Abstract
Objective
The aim of this work was to study the optimal cultivation conditions for β-galactosidase production by Bacillus licheniformis ATCC 12759.
Materials and methods
The screening of β-galactosidase production from B. licheniformis ATCC 12759 was performed by solid state fermentation method on media rich with rice bran (RB). Different factors were tested for the optimization of β-galactosidase production.
Results
Certain fermentation parameters involving incubation time, incubation temperature, inoculum level, moisture content, initial pH, agitation speed, size of fermentation medium and optimum temperature of β-galactosidase activity were studied separately. Maximal amount of β-galactosidase production was obtained when solid-state fermentation (SSF) was carried out using RB, having inoculum level 35%, moisture content of 20%, initial pH 7.5 at 37°C for 48 h.
Conclusion
Results indicated that optimal fermentation conditions play a key role in the maximum production of β-galactosidase from B. licheniformis ATCC 12759. This study shows the potential of the studied enzymes to be promoting candidates for the degradation of lactose and production of important bioproducts.
Özet
Amaç
Bu çalışmada Bacillus licheniformis ATCC 12759’dan β-galaktosidaz üretimi için optimal kültür şartlarının belirlenmesi amaçlanmıştır.
Materyal ve Metod
Bacillus licheniformis ATCC 12759’dan β-galaktosidaz üretiminin taranması pirinç kepegiyle zenginleştirilmiş ortamda katı faz fermantasyon metodu ile gerçekleştirildi. β-galaktosidazın optimizasyonu için çeşitli faktörler test edildi.
Bulgular
İnkübasyon zamanı, inkübasyon sıcaklığı, inokülüm oranı, nem içeriği, başlangıç pH, çalkalanma hızı ve β-galaktosidaz aktivitesinin optimum sıcaklığını içeren belirli fermantasyon parametreleri ayrı ayrı incelendi. 48. saatte, 37ºC’de, başlangıç pH 7.5, nem içeriği 20%, inokülüm oranı 35% pirinç kepeği kullanılarak KSF ile gerçekleştirildiğinde maksimum miktarda β-galaktosidaz üretimi elde edildi.
Sonuç
Sonuçlar, optimal fermantasyon koşullarının B. licheniformis ATCC 12759’dan maksimum β-galaktosidaz üretiminde önemli rol oynadığını gösterdi. Bu çalışma incelenen enzimin laktozun parçalanması ve önemli biyoürünleri için potansiyelini göstermektedir.
Introduction
β-galactosidase [EC3.2.1.23] catalyzes the hydrolytic process of β-1,4-D-galactosidic linkages found in lactose (β-D-galactopyr-anosyl-(1→4)-D-glucopyranose) and releases D-glucose and D-galactose as an end product [1]. As a main milksugar, lactose needs to be sufficiently metabolized to fulfill the energy demand of human beings and makes possible the consumption of milk and other dairy products by lactose intolerant people [2]. In addition, some β-galactosidases can transfer the galactosyl residues of lactose to saccharide acceptors to yield galactooligosaccharides (GOS) [3], [4], [5]. GOS is an important prebiotic which can improve gut health by stimulating the proliferation probiotic intestinal bacteria [5]. Some recent studies have demonstrated that GOS can positively modify the immune function of overweight adults and increase calcium absorption of young girls [6], [7], [8].
The sources of β-galactosidase are extensively distributed in nature, namely in microorganisms, plants and animal organs [9], [10]. β-galactosidases from microbial sources exhibit a great industrial relevance mainly due to their easy handling, greater catalytic activity and high production yield [11]. As a result of commercial interest in β-galactosidase, a large number of microorganisms have been assessed as potential sources of this enzyme.
Industrially important enzymes have traditionally been obtained from submerged fermentation (SmF) because of the ease of handling and greater control of environmental factors such as temperature and pH [12]. But, nowadays, the demand for β-galactosidase production, as well as the development of an effective and inexpensive β-galactosidase production process has increased significantly. Industrial production of enzymes can be made economical by utilizing low-cost substrates such as agricultural byproducts in the production medium [13]. Compared with SmF, solid-state fermentation (SSF) has many advantages, such as superior productivity, greater simplicity, lower capital investment, less energy requirement and waste water output, better product recovery, etc., and is reported to be the most appropriate process for developing countries [14]. On preliminary cost analysis, a net savings of about 60 and 50% on fermentation medium cost and the expenditure on down-stream processing, respectively, as compared to the presently employed SmF technique was evident [15], [16]. It can be of special interest in those processes where the crude fermented products may be used directly as the enzyme sources for biocatalysis and biotransformation [17].
A reduction in β-galactosidase production cost is very important. The use of abundantly available lignocellulosic crop residues, such as rice husk, rice bran (RB), and wheat bran, for culture of microbes producing β-galactosidase offers an approach to reach this goal. In general, β-galactosidase is intracellular enzyme and to study intracellular enzymes is difficult. But β-galactosidase production of such microorganisms is very easy and economic because it is extracellular enzyme. This paper describes the screening of various agro-industrial substrates and the development of a suitable low cost fermentation medium for the optimizing following parameters with emphasis on incubation time, extraction medium, temperature, initial pH, moisture level, inoculum size, supplementation of carbon, nitrogen sources and metal salts by using RB as solid substrate.
Materials and methods
Bacterial strains and culture conditions
β-Galactosidase producing Bacillus licheniformis ATCC 12759 which was procured from MicroBioLogics, Inc. was used as biological material. Bacillus licheniformis ATCC 12759 was grown on nutrient agar at 37°C for 24 h for inoculum preparation. A loopful of the growth was transferred to Laura broth (LB) liquid medium [1% yeast extract, 0.5% peptone, 0.5% NaCl, (w/v), pH 7.0].
Enzyme production in SSF
Solid-state fermentation
SSF was carried out by taking 3 g of dry substrate in a 100 mL Erlenmeyer flask to which distilled water was added to adjust the required moisture level. The contents of the flasks were mixed and autoclaved at 121°C for 15 min. Flasks with inoculated were shaken at 150 rpm at 37°C for 144 h. The contents of the flasks were harvested and assayed every 24 h.
Enzyme extraction
The enzyme from the fermented bacterial bran was extracted twice with tap water. The slurry was squeezed through damp cheesecloth. Extracts were pooled and centrifuged at 4°C for 15 min at 10,000 rpm to separate small particles of different substrates, cells and spores. The brown, clear supernatant was used in enzyme assays.
Enzyme assay
The reaction mixture containing 500 μL 6 mM 2-nitrophenly β-D-galactoprynoside in 0.1 M sodium phosphate buffer (pH 6.8) and 200 μL of enzyme solution was incubated for 30 min at 37°C. The reaction was ended by adding 0.5 mL of 1 M Na2CO3 and the concentration of o-nitrophenol (ONP) released from ONPG was determined by measuring the absorbance at 420 nm, using a standard calibration curve. The enzyme activity was expressed as specific activity (U mg−1 soluble protein) and one unit of β-galactosidase activity (U) was defined as the amount of enzyme that liberates 1 nmol ONP per minute [18]. All experiments were conducted in triplicate and the mean at three with standard deviation (SD) was represented.
Effect of process parameters on β-galactosidase production in SSF
The optimization of medium components and fermentation process is of primary importance in any fermentation process. Combinations of the best substrates were employed for further optimization of process parameters, namely initial moisture content (20, 30, 40, 50 and 60%), incubation time (24, 48, 72, 96, 120 and 144 h), incubation temperature (30, 37, 40, 45 and 50°C), initial pH of the medium (pH: 4.0–10.0), inoculum size (0.5, 1, 1.5, 2, 2.5, 3, 3.5, 4, 6 and 8×106 CFU/mL), agitation speed (60, 100, 120, 150, 180 and 200 rpm), scale up (100, 250, 500 and 1000 mL shake flasks), the effects of the different proportions in mixed substrates, while nutrient supplementation such as inorganic nitrogen sources 1% (by mass) (ammonium nitrate, sodium nitrate, ammonium chloride and ammonium sulfate), organic nitrogen sources (peptone, tryptone, yeast extract, beef extract, urea, and casein), and added metal salts 0.1% (by mass) FeSO4·7H2O, MgSO4·7H2O, CuSO4·5H2O, ZnSO4·7H2O and CaCl2 were optimized. To study the efficacy of various inducers, the medium was supplemented independently with 1% mannose, xylose, lactose, sucrose, fructose, galactose, glucose, and arabinose. Distilled water, tap water, 50 mM NaCl, 0.1 M phosphate buffer (pH=7.0), 1% solution of various detergents like Tween 40, CHAPS (3-[(3-cholamido-propyl)-dimethylammonio]-propane-sulfonate), sodium dodesil sulfate (SDS) and Triton X-100 were used independently to find the best extraction medium for the enzyme. Data were expressed as the average of three replicates.
Results
The time courses of β-galactosidase production by B. licheniformis ATCC 12759 are shown in Figure 1. The initial β-galactosidase biosynthesis by B. licheniformis ATCC 12759 was reached the maximum (3244.7 U/mg) at 120 h.
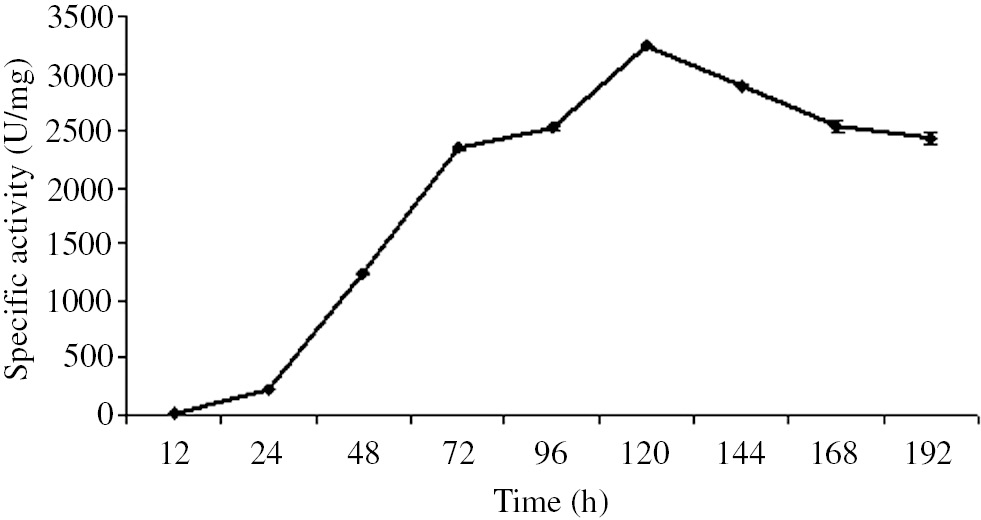
Effect of incubation time on β-galactosidase production by B. licheniformis ATCC 12759 in solid-state fermentation.
Investigation of the effect of temperature on enzyme production during fermentation showed that the optimum temperature for maximum yield of β-galactosidase (3396.8 U/mg) was 37°C (Figure 2).

Effect of temperature on β-galactosidase production by B. licheniformis ATCC 12759 in solid-state fermentation.
From the results (Table 1), it is clear that among all the solvents, tap water (3239.0 U/mg) gave the best extraction of β-galactosidase from the fermented solids.
Effect of extraction medium on β-galactosidase production by B. licheniformis ATCC 12759 in solid-state fermentation.
| Extraction medium | Specific activity (U/mg) |
|---|---|
| NaCl | 2563.6±35.1 |
| CHAPS | 2539.9±24.1 |
| Triton X-100 | 1773.5±16.9 |
| SDS | 2505.3±9.4 |
| Distilled water | 2675.3±110 |
| Phosphate buffer | 2746.7±18.4 |
| Tween 40 | 1742.7±16.8 |
| Tap water | 3229±12.7 |
(p<0.05).
The highest enzyme production (3530.4 U/mg) was obtained at an inoculum level of 35% (v/w) (Figure 3). The results from this study indicate that 35% inoculum size was optimal, balancing enzyme and biomass production.
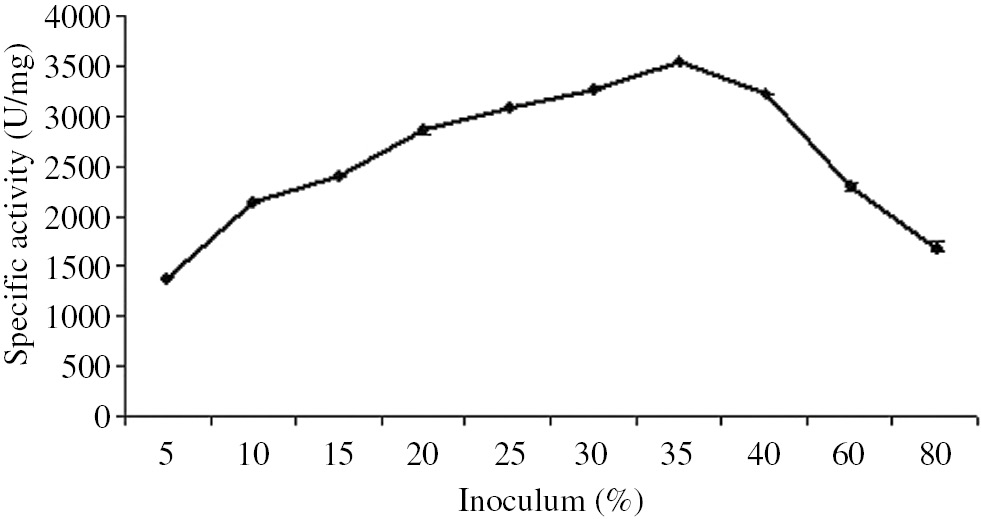
Effect of inoculum on β-galactosidase production by B. licheniformis ATCC 12759 in solid-state fermentation.
In the present investigation, five moisture levels ranging from 20 to 60% were established to study their effect on β-galactosidase production and the results obtained are shown in Figure 4. The highest production of β-galactosidase (3238.4 U/mg) was obtained when the initial moisture content was 20%.

Effect of moisture level on β-galactosidase production by B. licheniformis ATCC 12759 in solid-state fermentation.

Effect of initial pH on β-galactosidase production by B. licheniformis ATCC 12759 in solid-state fermentation.
Results showed that maximum enzyme production was observed at pH 7.0 (Figure 5). Therefore, in the subsequent experiments, the initial pH of the fermentation medium was adjusted to 7.0 with tap water.
β-galactosidase production was investigated at six different speeds (60–200 rpm). The optimal agitation speed for maximum β-galactosidase production (3473.3 U/mg) was obtained at 150 rpm (Table 2). The higher agitation levels (180–200 rpm) reduced β-galactosidase production from B. licheniformis ATCC 12759 due to sheer stress and heterogeneous mixing effects.
Effect of agitation speed on β-galactosidase production by B. licheniformis ATCC 12759 in solid-state fermentation.
| Agitation speed (rpm) | Specific activity (U/mg) |
|---|---|
| 60 | 3175.3±17 |
| 100 | 3244.6±12.1 |
| 120 | 3266.6±8 |
| 150 | 3473.3±12.7 |
| 180 | 3065.2±9 |
| 200 | 2646.4±12.1 |
(p<0.05).
To improve for a large-scale SSF, β-galactosidase production was investigated in four different fermentation medium sizes (100, 250, 500 and 1000 mL erlenmayer flasks). When solid state fermentation with B. licheniformis ATCC 12759 was carried out in Erlenmeyer flasks of various sizes with corresponding increase in with different quantities of RB moistened with appropriate amounts of tap water, the time course of enzyme production was similar to that in 100 mL Erlenmeyer flasks containing 2 g of RB (control), in most of the cases. The results were quite encouraging for the large scale production of the enzyme though the yields exhibited slight decline with the increase in substrate quantity which is probably due to lesser degree of aeration (Table 3). Future research will be focused on optimizing the nutrient conditions to obtain higher biomass, which is desirable for the industrial development of β-galactosidase product under low production cost [19].
Effect of medium volume on β-galactosidase production by B. licheniformis ATCC 12759 in solid-state fermentation.
| Medium size (mL) | Specific activity (U/mg) |
|---|---|
| 100 | 3138.7±13.7 |
| 250 | 3083.7±8.7 |
| 500 | 2951.5±20 |
| 1000 | 3032.7±3.7 |
(p<0.05).
To examine the effect of carbon source on the production of β-galactosidase B. licheniformis ATCC 12759, which produced a high amount of β-galactosidase with the highest specific activity (Table 4). The production of β-galactosidase by B. licheniformis ATCC 12759 was suppressed when the bacterium was grown on readily metabolizable sugars, since a low basal activity of β-galactosidase was detected in the culture medium.
Carbon sources on the production of β-galactosidase by B. licheniformis ATCC 12759.
| Carbon source (1%) | Specific activity (U/mg) |
|---|---|
| Control | 3674.3±78.3 |
| Sucrose | 3083.3±61.5 |
| Glucose | 2903.2±75.9 |
| Galactose | 3524.7±192.3 |
| Fructose | 2880.3±80.3 |
| Lactose | 2928.8±71.4 |
| Mannose | 2968.4±28.5 |
| Xylose | 2690.6±26.9 |
| Arabinose | 3328.0±33.2 |
(p<0.05).
The production of β-galactosidase by B. licheniformis ATCC 12759 was suppressed when the bacterium was grown on different organic and inorganic nitrogen sources (Table 5). Apart from a good carbon source, RB also could serve as a nitrogen source, thus an increase in the complex nitrogen source adversely influenced the production of β-galactosidase.
Nitrogen sources on the production of β-galactosidase by B. licheniformis ATCC 12759.
| Nitrogen source (1%) | Specific activity (U/mg) |
|---|---|
| Control | 3674.3±63.0 |
| Sodium nitrate | 2616.3±35.9 |
| Ammonium sulfate | 3200.4±93.5 |
| Ammonium nitrate | 2280.0±73.5 |
| Ammonium chloride | 1995.6±63.6 |
| Beef extract | 2110.4±47.8 |
| Tryptone | 3209.0±104.0 |
| Peptone | 1944.5±90.5 |
| Yeast extract | 1974.4±121.5 |
| Urea | 2735.8±54.2 |
| Casein | 1620.5±61.5 |
(p<0.05).
Addition of metal salts source such as FeSO4, MgSO4, CuSO4, ZnSO4 and CaCl2 to the medium were investigated. Comparison with the control (3475.6 U/mg), the production of β-galactosidase by B. licheniformis ATCC 12759 was suppressed when the bacterium was grown on metal salt sources (Table 6). Among the metal salt sources ZnSO4 (52.1 U/mg) was greatly inhibited production of β-galactosidase by B. licheniformis ATCC 12759. On the other hand the salt requirement for production of this particular enzyme was apparently provided by the nature of RB.
Effect of metal salt on β-galactosidase production by B. licheniformis ATCC 12759 in solid-state fermentation.
| Metal salt (0.01%) | Specific activity (U/mg) |
|---|---|
| Control | 3447.5±28.5 |
| MgSO4 | 2869.8±13.1 |
| CuSO4 | 2044.2±28.1 |
| CaCl2 | 2635.7±17.4 |
| ZnSO4 | 229.1±13.3 |
| FeSO4 | 2623.3±9 |
(p<0.05).
Discussion
Due to the potential usefulness of the β-galactosidase in the industrial applications, the development of methods for cheaper production of enzyme is very important. One alternative low cost production method is the use of SSF. In this study SSF has been found to be a cheap way of producing high levels of β-galactosidase by B. licheniformis ATCC 12759. The nature of solid substrate is the most important factor in SSF. This not only supplies the nutrients to the culture but also serves as an anchorage for the microbial cells. An ideal solid substrate provides all necessary nutrients to the microorganism for optimum function [20]. Developing a β-galactosidase production process based upon RB as the solid substrate is very attractive, since it is a readily available source of carbon. It was previously reported that RB was found to be the best substrate for cellulase and β-glucosidase production by T. reesei and P. citrinum YS40-5 [21], [22]. This was in contrast to earlier report, which described wheat bran as a potential substrate for β-galactosidase production [23]. This is the first report of utilization of RB as solid substrate for β-galactosidase production by B. licheniformis ATCC 12759 by SSF. As a result was significant RB, further studies were carried out using this substrate.
The incubation time is governed by characteristics of the culture and also based on growth rate and enzyme production. The enzyme yield showed a gradual decrease on further extension of fermentation period which could be due to the depletion of nutrients for enzyme synthesis and proteolytic degradation of already synthesized enzymes [24].
The effect of temperature on β-galactosidase production is related to the growth of the organism. β-galactosidase yield increased with increase in incubation temperature from 30 to 37°C and higher temperature levels were detrimental to the growth and β-galactosidase production by B. licheniformis ATCC 12759, which may be attributed to the mesophilic nature of the microbe [24]. Maximum production at lower temperatures may be advantageous as it can reduce the rate of evaporation during incubation [20].
In SSF, optimum extraction of the product of interest from the fermented mass with a suitable solvent is a necessity. Parameters governing this process must therefore be established to maximize recovery [25]. From the results, it is clear that among all the solvents, tap water gave the best extraction of β-galactosidase from the fermented solids. This might be due to dissolution of the all fermented media by tap water which then becomes phosphate buffer and hence able to extract enzyme protein from fermented biomass. In contrast, distilled water is extractant (available, save and low cost) used for extraction of levansucrase from solid sawdust fermentation [26].
The inoculum level was also an important factor for the production of β-galactosidase. High inoculum levels are inhibitory in nature. A higher inoculum size may increase moisture content and lead to a decrease in growth and enzyme production; this may be due to the limiting nutrients at higher inoculum size and a lower inoculum size may require a longer time for fermentation to form the desired product [27], [28], [29].
The critical importance of moisture level in SSF media and its influence on the biosynthesis of enzymes has been attributed to the interference of moisture in the physical properties of solid particles [20]. The fact that the bacteria can grow and produce maximum β-galactosidase at lower moisture content of fermentable substrate offers significant advantages in reducing risk of contamination. Either low or high initial moisture significantly decreased the enzyme production. Lower moisture levels lead to reduced solubility of the nutrients, a lower degree of substrate swelling and higher water tension. Higher initial moisture in SSF decreases porosity, changes the particle structure and promotes development of stickiness due to agglomeration of the substrate. This reduces mass transfer process and gas exchange subsequently restricting the supply of oxygen for the growth of microorganism and leading to suboptimal product formation [24], [30].
Among the physicochemical parameters, the pH of the growth medium plays an important role by inducing morphological changes in the organism and in enzyme secretion [27]. Similarly, Dagbagli and Goksungur reported that pH 7.35 optimum for β-galactosidase production by Kluyveromyces lactis NRRL Y-8279 [28]. Results show that enzyme production was generally stable from pH 7.0, which indicates excellent buffering property of the agroresidues used for SSF [27].
To examine the effect of carbon source on the production of β-galactosidase B. licheniformis ATCC 12759, which produced a high amount of β-galactosidase with the highest specific activity (Table 1). The production of β-galactosidase by B. licheniformis ATCC 12759 was suppressed when the bacterium was grown on readily metabolizable sugars, since a low basal activity of β-galactosidase was detected in the culture medium. Several investigators have described the carbon source regulation of β-galactosidase biosynthesis in various microorganisms [31], [32]. All indicated that the role of carbon source in the biosynthesis of β-galactosidase may vary and depend on the microorganisms tested. Similarly, Konsoula and Kyriakides [33] reported the production of β-galactosidase by B. subtilis was greatly suppressed when the bacterium was grown on readily metabolizable sugars, since a very low basal activity of enzyme was detected in the culture medium in the presence of glucose, maltose, maltotriose or lactose. Kim and Rajagopal [34] described that addition of glucose or lactose to the growth medium inhibited the synthesis of β-galactosidase by L. crispatus. Easily metabolizable carbohydrates may result in the better growth of the bacteria along with reduction in the enzyme formation [35]. Although RB contains about 20% oil, 15% protein, and approximately 50% carbohydrate, of which starch is the main component, it has never been proposed as a source of sugars [36]. Since RB was used, carbon requirement could be met from it. This is important in terms of the cost of production of β-galactosidase.
Nitrogen source is an important factor that effects production of enzyme. Nizamuddin et al. [23] reported different organic and inorganic nitrogen sources improved the β-galactosidase production on RB medium produced by A. oryzae.
Agitation speed is a very important factor in the fermentation process since it will increase the amount of dissolved oxygen in the cultivation medium [37]. Agitation affects both air bubble dispersion and mixing of nutrients during fermentation process [38]. Dagbagli and Goksungur [28] reported that maximum β-galactosidase production from K. lactis NRRL Y-8279 was achieved when the optimal agitation speed of 179.2 rpm was used. Nadeem et al. [38] also found that the enzyme production was strongly affected by the agitation.
Laboratory-scale experiments may provide a basis for scale-up purposes of the process on β-galactosidase production in SSF [19]. Different types of production vessels have been used for carrying out solid state fermentation [39]; but most of the laboratory studies on the production of enzymes using SSF technique have employed Erlenmeyer flasks [19], [20], [23], [40]. The results were quite encouraging for the large scale production of the enzyme though the yields exhibited slight decline with the increase in substrate quantity which is probably due to lesser degree of aeration [20]. Future research will be focused on optimizing the nutrient conditions to obtain higher biomass, which is desirable for the industrial development of β-galactosidase product under low production cost [19].
Conclusion
In this study, we report the optimal cultivation conditions for β-galactosidase production by B. licheniformis ATCC 12759. Maximal amount of β-galactosidase production was obtained when SSF was carried out using RB, having inoculum level 35%, moisture content of 20%, initial pH 7.5 at 37°C for 48 h. Commercial β-galactosidase production is usually produced by SmF; however, SSF appear promising due to the natural potential and advantages they offer. Based on the present study, it appears that RB, which is inexpensive and readily available agricultural substance, could replace the commercial and more expensive substances in the development of a suitable economic fermentation medium for obtaining high yields β-galactosidase. However, the present study was entirely a laboratory-scale study, and it has to be further improved for a large-scale SSF.
References
1. Kamran A, Bibi Z, Amana A, Qader SA. Lactose hydrolysis approach: isolation and production of β-galactosidase from newly isolated Bacillus strain B-2. Biocatal Agric Biotechnol 2016;5:99–103.10.1016/j.bcab.2015.12.010Search in Google Scholar
2. Haider T, Husain Q. Immobilizationof β galactosidase from Aspergillus oryzae via immunoaffinity support. Biochem Eng J 2009;43:307–14.10.1016/j.bej.2008.10.012Search in Google Scholar
3. Rodriguez-Colinas B, Poveda A, Jimenez-Barbero J, Ballesteros AO, Plou FJ. Galacto-oligosaccharide synthesis from lactose solution or skim milk using the β-galactosidase from Bacillus circulans. J Agric Food Chem 2012;60:6391–8.10.1021/jf301156vSearch in Google Scholar PubMed
4. Satar R, Ismail SA, Rehan M, Ansari SA. Elucidating the binding efficacy of β-galactosidase on graphene by docking approach and its potential application in galacto-oligosaccharide production. Bioprocess Biosyst Eng 2016;39:807–14.10.1007/s00449-016-1560-6Search in Google Scholar PubMed
5. Yin H, Bultema JB, Dijkhuizen L, van Leeuwen SS. Reaction kinetics and galactooligosaccharide product profiles of the β-galactosidases from Bacillus circulans, Kluyveromyces lactis and Aspergillus oryzae. Food Chem 2017;225:230–8.10.1016/j.foodchem.2017.01.030Search in Google Scholar PubMed
6. Vulevic J, Juric A, Tzortzis G, Gibson GR. Galactooligosaccharides reduces markers of metabolic syndrome and modulates the fecal microbiota and immune function of overweight adults. J Nutr 2013;143:324–31.10.3945/jn.112.166132Search in Google Scholar PubMed
7. Whisner CM, Martin BR, Schoterman MH, Nakatsu CH, McCabe LD, McCabe GP, et al. Galacto-oligosaccharides increase calcium absorption and gut bifidobacteria in young girls: a double-blind cross-over trial. Br J Nutr 2013;110:1292–303.10.1017/S000711451300055XSearch in Google Scholar PubMed
8. Liua Y, Chen Z, Jiang Z, Yan Q, Yanga S. Biochemical characterization of a novel β-galactosidase from Paenibacillus barengoltzii suitable for lactose hydrolysis and galactooligosaccharides synthesis. Int J Biol Macromol 2017;104:1055–63.10.1016/j.ijbiomac.2017.06.073Search in Google Scholar PubMed
9. Husain Q. β-galactosidases and their potential applications: a review. Crit Rev Biotechnol 2010;30:41–62.10.3109/07388550903330497Search in Google Scholar PubMed
10. Cardoso BB, Silvério SC, Abrunhosa L, Teixeira JA, Rodrigues LR. β-galactosidase from Aspergillus lacticoffeatus: a promising biocatalyst for the synthesis of novel prebiotics. Int J Food Microbiol 2017;257:67–74.10.1016/j.ijfoodmicro.2017.06.013Search in Google Scholar PubMed
11. Panesar PS, Panesar R, Singh RS, Kennedy JF, Kumar H. Microbial production, immobilization and applications of β-D-galactosidase. J Chem Technol Biotechnol 2006;81: 530–43.10.1002/jctb.1453Search in Google Scholar
12. Kar S, Datta TK, Ray RC. Optimization of thermostable α-amylase production by Streptomyces erumpens MTCC 7317 in solid-state fermentation using cassava fibrous residue. Braz Arc Bıo Technol 2010;53:301–9.10.1590/S1516-89132010000200008Search in Google Scholar
13. Pandey A, Soccol CR, Nigam P, Soccol VT. Biotechnological potential of agro-industrial residues. I: sugarcane bagasse. Bioresour Technol 2000;74:69–80.10.1016/S0960-8524(99)00142-XSearch in Google Scholar
14. Xu H, Sun L, Zhao D, Zhang B, Shi Y, Wu Y. Production of α-amylase by Aspergillus oryzae As 3951 in solid state fermentation using spent brewing grains as substrate. J Sci Food Agric 2008;88:529–35.10.1002/jsfa.3118Search in Google Scholar
15. Kumar PK, Lonsane BK. Gibberellic acid by solid state fermentation: consistent and improved yields. Biotechnol Bioeng 2004;30:267–71.10.1002/bit.260300217Search in Google Scholar
16. Tanyildizi MS, Özer D, Elibol M. Production of bacterial α-amylase by B. amyloliquefaciens under solid substrate fermentation. Biochem Eng J 2007;37:294–7.10.1016/j.bej.2007.05.009Search in Google Scholar
17. Pandey A, Selvakumar P, Soccol CR, Singh N, Poonam N. Solid state fermentation for the production of industrial enzymes. Curr Scı 1999;77:149–62.Search in Google Scholar
18. Vasiljevic T, Jelen P. Production of β-galactosidase for lactose hydrolysis in milk and dairy products using thermophilic lactic acid bacteria. Inn Food Sci Emerg Technol 2001;2:75–85.10.1016/S1466-8564(01)00027-3Search in Google Scholar
19. Wang CL, Li DF, Lu WQ, Wang YH, Lai CH. Influence of cultivating conditions on the α-galactosidase biosynthesis from a novel strain of Penicillium sp. in solid-state fermentation. Lett Appl Microbiol 2004;39:369–75.10.1111/j.1472-765X.2004.01594.xSearch in Google Scholar
20. Singh H, Soni SK. Production of starch-gel digesting amyloglucosidase by Aspergillus oryzae HS-3 in solid state fermentation. Process Biochem 2001;37:453–9.10.1016/S0032-9592(01)00238-2Search in Google Scholar
21. Latifian M, Esfahani ZH, Barzegar M. Evaluation of culture conditions for cellulase production by two Trichoderma reesei mutants under solid-state fermentation conditions. Bioresour Technol 2007;98:3634–7.10.1016/j.biortech.2006.11.019Search in Google Scholar PubMed
22. Ng IS, Li CW, Chan SP, Chir JL, Chen PT, Tong CG, et al. High-level production of a thermoacidophilic β-glucosidase from Penicillium citrinum YS40-5 by solid-state fermentation with rice bran. Bioresour Technol 2010;101:1310–7.10.1016/j.biortech.2009.08.049Search in Google Scholar
23. Nizamuddin S, Sridevi A, Narasimha G. Production of β-galactosidase by Aspergillus oryzae in solid-state fermentation. Afr J Biotechnol 2008;7:1096–100.Search in Google Scholar
24. Anisha GS, Rojan PJ, Nicemol J, Niladevi KN, Prema P. Production and characterization of partially purified thermostable a-galactosidases from Streptomyces griseoloalbus for food industrial applications. Food Chem 2008;111:631–5.10.1016/j.foodchem.2008.04.029Search in Google Scholar
25. Sangeeta N, Rintu B. Optimization of extraction and purification of glucoamylase produced by Aspergillus awamori in solid-state fermentation. Biotechnol and Bioprocess Eng 2009;14:60–6.10.1007/s12257-008-0107-3Search in Google Scholar
26. Ahmed SA. Optimization of production and extraction parameters of Bacillus megaterium levansucrase using solid-state fermentation. J Appl Sci Res 2008;4:1199–204.Search in Google Scholar
27. Gangadharan D, Sivaramakrishnan S, Nampoothiri KM, Pandey A. Solid culturing of Bacillus amyloliquefaciens for alpha amylase production. Food Technol Biotechnol 2006;44:269–74.Search in Google Scholar
28. Dagbagli S, Goksungur Y. Optimization of β-galactosidase production using Kluyveromyces lactis NRRL Y-8279 by response surface methodology. Electronic J Biotechnol 2008;11.10.2225/vol11-issue4-fulltext-12Search in Google Scholar
29. Baysal Z, Uyar F, Aytekin Ç. Solid state fermentation for production of α-amylase by a thermotolerant Bacillus subtilis from hot-springwater. Process Biochem 2003;38:1665–8.10.1016/S0032-9592(02)00150-4Search in Google Scholar
30. Kashyap P, Sabu A, Pandey A, Szakas G, Soccol CR. Extracellular L-glutaminaseproduction by Zygosaccharomyces rouxii under solid state fermentation. Process Biochem 2002;38:307–12.10.1016/S0032-9592(02)00060-2Search in Google Scholar
31. Pandey A, Soccol CR, Rodriguez LJ, Nigam P. Solid-state fermentation in biotechnology. Fundamentals and applications. New Delhi: Asiatech Publishers Inc., 2001: 98.Search in Google Scholar
32. Hsu CA, Yu RC, Chou CC. Production of h-galactosidase by Bifidobacteria as influenced by various culture conditions. Int J Food Microbiol 2005;104:197–206.10.1016/j.ijfoodmicro.2005.02.010Search in Google Scholar PubMed
33. Konsoula Z, Kyriakides ML. Co-production of α-amylase and β-galactosidase by Bacillus subtilis in complex organic substrates. Bioresource Technol 2007;98:150–7.10.1016/j.biortech.2005.11.001Search in Google Scholar PubMed
34. Kim JW, Rajagopal SN. Isolation and characterization of b-galactosidase from Lactobacillus crispatus. Folia Microbiol 2000;45:29–34.10.1007/BF02817446Search in Google Scholar PubMed
35. Batra N, Singh J. Production and characterization of a thermostable β-galactosidase from Bacillus coagulans RCS3. Biotechnol Appl Biochem 2002;236:1–6.10.1042/BA20010091Search in Google Scholar PubMed
36. Hernandez N, Rodriguez-Alegría ME, Gonzalez F, Lopez-Munguia A. Enzymatic treatment of rice bran to improve processing. J Am Oil Chem Soc 2000;77:177–80.10.1007/s11746-000-0028-2Search in Google Scholar
37. Purwanto LA, Ibrahim D. Sudrajat effect of agitation speed on morphological changes in Aspergillus niger hyphae during production of tannase. World J Chem 2009;4:34–8.Search in Google Scholar
38. Nadeem M, Qazi JI, Baig S. Effect of aeration and agitation rates on alkaline protease production by Bacillus licheniformis UV-9 Mutant. Turkish J Biochem 2009;34:89–96.Search in Google Scholar
39. Mitchell DA, Krieger N, Stuart DM, Pandey A. New developments in solid-state fermentation II. Rational approaches to the design, operation and scale-up of bioreactors. Process Biochem 2000;35:1211–25.10.1016/S0032-9592(00)00157-6Search in Google Scholar
40. Shankar SK, Mulimani VH. α-Galactosidase production by Aspergillus oryzae in solid-state fermentation. Bioresource Technol 2007;98:958–61.10.1016/j.biortech.2006.03.013Search in Google Scholar PubMed
©2018 Walter de Gruyter GmbH, Berlin/Boston
Articles in the same Issue
- Frontmatter
- Research Articles
- Comparative study of stirred and fluidized tank reactor for hydroxyl-kojic acid derivatives synthesis and their biological activities
- Synthesis and antimicrobial activities of some novel thiazole compounds
- Water based PHEMA hydrogels for controlled drug delivery
- Cultural conditions optimization for production of β-galactosidase from Bacillus licheniformis ATCC 12759 under solid-state fermentation
- A novel application of oriental beetle juvenile Anomala corpulenta to reflect Pb contamination in soil
- Purification and characterization of Rhizoctonia solani AG-4 strain ZB-34 α-amylase produced by solid-state fermentation using corn bran
- Grain amino acid composition of barley (Hordeum vulgare L.) cultivars subjected to selenium doses
- Microsatellite-based characterization of cotton genotypes for verticillium wilt and fiber quality traits
- Novel pectinase from Piriformospora indica, optimization of growth parameters and enzyme production in submerged culture condition
- Application of Bdellovibrio bacteriovorus for reducing fouling of membranes used for wastewater treatment
- Large unstained cells are correlated with inflammatory biomarkers in patients with invasive aspergillosis
- Detection of industrially potential enzymes of moderately halophilic bacteria on salted goat skins
- Strain improvement of newly isolated Lactobacillus acidophilus MS1 for enhanced bacteriocin production
- The effects of hydraulic calcium silicate containing endodontic materials on oxidative stress in erythrocytes and liver
Articles in the same Issue
- Frontmatter
- Research Articles
- Comparative study of stirred and fluidized tank reactor for hydroxyl-kojic acid derivatives synthesis and their biological activities
- Synthesis and antimicrobial activities of some novel thiazole compounds
- Water based PHEMA hydrogels for controlled drug delivery
- Cultural conditions optimization for production of β-galactosidase from Bacillus licheniformis ATCC 12759 under solid-state fermentation
- A novel application of oriental beetle juvenile Anomala corpulenta to reflect Pb contamination in soil
- Purification and characterization of Rhizoctonia solani AG-4 strain ZB-34 α-amylase produced by solid-state fermentation using corn bran
- Grain amino acid composition of barley (Hordeum vulgare L.) cultivars subjected to selenium doses
- Microsatellite-based characterization of cotton genotypes for verticillium wilt and fiber quality traits
- Novel pectinase from Piriformospora indica, optimization of growth parameters and enzyme production in submerged culture condition
- Application of Bdellovibrio bacteriovorus for reducing fouling of membranes used for wastewater treatment
- Large unstained cells are correlated with inflammatory biomarkers in patients with invasive aspergillosis
- Detection of industrially potential enzymes of moderately halophilic bacteria on salted goat skins
- Strain improvement of newly isolated Lactobacillus acidophilus MS1 for enhanced bacteriocin production
- The effects of hydraulic calcium silicate containing endodontic materials on oxidative stress in erythrocytes and liver

