Abstract
Background and aims
Several pain conditions have been shown to reduce the discriminative abilities of external stimuli. The aim of this study was to investigate how cutaneous sensitization affects the tempo-spatial discrimination for both painful laser stimulation and mechanical stimulation.
Methods
Fifteen healthy subjects were presented with two different stimulation paradigms, a continuous line stimulation and a 2-point stimulation. Line stimulations were delivered in two different directions in lengths of 25, 50, 75, and 100 mm. Two-point distances from 0 to 100 mm were tested. The subjects reported the perceived intensity, and either direction (line stimulations) or number of perceived points (2-point stimulations). All stimuli were tested both before and after topical capsaicin (8% concentration) sensitization (30 min).
Results
All mechanical line stimulations were reported correctly before capsaicin and 3 stimulations (out of 240) were reported incorrectly after capsaicin. For the laser line stimulation, the directional discrimination threshold (DDT) was 69.5 mm before capsaicin and 76.3 mm after capsaicin. The 2-point discrimination threshold for laser stimulation was 70.3 mm before capsaicin and 68.0 mm after, for the mechanical stimuli it was 31.5 mm before capsaicin and 31.0 mm after capsaicin. The perceived intensities were increased for the laser line stimulations after capsaicin (linear mixed model (LMM), p < 0.001) and increased with stimulation length (LMM, p < 0.001). For mechanical stimuli, NRS was increased following capsaicin (LMM, p < 0.001). The intensities for both mechanical and laser 2-point stimuli increased after capsaicin and increased with distance between points (LMM, p < 0.01).
Conclusions
The findings show how cutaneous sensitization appears to affect directional discrimination to a larger extent than the 2-point discrimination.
Implications
This study is the first to investigate how directional discrimination is altered during sensitization. If such measures can be optimized they may provide a new method to probe the neural mechanisms in pain patients.
1 Introduction
The exteroceptive system is responsible for sensing external stimuli in relation to time and space. Until now, its function for cutaneous stimuli has been studied primarily for the tactile modality [1], [2]. Previously it has been shown that the function and characteristics of the noxious system in relation to graphestesia (the ability to recognize numbers drawn on the skin) [3], and directional discrimination (the ability to recognize the direction of a continuous moving stimulus) [4]. Moreover, the simultaneous 2-point discrimination appears to be worse for noxious stimuli [3], [4]. Overall, the exteroceptive system appears to have lower accuracy for noxious stimuli compared to innocuous stimuli. Such measures are not just reflecting the function of the first order sensory neuron, but relies as well on the function of high-order neurons along the neuroaxis [1]. Previous animal studies have demonstrated the existence of direction-sensitive neurons in the primary sensory cortex responding primarily to directional movement [5], [6], and are more or less silent for single point stimuli [5], [6]. These studies applied various types of tactile stimuli, but no noxious stimuli were tested.
It may be speculated that the function of direction-sensitive neurons in the nociceptive system may also be affected by various clinical conditions such as chronic pain. The majority of research regarding the human pain system focuses on aspects related to pain intensity, e.g. evidence showing that the temporal summation mechanism is facilitated in chronic pain patients [7], [8], [9]. However, there is a continued need for new methods for probing the nociceptive system that are sensitive to underlying mechanisms [10]. In this respect, it is very interesting that the tactile acuity often is lower in chronic pain patients compared to healthy controls [11] for conditions such as arthritis, chronic low back pain and complex regional pain syndrome [11], [12]. Some evidence suggest that the tactile acuity of CRPS patients was positively correlated with the pain intensity [11]. In addition, several chronic pain conditions exhibit tactile sensory abnormalities [13]. Overall this indicates that outcome measures relating to spatial acuity, including the directional discrimination threshold (DDT), may have diagnostic relevance in pain patients.
To further investigate how sensitization affects the discriminative abilities, the current study used capsaicin to sensitize the skin to explore the directional and 2-point discrimination capabilities in an experimental model of hyperalgesia. As a pain model, topical capsaicin can induce both peripheral and central sensitization [14], and has been shown to reduce the 2-point discrimination [15]. In this study we hypothesized that capsaicin induced cutaneous sensitization would decrease the ability to discriminate direction of movement. The effect of sensitization was investigated for both laser stimuli, preferentially activating nociceptive receptors and tactile stimuli preferentially activating non-nociceptive receptors.
2 Methods
2.1 Subjects
Fifteen healthy subjects participated in this study (4 females and 11 males, mean age 28.8 years – range 22–48 years). The subjects were placed in a bed with inclined back rest during the experiment. The right forearm was kept horizontal and was stimulated with both non-painful mechanical and painful laser heat stimuli. All participants received written and oral information prior to the experiments, and gave written consent prior to the experiments, ensuring compliance with the Declaration of Helsinki. The experiment was approved by the local ethical committee (N-20140093).
2.2 Experimental protocol
The experiment consisted of four blocks, two before and two following capsaicin sensitization (Fig. 1). Each block included both a line and 2-point stimulation paradigm in randomized order. The stimulation modality (laser or mechanical) was randomized between the two blocks before and after capsaicin, so that both laser and mechanical stimulations were applied both before and after the capsaicin. Before testing began, the stimulation area was drawn on the volar forearm (Fig. 1), and any excessive hair growth was shaved. When shaving it was ensured that the stratum corneum was not affected but that only the hairs were removed.

Experimental protocol. Two stimulation blocks were completed before and after capsaicin application. Each block consisted of a line stimulation and 2-point stimulation paradigms in randomized orders. The stimulation modality in each block (laser or mechanical) was randomized. To monitor the effect of the capsaicin the HPT was determined before the capsaicin application and following block 4, this was done to ensure that heat hyperalgesia was still present during blocks 3 and 4. The capsaicin patch was 5×14 cm and applied in the volar forearm. All line and 2-point stimulations were administered within the capsaicin treated area. HPT was also tested within the capsaicin treated area.
The line stimulations were used to test the DDT by displacing a continuous stimulus along a line parallel to the forearm. The lines were administered in four different lengths (25, 50, 75 and 100 mm) and in two different directions – either towards the elbow (proximal orientation) or towards the wrist (distal orientation). Each combination of length and direction was repeated twice for both tactile and painful heat stimulations. Lines were stimulated with a fixed velocity of 35 mm/s [3]. After each line stimulation, the subject had to indicate the direction of the line (either proximally or distally, i.e. a forced choice task paradigm) and the perceived intensity on a NRS scale. The NRS scale was set as: 0 – no perception, 3 – pain threshold, 10 – maximum pain imaginable. This scale was used since both non-painful and painful stimuli were applied [4].
Two concurrent single point stimulations were used to test the 2-point discrimination threshold. The subjects were instructed that either 1 or 2 points would be administered. Following each stimulations, the subjects had to indicate the number of perceived points (1 or 2, i.e. forced choice task) and they had to indicate the perceived intensity on the NRS scale. The distance between the two points varied in steps of 10 mm ranging from 0 (only a single point stimulation) to 100 mm. The single point stimuli were used as a bias control. Each distance was applied twice in randomized order. The duration of the stimulation was approx. 1.5 s for both laser and mechanical stimuli.
2.3 Skin sensitization
An 8% Capsaicin patch (Qutenza®, Astellas) was used to sensitize the stimulated skin area. The patch contains 640 μg of capsaicin per cm2. The patch was cut to a size of 5×14 cm and placed on the volar forearm for 30 min. The size of the area was chosen to ensure that all tactile and laser stimuli were applied within the sensitized area. After removal of the capsaicin patch, the included cleansing gel was used to inactivate any capsaicin in the superficial skin. This however, does not affect the sensitization of the skin.
The heat pain threshold (HPT) was determined before capsaicin application and after block 4 using a contact thermode (Pathway, ATS, Medoc, Israel). The thermode was placed within the skin area treated with capsaicin. The temperature baseline was 32 °C, and the temperate rise rate was 1 °C/s. The subjects were instructed to press a button when the temperature reached pain threshold, hereafter the temperature returned to baseline (5 °C/s). A total of three trials were conducted to determine HPT before and after capsaicin application, each trial was separated by at least 60 s. In one subject HPT was not determined due to equipment failure, but all other measurements were included in the data analysis.
2.4 Laser stimulation
The noxious heat stimuli was delivered using a 100 W CO2 laser (Firestar ti-series, Synrad, USA), the beam was transmitted through a 5× beam expander and then into a scanner head (GSI Lumonics General Scanning XY10A) to allow rapid and accurate displacement of the laser beam. To obtain a beam diameter of 5 mm (1/e2) the laser beam was rapidly moved in small concentric circles (dithering), the dithering was so small that it did not affect the perception i.e. the stimulations were perceived as continuous.
The laser intensity was calibrated for each individual subject, so that a 50 mm line stimulation was perceived as having an intensity of four on the NRS scale. Similarly, the intensity of the point stimulations were calibrated so that a single point stimulation was perceived as four on the NRS scale. To obtain a simultaneous two point stimulation, the laser stimulated one point and was then rapidly moved the second point, and stimulated this point. When moving from one point to another the laser shut off. The sequence was repeated 20 times per second. This stimulation caused a continuous and concurrent stimulation at each point [3], [4].
The skin temperature was measured throughout the experiment using an infrared camera (Agema 900, FLiR, Sweden), the experiment was stopped if the skin temperature exceeded 50 °C. During the stimulations, both the subject and investigator wore protective googles.
2.5 Mechanical stimulation
The mechanical line stimulations were delivered using a Somedic Senselab Brush-05 (Somedic, Sweden), the brush is calibrated to 400 mN and has a width of approx. 5 mm and a length of approx. 15 mm. To ensure a stimulation speed of 35 mm/s and correct stimulation distances, a visible HeNe (Helium-Neon) laser was inserted into the path of the CO2 laser (CO2 laser was shut off). The beam of the HeNe laser was directed through the scanner head onto the skin. The mechanical line stimulations were stimulated in the same lengths and directions as the laser line stimulations.
To test the mechanical 2-point discrimination the skin was stimulated using a Vernier caliber with two blunted plastic filaments both with a diameter of approx. 5 mm. It was ensured that both filaments touched the skin simultaneously.
2.6 Threshold estimations
In order to find the discriminative thresholds for the line and 2-point stimulations, the responses were fitted to a sigmodal curve (Matlab, Natick, MA, USA) [3], [4], [16] (Eq. 1).
where b corresponds to the point where y=0.75, and a is the slope of the curve at this point. For the line stimulations c and d were both 0.5, this means that 0.75 corresponds to the point where the subjects answer 75% correct, i.e. half between merely guessing and absolute certainty. For the 2-point stimulations, c was 1 and d was 0. Prior to the fit, the responses to the 2-point stimulations (either 1 or 2), were subtracted 1, i.e. 1’s were converted to 0, and 2’s were converted to 1 [3].
2.7 Statistics
Normality was confirmed before statistical tests were applied.
The NRS for the line stimulations were analyzed using a linear mixed model (LMM). The stimulation direction, length and pre/post capsaicin were used as factors. Similarly the NRS from the 2-point stimuli were analyzed in another LMM with the distance between the points and pre/post capsaicin as factors. Each analysis was made independently for each modality (mechanical or laser).
A logistic regression model was used to investigate difference in correctness for the laser line responses. Factors were set as stimulation direction, correctness and whether the stimulation was perceived as painful. To ensure that there was no order effects between the blocks before and after capsaicin the logistical regression model also included the stimulation order as a covariate.
A Student’s t-test was applied to compare HPT before and after capsaicin application.
p-Values less than 0.05 was considered significant. All statistical tests were performed using SPSS 23 (IBM, Armonk, NY, USA). Results are reported as mean±standard deviation and the fitted values are reported as mean±95% confidence intervals (CI).
3 Results
3.1 Directional discrimination threshold
For the laser stimulations, the DDTs for the distal direction were 67.2 mm (95% CI: 19.9–114.6 mm) before capsaicin and 70.9 mm (95% CI: 37.7–104.1 mm) after capsaicin. For the proximal direction DDT was 68.7 mm (95% CI: 50.9–86.6 mm) before capsaicin and 80.3 mm (95% CI: 12.8–147.9 mm) following capsaicin. Since there were no difference in direction, the data was pooled for both directions, thus, DDT was 69.5 mm (95% CI: 51.0–88.1 mm) before capsaicin and 76.3 mm (95% CI: 31.4–121.2 mm) after capsaicin. However, the 95% CI did overlap in all of these cases, despite an increase in DDT of at least 6% and in some cases as much as 17%. Following the capsaicin application, the variance of the responses was increased, as shown by a widening of the 95% CI (Fig. 2).
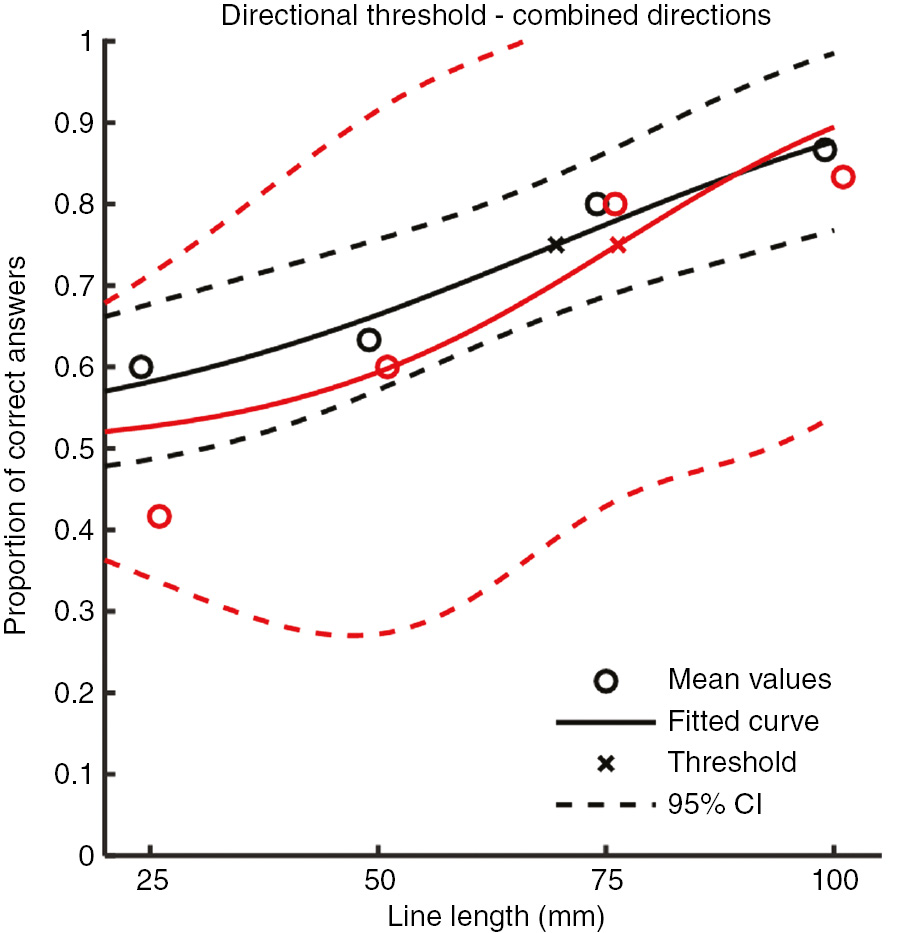
Directional discrimination threshold for laser stimulation. Black indicates responses before capsaicin and red indicate responses following capsaicin. For 7 out of 8 stimulation lengths the post capsaicin responses were less correct than before capsaicin, however, the 95% CI overlaps for all stimulation lengths. A logistic regression analysis showed that the correctness of the response to the laser line stimulations was significantly lower after the capsaicin administration (Log. Reg., p<0.05) indicating an impairment of the directional discrimination following capsaicin.
For mechanical stimuli, all stimulations were perceived correctly before the capsaicin application (not depicted). However, following capsaicin 3 (out of 240) of the mechanical line stimulations were perceived incorrectly, but no fit could be created to estimate the DDT.
For the laser line stimuli, NRS was significantly increased following capsaicin sensitization (Fig. 3, LMM, p<0.001), and the NRS increased with stimulation length (Fig. 3, LMM, p<0.001), but there were no difference in NRS in relation to stimulation directions (Fig. 3, LMM, p=0.059).

Reported NRS following line stimulations. (A, B): laser stimulation, (C, D): mechanical stimulation. Black indicates responses before capsaicin and red indicate responses following capsaicin. The horizontal black line at NRS=3 indicates pain threshold. Triangles indicate direction of stimulus. (A, C): NRS in relation to stimulation length. (B, D): NRS in relation to correctness of answer. For the laser stimuli (A, B), the perceived intensity increased with stimulation length (ANOVA, p<0.001), but not mechanical (C, D). There was no effect of stimulation direction for neither stimulation modality. Note: different truncation of y axis for better visualization. For mechanical stimulation only three answers were incorrect, two for the distal direction and one for the proximal direction.
For the mechanical stimuli, NRS was significantly increased following capsaicin sensitization (Fig. 3, LMM, p<0.001). The stimulation length did not affect the perceived intensity for the mechanical stimuli (Fig. 3, LMM, p=0.085). There was no difference in perceived intensity depending on stimulation direction (Fig. 3, LMM, p=0.873).
The logistic regression showed that the correctness of the response to the laser line stimulations was significantly lower after the capsaicin administration (Log. Reg., p<0.05) and the correctness was significantly higher if the stimulation was perceived as painful (Log. Reg., p<0.01). Finally, the logistic regression showed that the stimulation order did not explain the correctness of the response (Log. Reg., p=0.369), indicating that the changes in directional discrimination was not due to an order effect.
3.2 Two-point discrimination threshold
The 2-point discrimination for laser stimuli was 70.3 mm (95% CI: 58.2–82.6 mm) before capsaicin and 68.0 mm (95% CI: 57.2–78.7 mm) after capsaicin, for the mechanical stimuli it was 31.5 mm (95% CI: 29.9–33.0 mm) before capsaicin and 31.0 mm (95% CI: 29.5–32.6 mm) after capsaicin (Fig. 4). The overlap of the 95% CI indicates no differences. While the capsaicin sensitization does not appear to affect the 2-point discrimination threshold, the slope of the fitted curve at threshold, however, does appear to change for both laser and mechanical stimuli (Fig. 4). For laser stimuli the slope at the threshold decreased from 0.031 (perceived points/mm) for to 0.027 (perceived points/mm) following capsaicin, while for the mechanical stimuli the slope changed from 0.218 to 0.117 following capsaicin.

Two-point discrimination threshold and NRS responses. (A–C): laser stimulations, (D–F): mechanical stimulation. Black indicates responses before capsaicin and red indicate responses following capsaicin. The horizontal black line at NRS=3 indicates pain threshold. (A, D): fitted curves to find the 2-point discrimination threshold. (B, E): NRS in relation to the distance between the points. (C, F): NRS in relation to the perceived number of points, NRS was higher for stimuli perceived as one point for laser stimuli before capsaicin (C, black) (Student’s t-test, p<0.001) and for mechanical stimuli after capsaicin (F, red) (Student’s t-test, p<0.05). Note: Y-axis were truncated for better visualization.
For the laser 2-point stimuli, NRS significantly increased following capsaicin (LMM; p<0.001), and the NRS increased with the distance between points (LMM, p<0.001).
For mechanical 2-point stimuli, NRS was significantly increased following capsaicin (LMM; p<0.001), and the NRS increased with the distance between points (LMM, p<0.01).
3.3 Effect of capsaicin sensitization
HPT was significantly reduced from 44.8±2.2 °C to 40.3±2.6 °C following the capsaicin application (t-test, p<0.001 – Fig. 5). This indicates that the capsaicin sensitization is still in effect following stimulation block 4. The average perceived intensity during the capsaicin application was 4.6±2.2 on the NRS scale.

Heat pain threshold (HPT) – pre and post capsaicin sensitization. HPT was measured within the area where capsaicin was applied. HPT was significantly reduced following capsaicin application (t-test, p<0.001).
4 Discussion
In this study, the effect of cutaneous capsaicin sensitization on directional discrimination and 2-point discrimination was investigated. It was shown that the DDT appears to increase (correctness of responses were lower after capsaicin administration) whilst the 2-point discrimination threshold did not. Expectedly, we also show that the perceived intensities were increased following capsaicin application. The fact that the DDT was affected by capsaicin, whereas the 2-point discrimination threshold was not, could possibly indicate that the underlying discrimination mechanism between these two types of stimuli are inherently different.
4.1 The effect of cutaneous sensitization on discrimination
Previous studies have shown that several pain conditions affect the perception of tactile stimuli and cause reduced tactile acuity [12] as well as causing tactile sensory abnormalities, such as tactile hypoesthesia [13]. Similarly, experimental induced sensitization using capsaicin has been reported to increase the mechanical 2-point discrimination threshold [15]. Thus, in this study we applied both painful and non-painful stimuli to investigate how the discriminative properties are altered by cutaneous sensitization with topical capsaicin.
The current study show that the average DDT for painful laser stimulation does increase by 6–17% on average (Fig. 2) and a logistic regression analysis did show a significant increase in DDT following capsaicin sensitization. It should also be noted that the 95% CI increased following the sensitization. Thus, this study is the first to show the ability to discriminate the direction of painful stimulus appear to be impaired following capsaicin sensitization. In addition, this study also show that the directional discrimination is significantly better if the stimulation is perceived as painful. This means that directional discrimination may improve if using higher stimulation intensities. Since the capsaicin sensitization increases the perceived intensities of the stimuli, one could assume this would improve the discrimination. This is however, not the case, as the capsaicin appears to impair the directional discrimination. This shows the changed directional discrimination depend on other mechanisms than those merely related to intensity, and the directional discrimination paradigm could potentially be used to investigate these mechanisms.
Furthermore, this study show that the 2-point discrimination threshold (Fig. 4) did not change, but the slope of the fitted curve is flatter, meaning the threshold is not as well defined following the capsaicin sensitization. This finding actually fits well with the study by Kauppila et al. which show that the mechanical 2-point discrimination threshold increases following capsaicin sensitization. There are, however, several methodological difference between that study [15] and the present study. Most importantly, the study by Kauppila et al. defined the 2-point discrimination threshold as 80% correct answers, unlike 50% in most other studies [3], [4], [16]. In fact Fig. 4D shows that the distance between the points resulting in 80% answer correctness is increased following capsaicin sensitization i.e. showing good agreement with the study by Kauppila et al.
One could argue that the increase in the 95% CI for the DDT and the flatter curve of the 2-point discrimination could be caused by noise in the perception due to the induced sensitization. Alternatively, it could also simply reflect that the subjects had difficulties discriminating between the test stimuli and the continuous sensations caused by the capsaicin sensitization despite that the subjects were asked to disregard the continuous sensations that may be caused by the capsaicin and focus on either the line or 2-point stimulus.
Another potential mechanism which could affect the discriminative abilities may be cortical reorganization [12], which previously has been shown to be correlated both to the degree of pain but also the spatial acuity [17] and the perception threshold [18]. Cortical reorganization has been shown to cause changes in the exteroceptive system. However, it have been suggested that cortical changes are related to the chronicity of the pain [18], thus, it may be expected that such changes takes longer time to establish than the extent of the experimental pain model used in this study. Other studies have, in contrast, found evidence indicating minor cortical changes induced by capsaicin in the acute phase [19] at least for mechanical brush stimuli. This may explain why the post-capsaicin directional discrimination for mechanical stimuli in the present study exhibits 3 erroneous responses compared to no errors pre-capsaicin.
Lateral inhibition or surround inhibition is the neural process in which an excited sensory neuron inhibits its surrounding neurons by inhibitory projections between the neurons. This mechanism has previously been studied for the visual sense [20], but also for the cutaneous senses, e.g. during heat stimuli [21]. Our research group has previously found evidence suggesting that lateral inhibition is present in the nociceptive system [22]. Previously we have hypothesized that the directional discrimination depends on the lateral inhibition mechanism [4]. It is unsure how lateral inhibition is affected during sensitization. However, it is well-known that capsaicin will cause both peripheral and central sensitization of the nociceptive system [23]. The sensitization and/or the pain may affect the endogenous inhibitory mechanisms and thus it would be expected that lateral inhibition would also be affected. Additionally, the changes in tactile acuity seen during chronic pain may be due to reduced lateral inhibition [22]. Therefore, it is likely that lateral inhibition is changed following a capsaicin sensitization. Thus, the increased DDT seen in this study could be somewhat linked to an altered lateral inhibition. In addition, capsaicin sensitization has been shown to increase the receptive field size of Wide Dynamic Range neurons, which, similar to a change in lateral inhibition, will impair the discriminative abilities [15], thus, either of these two mechanisms could be involved in the reduced discrimination abilities following capsaicin sensitization.
Finally, it is interesting to note that the discrimination of a moving tactile stimulus has been shown to be reduced in patients with spinal cord disorders [24]. Hankey and Edis applied a scratch stimulus to investigate both patients and controls, and it was found that patients had more difficulty discriminating the direction of the scratch. This led the authors to suggest that the method could be used clinically to screen such a patient group [24], this could perhaps also be considered in pain patients.
4.2 The effect of cutaneous sensitization on perception intensity
Capsaicin will sensitize the nociceptors by activating the TRP-V1 ion channel [23], since this receptor is also activated by noxious heat stimulation, it is not surprising that the perceived intensities to laser stimulation were increased following the capsaicin administration [25], since the laser stimuli will activate this ion channel. Thus, the mechanism behind the increased pain perception to the laser stimuli is most likely due to the topical capsaicin creating a primary heat hyperalgesia, caused by sensitization of peripheral nociceptors [26]. In addition, the perceived intensity of the mechanical stimuli also increases, but the perceived intensities were not above pain threshold. Capsaicin has previously been reported to also induce changes such as allodynia and mechanical hyperalgesia [14], [27], [28] so the hyperesthesia seen in this study fits well with the literature.
4.3 Methodological considerations
Capsaicin can be applied as a cutaneous pain model both by topical and intradermal injection [23], [29]. The intradermal injection will typically lead to more robust changes [29], however, it is very difficult to control the affected area, and therefore, the current study used topical capsaicin to achieve a well-defined sensitized area. Additionally, intradermal capsaicin is known to induce more mechanical hyperalgesia than topical administration [23]. Furthermore, the intradermal capsaicin injection typically does not give as a profound primary heat hyperalgesia as seen using topical capsaicin [28]. The topical capsaicin will induce both peripheral and central sensitization [29], both of which could potentially affect the discrimination. However, in the current study it is not directly possible to deduce how the changed directional discrimination depends on either peripheral or central changes. Thus, to further investigate the purely central mechanisms of discrimination it may be of interest to repeat the current study within the secondary hyperalgesic area irrespective that hyperalgesia to heat might be minimal.
The 8% capsaicin patch used in this study, can also be used to treat neuropathic pain [23], however, for this use the application time is longer (60–90 min), than what was used in this study (30 min). The background for this treatment is the fact that prolonged exposure to capsaicin will desensitize the cutaneous nerve fibers expressing the TRP-V1 channel. This has been developed into an experimental protocol which applies the 8% capsaicin patch for 2×22 h to robustly desensitize the skin [30]. However, the exposure times in that protocol are much longer than those applied in this study. Furthermore, if the capsaicin patch had resulted in an acute deafferention or desensitization, this would lead to an increased HPT [30], which was not observed in the present study, where in fact, the HPT decreased (Fig. 5). Thus, our results indicate that the skin was indeed sensitized to thermal stimuli during blocks 3 and 4.
Finally, the localization of chemical stimuli (capsaicin) showed lower acuity than for tactile stimuli [31]. Therefore, care was taken to ensure that the subjects focused on the phasic stimulation, administered either by laser or mechanical stimuli, rather than the tonic sensation caused by the capsaicin.
-
Authors’ statements
-
Research funding: This study was funded the Danish National Research Foundation (DNRF121).
-
Conflict of interest: Authors state no conflict of interest.
-
Informed consent: Informed consent has been obtained from all individuals included in this study.
-
Ethical approval: The research related to human use complies with all the relevant national regulations, institutional policies and was performed in accordance with the tenets of the Helsinki Declaration, and has been approved by the local ethical committee (N-20140093).
References
[1] Johnson KO, Phillips JR. Tactile spatial resolution. I. Two-point discrimination, gap detection, grating resolution, and letter recognition. J Neurophysiol 1981;46:1177–91.10.1152/jn.1981.46.6.1177Search in Google Scholar PubMed
[2] Freeman C, Okun MS. Origins of the sensory examination in neurology. Semin Neurol 2002;22:399–407.10.1055/s-2002-36762Search in Google Scholar PubMed
[3] Mørch CD, Andersen OK, Quevedo AS, Arendt-Nielsen L, Coghill RC. Exteroceptive aspects of nociception: insights from graphesthesia and two-point discrimination. Pain 2010;151:45–52.10.1016/j.pain.2010.05.016Search in Google Scholar PubMed PubMed Central
[4] Frahm KS, Mørch CD, Andersen OK. Tempo-spatial discrimination is lower for noxious stimuli than for innocuous stimuli. Pain 2018;159:393–401.10.1097/j.pain.0000000000001095Search in Google Scholar PubMed
[5] Costanzo RM, Gardner EP. A quantitative analysis of responses of direction-sensitive neurons in somatosensory cortex of awake monkeys. J Neurophysiol 1980;43:1319–41.10.1152/jn.1980.43.5.1319Search in Google Scholar PubMed
[6] Hyvarinen J, Poranent A. Movement-sensitive and direction and orientation-selective cutaneous receptive fields in the hand area of the post-central gyrus in monkeys. J Physiol 1978;283:523–37.10.1113/jphysiol.1978.sp012517Search in Google Scholar PubMed PubMed Central
[7] Graven-Nielsen T, Aspegren Kendall S, Henriksson KG, Bengtsson M, Sörensen J, Johnson A, Gerdle B, Arendt-Nielsen L. Ketamine reduces muscle pain, temporal summation, and referred pain in fibromyalgia patients. Pain 2000;85:483–91.10.1016/S0304-3959(99)00308-5Search in Google Scholar PubMed
[8] Arendt-Nielsen L, Drewes AM, Hansen JB, Tage-Jensen U. Gut pain reactions in man: an experimental investigation using short and long duration transmucosal electrical stimulation. Pain 1997;69:255–62.10.1016/S0304-3959(96)03244-7Search in Google Scholar PubMed
[9] Nikolajsen L, Hansen CL, Nielsen J, Keller J, Arendt-Nielsen L, Jensen TS. The effect of ketamine on phantom pain: A central neuropathic disorder maintained by peripheral input. Pain 1996;67:69–77.10.1016/0304-3959(96)03080-1Search in Google Scholar PubMed
[10] Woolf CJ, Bennett GJ, Doherty M, Dubner R, Kidd B, Koltzenburg M, Lipton R, Loeser JD, Payne R, Torebjork E. Towards a mechanism-based classification of pain? Pain 1998;77:227–9.10.1016/S0304-3959(98)00099-2Search in Google Scholar PubMed
[11] Catley MJ, O’Connell NE, Berryman C, Ayhan FF, Moseley LG. Critical review is tactile acuity altered in people with chronic pain? A systematic review and meta-analysis. J Pain 2014;15:985–1000.10.1016/j.jpain.2014.06.009Search in Google Scholar PubMed
[12] Moseley GL, Zalucki NM, Wiech K. Tactile discrimination, but not tactile stimulation alone, reduces chronic limb pain. Pain 2008;137:600–8.10.1016/j.pain.2007.10.021Search in Google Scholar PubMed
[13] Moriwaki K, Yuge O. Topographical features of cutaneous tactile hypoesthetic and hyperesthetic abnormalities in chronic pain. Pain 1999;81:1–6.10.1016/S0304-3959(98)00257-7Search in Google Scholar PubMed
[14] Olesen AE, Andresen T, Staahl C, Drewes AM. Human experimental pain models for assessing the therapeutic efficacy of analgesic drugs. Pharmacol Rev 2012;64:722–79.10.1124/pr.111.005447Search in Google Scholar PubMed
[15] Kauppila T, Mohammadian P, Nielsen J, Andersen OK, Arendt-nielsen L. Capsaicin-induced impairment of tactile spatial discrimination ability in man: indirect evidence for increased receptive fields in human nervous system. Brain Res 1998;797:361–7.10.1016/S0006-8993(98)00431-4Search in Google Scholar
[16] Schlereth T, Magerl W, Treede R. Spatial discrimination thresholds for pain and touch in human hairy skin. Pain 2001;92:187–94.10.1016/S0304-3959(00)00484-XSearch in Google Scholar
[17] Maihöfner C, Neundörfer B, Birklein F, Handwerker HO. Mislocalization of tactile stimulation in patients with complex regional pain syndrome. J Neurol 2006;253:772–9.10.1007/s00415-006-0117-zSearch in Google Scholar PubMed
[18] Flor H, Braun C, Elbert T, Birbaumer N. Extensive reorganization of primary somatosensory cortex in chronic back pain patients. Neurosci Lett 1997;224:5–8.10.1016/S0304-3940(97)13441-3Search in Google Scholar
[19] Witting N, Kupers RC, Svensson P, Arendt-Nielsen OL, Gjedde A, Jensen TS. Experimental brush-evoked allodynia activates posterior parietal cortex. Neurology 2001;57:1817–24.10.1212/WNL.57.10.1817Search in Google Scholar PubMed
[20] Hartline HK, Wagner HG, Ratliff F. Inhibition in the eye of Limulus. J Gen Physiol 1956;39:651–73.10.1085/jgp.39.5.651Search in Google Scholar PubMed PubMed Central
[21] Békésy GV. Lateral inhibition of heat sensations on the skin. J Appl Physiol 1962;17:1003–8.10.1152/jappl.1962.17.6.1003Search in Google Scholar PubMed
[22] Quevedo AS, Mørch CD, Andersen OK, Coghill RC. Lateral inhibition during nociceptive processing. Pain 2017;158:1046–52.10.1097/j.pain.0000000000000876Search in Google Scholar PubMed PubMed Central
[23] O’Neill J, Brock C, Olesen AE, Andresen T, Nilsson M, Dickenson AH. Unravelling the mystery of capsaicin: a tool to understand and treat pain. Pharmacol Rev 2012;64:939–71.10.1124/pr.112.006163Search in Google Scholar PubMed PubMed Central
[24] Hankey GJ, Edis RH. The utility of testing tactile perception of direction of scratch as a sensitive clinical sign of posterior column dysfunction in spinal cord disorders. J Neurol Neurosurg Psychiatry 1989;52:395–8.10.1136/jnnp.52.3.395Search in Google Scholar PubMed PubMed Central
[25] Hüllemann P, von der Brelie C, Manthey G, Düsterhöft J, Helmers AK, Synowitz M, Baron R. Reduced laser-evoked potential habituation detects abnormal central pain processing in painful radiculopathy patients. Eur J Pain (United Kingdom) 2017;21:918–26.10.1002/ejp.994Search in Google Scholar PubMed
[26] Lamotte RH, Lundberg LER, Torebjork HE. Pain, hyperalgesia and acitivityin nociceptive C units in humans after intradermal injection of capsaicin. J Physiol 1992;448:749–64.10.1113/jphysiol.1992.sp019068Search in Google Scholar PubMed PubMed Central
[27] Walder RY, Radhakrishnan R, Loo L, Rasmussen LA, Mohapatra DP, Wilson SP, Sluka KA. TRPV1 is important for mechanical and heat sensitivity in uninjured animals and development of heat hypersensitivity after muscle inflammation. Pain 2012;153:1664–72.10.1016/j.pain.2012.04.034Search in Google Scholar PubMed PubMed Central
[28] Klein T, Magerl W, Rolke R, Treede RD. Human surrogate models of neuropathic pain. Pain 2005;115:227–33.10.1016/j.pain.2005.03.021Search in Google Scholar PubMed
[29] Arendt-Nielsen L, Andersen OK. Capsaicin in human experimental pain models of skin, muscle and visceral sensitization. In: Malmberg AB, Bley KR, editors. Turning up the heat on pain: TRPV1 receptors in pain and inflammation. Basel: Birkh{ä}user Basel, 2005:117–44.10.1007/3-7643-7379-2_7Search in Google Scholar
[30] Henrich F, Magerl W, Klein T, Greffrath W, Treede RD. Capsaicin-sensitive C- and A-fibre nociceptors control long-term potentiation-like pain amplification in humans. Brain 2015;138:2505–20.10.1093/brain/awv108Search in Google Scholar PubMed
[31] Green BG, Flammer LJ. Localization of chemical stimulation: capsaicin on hairy skin. Somatosens Mot Res 1989;6:553–66.10.3109/08990228909144692Search in Google Scholar PubMed
©2019 Scandinavian Association for the Study of Pain. Published by Walter de Gruyter GmbH, Berlin/Boston. All rights reserved.
Articles in the same Issue
- Frontmatter
- Editorial comments
- Information, back pain, and radiology
- Painful Willis-Ekbom disease: unbearable and distinct form of restless legs?
- Systematic review
- The effect of exercise therapy combined with psychological therapy on physical activity and quality of life in patients with painful diabetic neuropathy: a systematic review
- Clinical pain research
- A 4-year follow-up of non-freezing cold injury with cold allodynia and neuropathy in 26 naval soldiers
- Predicting chronic pain after major traumatic injury
- Dynamic assessment of the pupillary reflex in patients on high-dose opioids
- Decline of substance P levels after stress management with cognitive behaviour therapy in women with the fibromyalgia syndrome
- Characterization of painful Restless Legs Syndrome sensations in an English-speaking South African population
- Opioid prescribing habits differ between Denmark, Sweden and Norway – and they change over time
- Psychological factors can cause false pain classification on painDETECT
- Are attitudes about pain related to coping strategies used by adolescents in the community?
- Field testing of the revised neuropathic pain grading system in a cohort of patients with neck and upper limb pain
- Patient reported outcomes and neuropsychological testing in patients with chronic non-cancer pain in long-term opioid therapy: a pilot study
- Observational studies
- Lessons learned from piloting a pain assessment program for high frequency emergency department users
- The Portuguese 35-item Survey of Pain Attitudes applied to Portuguese women with Endometriosis
- Original experimental
- Development of a new bed-side-test assessing conditioned pain modulation: a test-retest reliability study
- Test-retest repeatability of questionnaire for pain symptoms for school children aged 10–15 years
- Unique brain regions involved in positive versus negative emotional modulation of pain
- What decreases low back pain? A qualitative study of patient perspectives
- Cutaneous nociceptive sensitization affects the directional discrimination – but not the 2-point discrimination
- Educational case report
- A painful foot with diagnostic and therapeutic consequences
- Short communications
- Educational interventions to improve medical students’ knowledge of acute pain management: a randomized study
- Possible inflammatory pain biomarkers in postamputation pain
- An online investigation into the impact of adding epidemiological information to imaging reports for low back pain
- Letter to the Editor
- Reply to Letter to the Editor “Clinical registries are essential tools for ensuring quality and improving outcomes in pain medicine” by Baciarello et al.
Articles in the same Issue
- Frontmatter
- Editorial comments
- Information, back pain, and radiology
- Painful Willis-Ekbom disease: unbearable and distinct form of restless legs?
- Systematic review
- The effect of exercise therapy combined with psychological therapy on physical activity and quality of life in patients with painful diabetic neuropathy: a systematic review
- Clinical pain research
- A 4-year follow-up of non-freezing cold injury with cold allodynia and neuropathy in 26 naval soldiers
- Predicting chronic pain after major traumatic injury
- Dynamic assessment of the pupillary reflex in patients on high-dose opioids
- Decline of substance P levels after stress management with cognitive behaviour therapy in women with the fibromyalgia syndrome
- Characterization of painful Restless Legs Syndrome sensations in an English-speaking South African population
- Opioid prescribing habits differ between Denmark, Sweden and Norway – and they change over time
- Psychological factors can cause false pain classification on painDETECT
- Are attitudes about pain related to coping strategies used by adolescents in the community?
- Field testing of the revised neuropathic pain grading system in a cohort of patients with neck and upper limb pain
- Patient reported outcomes and neuropsychological testing in patients with chronic non-cancer pain in long-term opioid therapy: a pilot study
- Observational studies
- Lessons learned from piloting a pain assessment program for high frequency emergency department users
- The Portuguese 35-item Survey of Pain Attitudes applied to Portuguese women with Endometriosis
- Original experimental
- Development of a new bed-side-test assessing conditioned pain modulation: a test-retest reliability study
- Test-retest repeatability of questionnaire for pain symptoms for school children aged 10–15 years
- Unique brain regions involved in positive versus negative emotional modulation of pain
- What decreases low back pain? A qualitative study of patient perspectives
- Cutaneous nociceptive sensitization affects the directional discrimination – but not the 2-point discrimination
- Educational case report
- A painful foot with diagnostic and therapeutic consequences
- Short communications
- Educational interventions to improve medical students’ knowledge of acute pain management: a randomized study
- Possible inflammatory pain biomarkers in postamputation pain
- An online investigation into the impact of adding epidemiological information to imaging reports for low back pain
- Letter to the Editor
- Reply to Letter to the Editor “Clinical registries are essential tools for ensuring quality and improving outcomes in pain medicine” by Baciarello et al.

