinolates, abundant in fresh roots, consistently decrease in the dried material because they are involved in the ex novo biosynthesis of macamides, together with free fatty acids [12].
Maca extracts are used for the treatment of a wide range of pathological conditions such as sexual infertility, osteoporosis, and benign prostatic hyperplasia [13], [14]. On the contrary, limited evidence of efficacy against persistent pain has been described up to now [15]. However, in vitro studies confirmed macaenes family components’ involvement in enhancing cannabinoid transmission [16]. The cholinergic stimulation induced by L. meyenii extract (mainly by polyphenols) has been related to memory improvement [17] as well as antioxidant properties that may sustain the antidepressant-like effects of the plant extract [18]. Moreover, maca improves the antioxidant cell machinery [19]. Cannabinoid and cholinergic systems [20], [21], as well as redox unbalance [22], [23], [24], [25], play a relevant role in persistent pain processes. Maca consumption has been associated with lower serum levels of the proinflammatory and pro-algic cytokine IL-6 in populations living in the Peruvian Central Andes [8]. Starting from the theoretical approach that maca is able to modulate endogenous systems normally involved in pain control, the aim of this study was to evaluate the efficacy, after acute intake (oral administration), of a commercial water extract of L. meyenii, controlled in its composition by high performance liquid chromatography with diode array detector (HPLC-DAD) and high performance liquid chromatography-mass spectrometry (HPLC-MS). The study was carried out in rat or mouse models of persistent pain induced by the intra-articular injection of monoiodoacetate (MIA), chronic constriction injury (CCI) of the sciatic nerve and chemotherapy-induced neuropathy.
2 Materials and methods
2.1 Sample preparation
The dried extract was purchased from KOS Spa, Italy, and was obtained from the roots of maca (L. meyenii Walp.) harvested in the Peruvian highlands in South America. According to the supplier’s documentation, the extract was obtained by water extraction, and to achieve a final powdered homogeneous sample, maltodextrins were added, obtaining a final 4:1 ratio of dry extract/excipient. Further technical specifications were not available from the supplier. This sample was fractionated as described subsequently: 50 mg of powder were dissolved in 1 mL of water (W) or acetonitrile/methanol (AM) 1:1 v/v; after 1 h of stirring, the solid residues were removed by centrifugation and the solutions recovered to obtain W and AM extracts, respectively. These samples were then analyzed by HPLC-MS and tandem mass spectrometry (MS-MS).
2.2 HPLC-MS and MS-MS analysis
The HPLC-MS analyses were done using a Platin Blue UHPLC (Knauer, Germany) directly coupled to a 6410B triple quadrupole mass spectrometer (Agilent Technologies, USA) through an ESI interface. Two different columns were used: a Gemini C18 150×2 mm, 3 μm, 110 Å, and a Kinetex HILIC 150×2.1 mm, 2.6 μm, 100 Å (Phenomenex, Italy). For each column, a multistep linear gradient elution method was applied, in both cases using a 0.1% formic acid solution (eluent A) and acetonitrile, containing 0.1% formic acid (eluent B). The elution programs for the two columns are reported in the Supplementary information Table S1.
At the end of the run, the columns were re-equilibrated at the starting condition for 10 min. The injection volume was 10 μL; the column oven temperature was 30°C. The diode-array UV detector recorded the full UV spectra in the range 210–500 nm, and the 240, 280 and 330 nm wavelengths. MS acquisitions were performed in scan mode (100–800 m/z scan range at 650 ms scan−1), switching between positive and negative polarities. The ESI interface parameters were: nebulizer gas 30 psi, gas flow 10 L min−1, gas temperature 310°C, capillary voltages 4.2 kV and 4.0 kV in positive and negative modes, respectively.
2.3 Animals
Male Sprague-Dawley rats (Envigo, Varese, Italy) weighing approximately 200–250 g and male Swiss albino mice (Envigo, Varese, Italy) weighing approximately 23–25 g at the beginning of the experimental procedure were used. Animals were housed in CeSAL (Centro Stabulazione Animali da Laboratorio, University of Florence) and used at least 1 week after their arrival. Four rats and five mice were housed per cage (size 26–41 cm); animals were fed with standard laboratory diet and tap water ad libitum, and kept at 23°C±1°C with a 12-h light/dark cycle, light at 7 a.m. All animal manipulations were carried out according to Directive 2010/63/EU of the European parliament and of the European Union council (22 September 2010) on the protection of animals used for scientific purposes. The ethical policy of the University of Florence complies with the Guide for the Care and Use of Laboratory Animals of the U.S. National Institutes of Health (NIH Publication No. 85-23, revised 1996; University of Florence assurance number: A5278-01). Formal approval to conduct the experiments described was obtained from the Italian Ministry of Health (No. 54/2014-B) and from the Animal Subjects Review Board of the University of Florence. Experiments involving animals have been reported according to ARRIVE guidelines [26].
2.4 MIA-induced osteoarthritis
Unilateral osteoarthritis was also induced in rats by injection of MIA (Sigma-Aldrich, Italy) into the knee joint according to a described method [27], [28]. Exactly 2 mg MIA in 25 μL saline was delivered into the left articular cavity. Control rats received 25 μL of saline solution (day 1) in the knee joint. Behavioral and biochemical measures were performed on day 14.
2.5 CCI-induced neuropathy
Neuropathy was induced in rats according to a procedure described by Bennett and Xie [29]. Another group of rats were subjected to sham surgery in which the sciatic nerve was only exposed but not ligated. The animals were allowed to recover from surgery and then housed one per cage with free access to water and standard laboratory chow. Behavioral tests were performed on day 14.
2.6 Oxaliplatin-induced neuropathy
Mice were treated with 2.4 mg kg−1 oxaliplatin, administered i.p. for five consecutive days every week for 2 weeks (10 i.p. injections) [30], [31]. Oxaliplatin was dissolved in 5% glucose solution. Control animals received an equivalent volume of vehicle. Behavioral tests were performed on day 14.
2.7 Paclitaxel-induced neuropathy
Mice were treated with 2.0 mg kg−1 paclitaxel, administered i.p. four times on alternating days 1, 3, 5, and 7. Paclitaxel was dissolved in 10% Cremophor E-L of saline solution [32]. Control animals received an equivalent volume of vehicle. Behavioral and biochemical tests were performed on day 14.
2.8 Maca extract administration
Maca extract (KOS Spa, Italy) was suspended in 1% carboxymethylcellulose sodium salt and administered orally. Acute administrations (0.5, 1.5, 3 and 10 g kg−1; conventionally, 1 kg was considered as corresponding to 10 mL) were performed on day 14 after damage induction (CCI and MIA) or anticancer drug treatment start (oxaliplatin and paclitaxel). Behavioral tests were performed before and over time (60 min) after maca extract administration.
2.9 Paw pressure test
The nociceptive threshold in the rat was determined with an analgesimeter (Ugo Basile, Varese, Italy), according to a method described by Leighton et al. [33]. Rats scoring less than 40 g or more than 75 g during the test before drug administration were rejected (25%). For analgesia measures, mechanical pressure application was stopped at 120 g.
2.10 Incapacitance test
Weight-bearing changes were measured using an incapacitance apparatus (Linton Instrumentation, UK) detecting changes in postural equilibrium after a hind limb injury [34]. Rats were trained to stand on their hind paws in a box with an inclined plane (65° from horizontal). The value considered for each animal was the mean of five consecutive measurements. Data are expressed as the difference between the weight applied on the limb contralateral to the injury and the weight applied on the ipsilateral one (ΔWeight).
2.11 Cold plate test
The animals (mice) were placed in a stainless steel box (12 cm×20 cm×10 cm) with a cold plate as floor. The temperature of the cold plate was kept constant at 4°C±1°C. Pain-related behaviors (i.e. lifting and licking of the hind paw) were observed and the time(s) of the first sign was recorded. The cutoff time of the latency of paw lifting or licking was set at 60 s [35].
2.12 Rotarod test
The rotarod apparatus for rats (Ugo Basile, Varese, Italy) consisted of a base platform and a rotating rod with a diameter of 6 cm and a nonslippery surface. The rod was placed at a height of 25 cm from the base. The rod, 36 cm in length, was divided into four equal sections using five disks. Thus, up to four rats were tested simultaneously on the apparatus, with a rod-rotating speed of 10 rpm. The integrity of motor coordination was assessed on the basis of the number of falls from the rod in 30 s. Those animals scoring less than three and more than six falls in the pretest were rejected. The performance time was measured every 15 min for four times. Animals with normal coordination progressively reduced the number of falls during the experimental sessions.
2.13 Statistical analysis
Results were expressed as means±SEM and the analysis of variance was performed by ANOVA test. A Bonferroni’s significant difference procedure was used as a post hoc comparison. P values less than 0.05 were considered significant. Data were analyzed using the “Origin 8.1” software.
3 Results
Several analytical methods were proposed to investigate macamides [11] and glucosinolates [7] recognized as the main bioactive phytochemicals of maca. According to these findings, the aim of this work was to check the presence of these two groups of molecules in the commercial extract selected for this in vivo study. The screening was done by HPLC-DAD-MS working on two extracts, at different polarities, obtained from the same commercial sample. The chromatographic profiles of the W extract on the Gemini® column did not reveal the presence of analytes detectable in the 220–330 nm range. The same test on the HILIC column, specifically selected to highlight the glucosinolates, confirmed the absence of these compounds and the presence of several diglycosides and oligoglycosides, as expected. On the contrary, the AM extract after the HPLC-DAD-MS screening in positive and negative ionization mode, showed the best result in positive mode. By LC-MS several macamides were detected by their intense [M+H]+ and/or [M+Na]+ ions. Supplementary Figure S1A shows the base peak chromatogram in the 300–450 m/z range, selected in agreement with a recent article focused on the comparison of macamides in different batches of maca hypocotyls [9]. To summarize, some representative extract ion profiles at the selected masses are reported in Supplementary Figure S1B, whereas all the detected macamides are listed in Table 1.
List of the detected macamides in the AM extract of L. meyenii in positive ion mode and potential identification.
| Rt (min) | [M+H]+ (% intensity) | [M+Na]+ (% intensity) | Identity |
|---|---|---|---|
| 42.20 | 384 (5) | 406 (95) | N-Benzyl-9-oxo-(12Z, 15Z)-octadecadienamide |
| 42.36 | 384 (5) | 406 (95) | N-Benzyl-13-oxo-(9E, 11E)-octadecadienamide |
| 42.54 | 386 (5) | 408 (95) | N-Benzyl-9-oxo-12Z-octadecanamide |
| 43.09 | 368 (80) | 390 (20) | N-Benzyl-13-oxo-(9Z, 12Z, 15Z)-octadecatrienamide |
| 43.61 | 370 (100) | – | N-Benzyl-(9Z, 12Z)-octadecadienamide |
| 44.20 | 346 (50) | 368 (50) | N-Benzyl-hexadecanamide |
| 44.29 | 372 (80) | 394 (20) | N-Benzyl-9Z-octadecanamide |
| 42.88 | 374 (100) | – | N-Benzyl-octadecanamide |
The DAD profile also allowed us to point out a group of lipophilic metabolites with retention time values close to that of macamides and characterized by their UV-Vis spectra and ion molecular weight as reported in Supplementary Figure S2. To date, further information is not available on these compounds.
The pain reliever effect of L. meyenii extract was evaluated in several models of persistent pain by behavioral tests. In Figure 1, the effect of MIA administration in rats is shown. Fourteen days after MIA injection, the paw pressure test lead us to appreciate that the weight tolerated on the ipsilateral paw was significantly reduced compared with the contralateral (data not shown). A total of 10 g kg−1 of L. meyenii extract time dependently reduced MIA-evoked mechanical hypersensitivity increasing the weight tolerated up to 70.8±2.2 g (Figure 1A). Figure 1B shows the difference between the weight burdened on the contralateral and the ipsilateral limb. ∆Weight was significantly increased in MIA+vehicle-treated rats (67.6±1.6 g) in comparison to vehicle+vehicle-treated rats (5.0±1.5 g). Lepidium meyenii (10 g kg−1) was able to statistically reduce hind limb weight-bearing alterations peaking 15 min after treatment (41.1±6.0 g). Lepidium meyenii extract was also effective on neuropathic pain induced in rats by lesion to the peripheral nervous system. Rat paw pressure test revealed that 14 days after CCI, the weight tolerated on the ipsilateral paw significantly decreased (43.3±3.3 g) with respect to the contralateral (68.3±3.1 g) (data not shown). As depicted in Figure 2A, 10 g kg−1 of L. meyenii completely reverted the CCI-induced hyperalgesia for 30 min after administration. Moreover, the effect of the extract was also evaluated by measuring spontaneous pain of CCI rats with the incapacitance test. As shown in Figure 2B, 10 g kg−1 of L. meyenii extract reduced the difference between the weight burdened on the contralateral and the ipsilateral limb by approximately 45%. A different kind of neuropathic pain was induced in mice by the administration of the neurotoxic drugs oxaliplatin and paclitaxel. The platin-derivative anticancer compound was repeatedly administered and, on day 14, it induced a lowered threshold to cold stimuli that was measured by cold plate test (Figure 3A the licking latency of oxaliplatin+vehicle-treated mice is 11.6±0.7 s vs. 21.2±1.2 s of vehicle+vehicle-treated mice). Lepidium meyenii extract, acutely administered in a dose range of 0.5–10 g kg−1, reverted oxaliplatin-induced neuropathic pain in a dose-dependent manner. A total of 10 g kg−1 evoked the longest significant effect peaking 30 min after administration (20±0.9 s). Moreover, 3 and 1.5 g kg−1 induced a statistically significant effect. In Figure 3B, the effect of repeated paclitaxel administrations in mice is shown. On day 14, the taxane derivative decreased the time that animals tolerate on a cold surface (11.5±0.9 s, paclitaxel+vehicle vs. 23.4±1.7 s, vehicle+vehicle). Lepidium meyenii extract was administered at a dose range of 0.5–10 g kg−1. As depicted in Figure 3B, 0.5, 1.5, and 3 g kg−1 doses induced a significant effect, whereas the highest dose significantly increased licking latency peaking (21.6±1.6 s) 30 min after the administration. Table 2 displays the effect of the maximum tested dose of L. meyenii (10 g kg−1) on animals’ motor coordination as evaluated by rotarod test. Fifteen minutes after extract administration, the animals were placed on the rotating rod (10 rpm) for 10 min and the number of falls was counted. Administration of L. meyenii extract did not induce significant alterations of motor coordination in comparison with the control group.
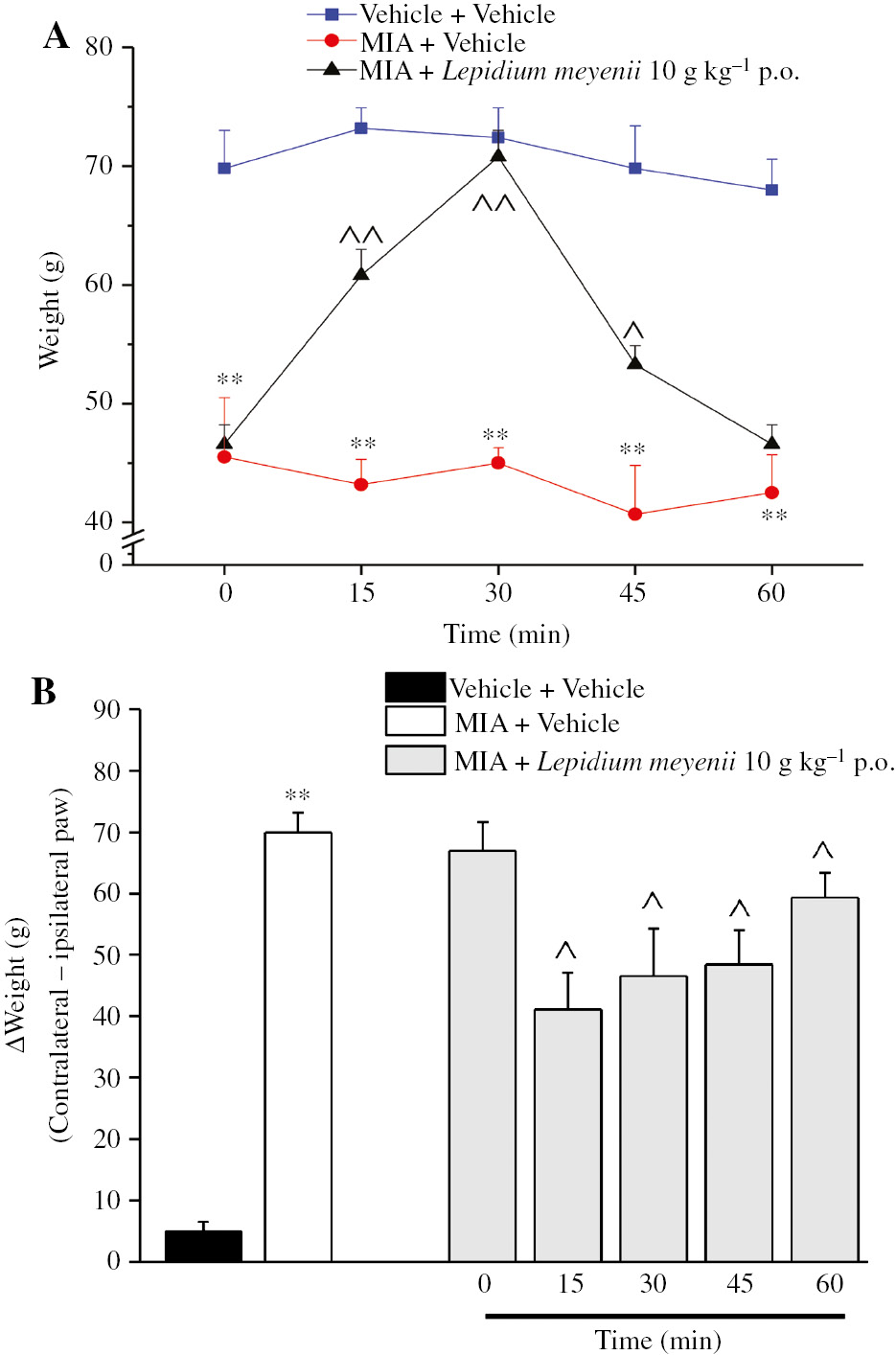
Articular pain.
On day 14 after MIA injection, L. meyenii extract (10 g kg−1) was p.o. administered and pain threshold was evaluated over time. (A) Response to a noxious mechanical stimulus evaluated by paw pressure test on the ipsilateral paw. (B) Hind limb weight-bearing alterations were measured by incapacitance test. n=10; *P<0.01 versus vehicle+vehicle-treated rats; ^P<0.05 and ^^P<0.01 versus MIA+vehicle-treated rats.
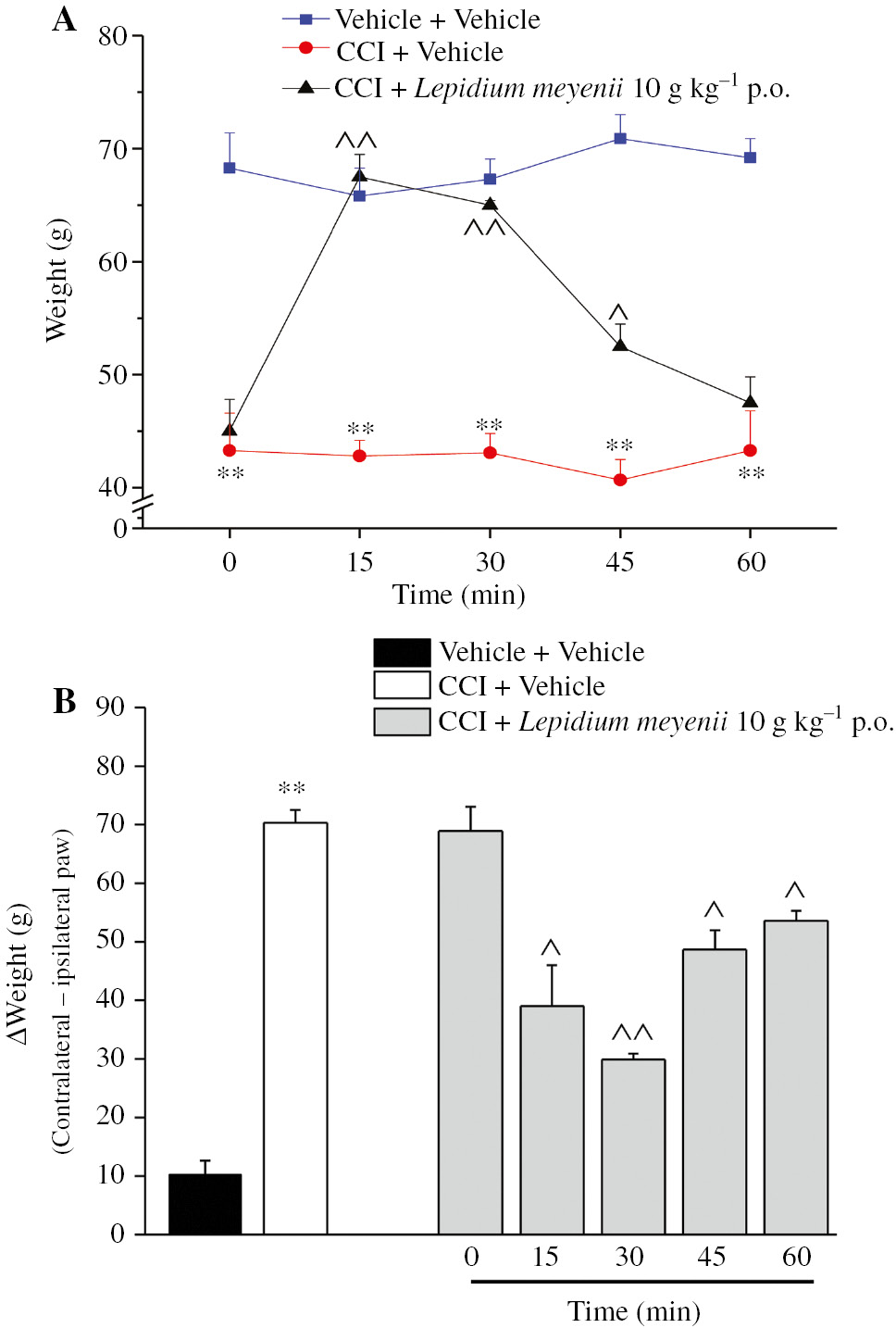
CCI-induced neuropathic pain.
On day 14 after nerve surgery, L. meyenii extract (10 g kg−1) was p.o. administered and pain threshold was evaluated over time. (A) Response to a noxious mechanical stimulus evaluated by paw pressure test on the ipsilateral paw. (B) Hind limb weight-bearing alterations were measured by incapacitance test 14 days after surgery. n=10; **P<0.01 versus vehicle+vehicle-treated rats; ^P<0.05 and ^^P<0.01 versus CCI+vehicle-treated rats.
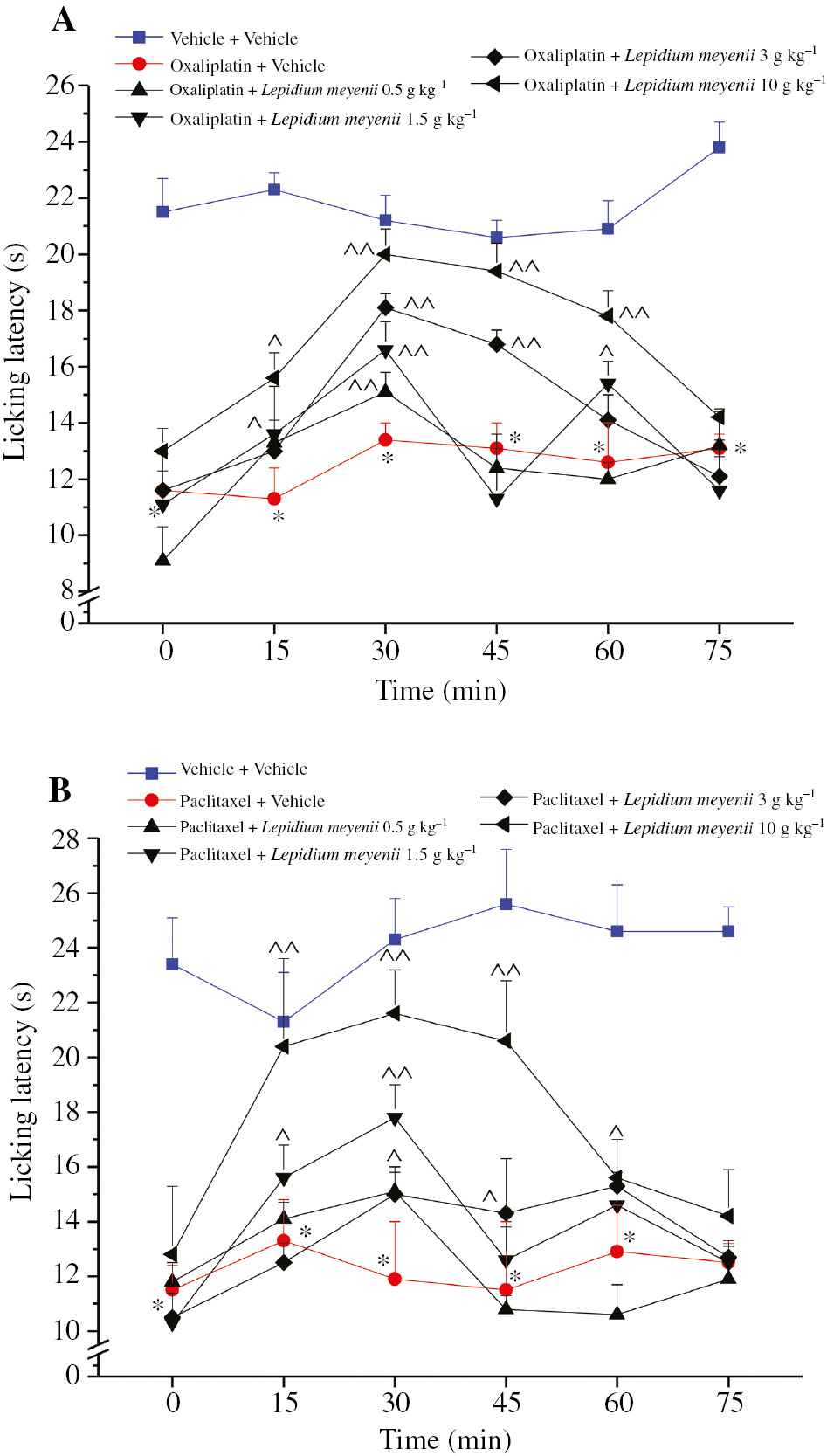
Chemotherapy-induced neuropathic pain.
(A) Oxaliplatin-induced neuropathic pain. Mice were treated i.p. daily with 2.4 mg kg−1 oxaliplatin. (B) Paclitaxel-induced neuropathic pain. Mice were treated i.p. (on days 1, 3, 5, and 7) with 2 mg kg−1 paclitaxel. The response to a thermal non-noxious stimulus was evaluated by cold plate test. Behavioral test was performed on day 14. Before and after L. meyenii extract (0.5–10 g kg−1) p.o. administration. n=10; *P<0.05 versus vehicle+vehicle; ^P<0.05 and ^^P<0.01 versus oxaliplatin+vehicle; ^P<0.05 and ^^P<0.01 versus paclitaxel+vehicle.
Effect of L. meyenii extract on mouse motor coordination (rotarod test).
| Treatment | Dosage (g kg−1) | Falls number (min) | ||||
|---|---|---|---|---|---|---|
| 0 | 15 | 30 | 45 | 60 | ||
| Vehicle | 7.0±0.8 | 5.0±1.0 | 1.0±0.2 | 1.0±0.4 | 0.0±0.0 | |
| Lepidium meyenii | 10 | 5.3±1.3 | 2.0±1.0 | 0.6±0.3 | 1.6±0.3 | 0.0±0.0 |
Animals received L. meyenii extract (10 g kg−1) per os; 15 min (time 0) after the administration they were placed on the rotating rod (10 rpm) for 30 s and the number of falls was evaluated. The performance time was measured every 15 min to evaluate the ability of the animals to acquire the movement. Each value represents the mean±SEM of eight rats per group, performed in two different experimental sets. P>0.05 between groups.
4 Discussion
The present results describe the pain-relieving effects of an aqueous extract of L. meyenii, in three different models of persistent pain. In particular, the intra-articular injection of MIA provides a monolateral osteoarthritis characterized by a persistent inflammatory pain which, starting on day 14 after injection, possesses a neuropathic component [36]. Ten gram per kilogram of maca extract reduces pain induced by a mechanical noxious stimulus on the ipsilateral paw. Moreover, maca-dependent pain relief allows also to reduce postural unbalance, an outward of osteoarthritis progression, as measured by hind limb weight bearing alterations [34]. The anti-neuropathic properties of the L. meyenii extract are confirmed after the ligation of the sciatic nerve, a neuropathy model where the alterations of the nerve morphology are accompanied by remarkable inflammatory state emerged in terms of cellular infiltrate and edema [37], [38]. Maca extract, at maximum dosage, is able to revert mechanical hypersensitivity for 30 min, after injection, as shown by paw pressure test. Ten gram per kilogram of L. meyenii significantly reduces the postural unbalance induced by the nerve ligation as revealed by the incapacitance test. Our results also demonstrate that a single oral administration of maca extract (10 g kg−1) is able to completely revert the thermal allodynia induced in mice after oxaliplatin or paclitaxel chronic treatment. The platinum and the taxane derivatives are used for treatment of several types of cancer as colorectal cancer, lung cancer or ovarian cancer [39], [40]. Their chronic administration causes a peripheral neuropathy characterized by hypersensitivity to cold stimuli [41], [42]. These models of neuropathic pain are free from inflammatory components and are characterized by specific molecular alteration of the peripheral and central nervous system [35]. The maintenance of efficacy in these different kinds of pain suggests an interesting pharmacodynamic profile. The dosages used are quite high but compatible with the use as food supplement. Furthermore, no signs of evident toxicity are highlighted in all treatment groups; in particular, as shown by the rotarod test performances, no motor or neurological alteration was detectable. This maca sample was obtained using an aqueous extraction of the root, as declared in the label, and consequently was not rich in lyphophilic components. Nevertheless, the high sensitivity of a mass spectrometer, differently from the DAD, allowed to detect the unexpected presence of several macamides tentatively identified according to their molecular weight [9].
Furthermore, by use of the HILIC column, suitable to analyzing very polar compounds, the absence of glucosinolates in our sample was confirmed. This finding is in agreement with a recent work that correlates the low content of glucosinolates in dried maca roots to the increase of macamides after the drying process. The hydrolytic phenomena associated with open-air drying are responsible of the formation of the precursors of macamides, which continue to be synthetized until a water content close to 13% is achieved [12]. Even if in low amounts, the macamides can contribute to the observed effect in rats.
At the end, it is worth noting the presence of a group of unknown metabolites with UV-Vis spectra indicating a chromophoric system that can indicate a possible antioxidant capacity that can contribute to the biological effects of this commercial maca sample. At the same time, the shape of these spectra did not confirm the presence of typical flavonoids or flavones, some of them previously found in maca. In vitro studies have demonstrated that macaenes and macamides are able to inhibit the FAAH enzyme, normally responsible for inactivation of anandamide, the main endogenous endocannabinoid. Anandamide is involved in several physiological processes such as painful sensitivity and neuroprotection [16]. Selective FAAH inhibitors enhance anandamide endogenous levels that cause antinociceptive effect by agonism on the CB2 receptor [21]. Furthermore, many preclinical studies demonstrated that FAAH inhibitors reduce chronic pain without the development of tolerance after repeated administrations [43]. The absence of flavonoids in the present extract was confirmed by the investigation on the UV-vis and mass spectra registered, respectively, by HPLC-DAD and by MS at different fragmentation energies. Only a recent work cited two new flavolignans [44]. By the mass experiments, it was possible to exclude the presence of these compounds in our extract. At the same time, other known phenolic compounds were also reported in the same work, as pinoresinol and tricine were not detected. These results strongly suggest the exclusion of both cholinergic system modulation and antioxidant effects of phenolic compounds from the pharmacodynamic mechanisms. The absence of the glucosinolates, confirmed by mass spectrometric analyses, allowed us to exclude the contribution of these phytochemicals for the biological effect observed for the maca extract.
In conclusion, L. meyenii extract is effective as a pain reliever in different models of persistent pain, both in those with a predominant inflammatory component and in those with a predominant neuropathic aspect. The absence of evident toxicity, confirmed by its common use as a customary refreshment as well as a traditional remedy by Peruvian people, shows that L. meyenii extract is a possible treatment against osteoarticular and neuropathic pain.
Acknowledgments
This research was funded by the Italian Ministry of Instruction, University and Research (MIUR) and by the University of Florence.
Disclosure statement: No competing financial interests exist.
References
1. Goldberg DS, McGee SJ. Pain as a global public health priority. BMC Public Health 2012;11:770.10.1186/1471-2458-11-770Search in Google Scholar PubMed PubMed Central
2. Sandkuhler J. Models and mechanisms of hyperalgesia and allodynia. Physiol Rev 2009;89:707–58.10.1152/physrev.00025.2008Search in Google Scholar PubMed
3. Xu B, Descalzi G, Ye HR, Zhuo M, Wang YW. Translational investigation and treatment of neuropathic pain. Mol Pain 2012;9:8–15.10.1186/1744-8069-8-15Search in Google Scholar PubMed PubMed Central
4. Valerio LG, Jr, Gonzales GF. Toxicological aspects of the South American herbs cat’s claw (Uncaria tomentosa) and maca (Lepidium meyenii): a critical synopsis. Toxicol Rev 2005;24:11–35.10.2165/00139709-200524010-00002Search in Google Scholar PubMed
5. Ochoa C, Ugent D. Maca (Lepidium meyenii Walp.; Brassicaceae): a nutritious root crop of the central Andes. Econ Bot 2011;55:344–5.10.1007/BF02866557Search in Google Scholar
6. Quirós CF, Aliaga R. Maca. In: Herman M, Heller J, editors. Andean roots and tubers: ahipa arracacha, maca and yacon. Rome: International Plant Genetic Resources Institute, 1997:173–98.Search in Google Scholar
7. Yabar E, Pedreschi R, Chirinos R, Campos D. Glucosinolate content and myrosinase activity evolution in three maca (Lepidium meyenii Walp.) ecotypes during preharvest, harvest and postharvest drying. Food Chem 2011;127:1576–83.10.1016/j.foodchem.2011.02.021Search in Google Scholar
8. Gonzales GF. Ethnobiology and ethnopharmacology of Lepidium meyenii (maca), a plant from the Peruvian Highlands. Evid Based Complement Alternat Med 2012;2012:193496.10.1155/2012/193496Search in Google Scholar PubMed PubMed Central
9. Pan Y, Zhang J, Li H, Wang YZ, Li WY. Characteristic fingerprinting based on macamides for discrimination of maca (Lepidium meyenii) by LC-MS/MS and multivariate statistical analysis. J Sci Food Agric 2016;96:4475–83.10.1002/jsfa.7660Search in Google Scholar PubMed
10. Chain FE, Grau A, Martins JC, Catalan CA. Macamides from wild ‘maca’, Lepidium meyenii Walpers (Brassicaceae). Phytochem Lett 2014;8:145–8.10.1016/j.phytol.2014.03.005Search in Google Scholar
11. Ganzera M, Zhao J, Muhammad I, Khan IA. Chemical profiling and standardization of Lepidium meyenii (maca) by reversed phase high performance liquid chromatography. Chem Pharm Bull 2002;50:988–91.10.1248/cpb.50.988Search in Google Scholar PubMed
12. Esparza E, Hadzicha A, Kofera W, Mithöfer A, Cosio EG. Bioactive maca (Lepidium meyenii) alkamides are a result of traditional Andean postharvest drying practices. Phytochemistry 2015;116:138–48.10.1016/j.phytochem.2015.02.030Search in Google Scholar PubMed
13. Gasco M, Villegas L, Yucra S, Rubio J, Gonzales GF. Dose-response effect of red maca (Lepidium meyenii) on benign prostatic hyperplasia induced by testosterone enanthate. Phytomedicine 2007;14:460–4.10.1016/j.phymed.2006.12.003Search in Google Scholar PubMed
14. Wang ZQ, Porreca F, Cuzzocrea S, Galen K, Lightfoot R, Masini E, et al. Newly identified role for superoxide in inflammatory pain. J Pharmacol Exp Ther 2004;309:869–78.10.1124/jpet.103.064154Search in Google Scholar PubMed
15. Mehta K, Gala J, Bhasale S, Naik S, Modak M, Thakur H, et al. Comparison of glucosamine sulfate and a polyherbal supplement for the relief of osteoarthritis of the knee: a randomized controlled trial [ISRCTN25438351]. BMC Complement Altern Med 2007;7:34.10.1186/1472-6882-7-34Search in Google Scholar PubMed PubMed Central
16. Almukadi H, Wu H, Böhlke M, Kelley CJ, Maher TJ, Pino-Figueroa A. The macamide N-3-methoxybenzyl-linoleamide is a time-dependent fatty acid amide hydrolase (FAAH) inhibitor. Mol Neurobiol 2013;48:333–9.10.1007/s12035-013-8499-2Search in Google Scholar PubMed
17. Rubio J, Yucra S, Gasco M, Gonzales GF. Dose-response of black maca (Lepidium meyenii) in mice with memory impairment induced by ethanol. Toxicol Mech Methods 2011;21:628–38.10.3109/15376516.2011.583294Search in Google Scholar PubMed
18. Ai Z, Cheng AF, Yu YT, Yu LJ, Jin W. Antidepressant-like behavioral, anatomical, and biochemical effects of petroleum ether extract from maca (Lepidium meyenii) in mice exposed to chronic unpredictable mild stress. J Med Food 2014;17:535–42.10.1089/jmf.2013.2950Search in Google Scholar PubMed PubMed Central
19. Vecera R, Orolin J, Skottová N, Kazdová L, Oliyarnik O, Ulrichová J, et al. The influence of maca (Lepidium meyenii) on antioxidant status, lipid and glucose metabolism in rat. Plant Foods Hum Nutr 2007;62:59–63.10.1007/s11130-007-0042-zSearch in Google Scholar PubMed
20. Bartolini A, Di Cesare Mannelli L, Ghelardini C. Analgesic and antineuropathic drugs acting through central cholinergic mechanisms. Recent Patents CNS Drug Discov 2011;6:119–40.10.2174/157488911795933901Search in Google Scholar PubMed PubMed Central
21. Chiou LC, Hu SJ, Ho YC. Targeting the cannabinoid system for pain relief? Acta Anaesthesiol Taiwan 2013;51:163–70.10.1016/j.aat.2013.10.004Search in Google Scholar PubMed
22. Di Cesare Mannelli L, Zanardelli M, Failli P, Ghelardini C. Oxaliplatin-induced neuropathy: oxidative stress as pathological mechanism. Protective effect of silibinin. J Pain 2012;13:276–84.10.1016/j.jpain.2011.11.009Search in Google Scholar
23. Di Cesare Mannelli L, Bani D, Bencini A, Brandi ML, Calosi L, Cantore M, et al. Therapeutic effects of the superoxide dismutase mimetic compound MnIIMe2DO2A on experimental articular pain in rats. Mediators Inflamm 2013;2013:905360.10.1155/2013/905360Search in Google Scholar
24. Di Cesare Mannelli L, Zanardelli M, Failli P, Ghelardini C. Oxaliplatin-induced oxidative stress in nervous system-derived cellular models: could it correlate with in vivo neuropathy? Free Radic Biol 2013;61C:143–50.10.1016/j.freeradbiomed.2013.03.019Search in Google Scholar
25. Flatters SJ, Bennett GJ. Studies of peripheral sensory nerves in paclitaxel-induced painful peripheral neuropathy: evidence for mitochondrial dysfunction. Pain 2006;122:247–57.10.1016/j.pain.2006.01.037Search in Google Scholar
26. McGrath JC, Lilley E. Implementing guidelines on reporting research using animals (ARRIVE etc.): new requirements for publication in BJP. Br J Pharmacol 2015;172:3189–93.10.1111/bph.12955Search in Google Scholar
27. Guingamp C, Gegout-Pottie P, Philippe L, Terlain B, Netter P, Gillet P. Monoiodoacetate-induced experimental osteoarthritis: a dose-response study of loss of mobility, morphology, and biochemistry. Arthritis Rheum 1997;40:1670–9.10.1002/art.1780400917Search in Google Scholar
28. Pomonis JD, Boulet JM, Gottshall SL, Phillips S, Sellers R, Bunton T, et al. Development and pharmacological characterization of a rat model of osteoarthritis pain. Pain 2005;114:339–46.10.1016/j.pain.2004.11.008Search in Google Scholar
29. Bennett GJ, Xie YK. A peripheral mononeuropathy in rat that produces disorders of pain sensation like those seen in man. Pain 1988;33:87–107.10.1016/0304-3959(88)90209-6Search in Google Scholar
30. Cavaletti G, Tredici G, Petruccioli MG, Dondè E, Tredici P, Marmiroli P, et al. Effects of different schedules of oxaliplatin treatment on the peripheral nervous system of the rat. Eur J Cancer 2001;37:2457–63.10.1016/S0959-8049(01)00300-8Search in Google Scholar
31. Brindisi M, Maramai S, Gemma S, Brogi S, Grillo A, Di Cesare Mannelli L, et al. Development and pharmacological characterization of selective blockers of 2-arachidonoyl glycerol degradation with efficacy in rodent models of multiple sclerosis and pain. J Pain 2013;14:1585–600.10.1021/acs.jmedchem.5b01812Search in Google Scholar PubMed
32. Polomano RC, Mannes AJ, Clark US, Bennett GJ. A painful peripheral neuropathy in the rat produced by the chemotherapeutic drug, paclitaxel. Pain 2001;94:293–304.10.1016/S0304-3959(01)00363-3Search in Google Scholar
33. Leighton GE, Rodriguez RE, Hill RG, Hughes J. K-opioid agonist produce antinociception after i.v. and i.c.v. but not intrathecal administration in the rat. Br J Pharmacol 1998;93:553–60.10.1111/j.1476-5381.1988.tb10310.xSearch in Google Scholar
34. Bove SE, Calcaterra SL, Brooker RM, Huber CM, Guzman RE, Juneau PL, et al. Weight bearing as a measure of disease progression and efficacy of anti-inflammatory compounds in a model of monosodium iodoacetate-induced osteoarthritis. Osteoarthr Cartil 2003;11:821–30.10.1016/S1063-4584(03)00163-8Search in Google Scholar
35. Di Cesare Mannelli L, Pacini A, Bonaccini L, Zanardelli M, Mello T, Ghelardini C. Morphologic features and glial activation in rat oxaliplatin-dependent neuropathic pain. J Pain 2013;14:1585–600.10.1016/j.jpain.2013.08.002Search in Google Scholar PubMed
36. Ivanavicius SP, Ball AD, Heapy CG, Westwood FR, Murray F, Read SJ. Structural pathology in a rodent model of osteoarthritis is associated with neuropathic pain: increased expression of ATF-3 and pharmacological characterization. Pain 2007;128:272–82.10.1016/j.pain.2006.12.022Search in Google Scholar PubMed
37. Pacini A, Di Cesare Mannelli L, Bonaccini L, Ronzoni S, Bartolini A, Ghelardini C. Protective effect of alpha7 nAChR: behavioural and morphological features on neuropathy. Pain 2010;150:542–9.10.1016/j.pain.2010.06.014Search in Google Scholar PubMed
38. Di Cesare Mannelli L, Cinci L, Micheli L, Zanardelli M, Pacini A, McIntosh JM, et al. α-Conotoxin RgIA protects against the development of nerve injury-induced chronic pain and prevents both neuronal and glial derangement. Pain 2014;155:1986–95.10.1016/j.pain.2014.06.023Search in Google Scholar PubMed PubMed Central
39. Andrè T, Boni C, Mounedji-Boudiaf L, Navarro M, Tabernero J, Hickish T, et al. Oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment for colon cancer. Multicenter international study of oxaliplatin/5-fluorouracil/leucovorin in the adjuvant treatment of colon cancer (MOSAIC) investigators. N Engl J Med 2004;350:2343–51.10.1056/NEJMoa032709Search in Google Scholar PubMed
40. Peltier S, Oger J-M, Lagarce F, Couet W, Benoît JP. Enhanced oral paclitaxel bioavailability after administration of paclitaxel-loaded lipid nanocapsules. Pharm Res 2006;23:1243–50.10.1007/s11095-006-0022-2Search in Google Scholar PubMed
41. Gamelin E, Gamelin L, Bossi L, Quasthoff S. Clinical aspects and molecular basis of oxaliplatin neurotoxicity: current management and development of preventive measures. Semin Oncol 2002;29:21–33.10.1053/sonc.2002.35525Search in Google Scholar PubMed
42. Huang ZZ, Li D, Liu CC, Cui Y, Zhu HQ, Zhang WW, et al. CX3CL1-mediated macrophage activation contributed to paclitaxel-induced DRG neuronal apoptosis and painful peripheral neuropathy. Brain Behav Immun 2014;40:155–65.10.1016/j.bbi.2014.03.014Search in Google Scholar PubMed
43. Schlosburg JE, Blankman JL, Long JZ, Nomure DK, Pan B, Kinsey SG, et al. Chronic monoacylglycerol lipase blockade causes functional antagonism of the endocannabinoid system. Nat Neurosci 2010;13:1113–9.10.1038/nn.2616Search in Google Scholar PubMed PubMed Central
44. Bai N, He K, Roller M, Lai CS, Bai L, Pan MH. Flavonolignans and other constituents from Lepidium meyenii with activities in anti-inflammation and human cancer cell lines. J Agric Food Chem 2015;63:2458–63.10.1021/acs.jafc.5b00219Search in Google Scholar PubMed
Supplemental Material:
The online version of this article offers supplementary material (https://doi.org/10.1515/znc-2016-0251).
©2017 Walter de Gruyter GmbH, Berlin/Boston
Articles in the same Issue
- Frontmatter
- Ginkgetin inhibits proliferation of human leukemia cells via the TNF-α signaling pathway
- Effects of a water extract of Lepidium meyenii root in different models of persistent pain in rats
- Interaction of tubulin and protein kinase CK2 in Trypanosoma equiperdum
- Synthesis, biological activity and molecular modeling study of new Schiff bases incorporated with indole moiety
- A dihydrochalcone derivative and further steroidal saponins from Sansevieria trifasciata Prain
- Chemical composition and biological activities of leaf and fruit essential oils from Eucalyptus camaldulensis
- Fungal production of the polysaccharide pullulan from a plant hydrolysate
- Acetogenins and alkaloids during the initial development of Annona muricata L. (Annonaceae)
- Corrigendum
- Corrigendum to: Ginkgetin inhibits proliferation of human leukemia cells via the TNF-α signaling pathway
Articles in the same Issue
- Frontmatter
- Ginkgetin inhibits proliferation of human leukemia cells via the TNF-α signaling pathway
- Effects of a water extract of Lepidium meyenii root in different models of persistent pain in rats
- Interaction of tubulin and protein kinase CK2 in Trypanosoma equiperdum
- Synthesis, biological activity and molecular modeling study of new Schiff bases incorporated with indole moiety
- A dihydrochalcone derivative and further steroidal saponins from Sansevieria trifasciata Prain
- Chemical composition and biological activities of leaf and fruit essential oils from Eucalyptus camaldulensis
- Fungal production of the polysaccharide pullulan from a plant hydrolysate
- Acetogenins and alkaloids during the initial development of Annona muricata L. (Annonaceae)
- Corrigendum
- Corrigendum to: Ginkgetin inhibits proliferation of human leukemia cells via the TNF-α signaling pathway

