Abstract
Bio-based polyamide 56 (PA56), as an emerging green and environmentally friendly material, has garnered significant attention across various industries, including textiles, automotive, and electronics, due to its excellent mechanical properties, thermal resistance, and renewability. Despite these advantages, polyamide 56 faces challenges related to processability, impact resistance, flame retardancy, and other functional properties. To expand its application scope and enhance the added value of polyamide 56 products, improving its performance through various modification techniques has become a focal point of research. This paper provides a comprehensive review of recent advances in modification strategies for polyamide 56, including reinforcement, flame retardancy, toughening, and antibacterial treatments. Additionally, this paper discusses the future application prospects of polyamide 56 and offers valuable insights to guide ongoing research in this field.
1 Introduction
Polyamides (PAs) are widely used in various industries due to their exceptional mechanical strength, superior thermal stability, outstanding chemical resistance, and excellent processability. 1 , 2 Nevertheless, the production of conventional PAs is heavily dependent on petrochemical resources, a dependency that inevitably contributes to significant greenhouse gas emissions and environmental degradation, thus challenging the current “dual-carbon” policy goals. 3 In addition, the depletion of petrochemical resources and the ongoing energy crisis have further constrained the development and sustainability of petrochemical-based polyamides. Therefore, exploring and developing environmentally friendly and sustainable bio-based polyamide materials have become a prominent frontier and a key area of innovation in the field of polymer science. 4 , 5 , 6
Bio-based polyamide materials refer to the use of renewable biomass as raw materials (such as plant oils and polysaccharide derivatives) through biological, chemical and physical means to create monomers used in the synthesis of polyamide. The monomers are then polymerized to form high-performance materials with green, environmentally friendly, and renewable characteristics, such as PA1010, PA1012, PA56, PA10T, and PA11. Among these, PA56 not only retains the desirable properties characteristic of conventional polyamides, but also exhibits unique physical properties, making it a feasible alternative for high-performance applications in industries such as textiles and engineering plastics.
PA56 is synthesized through the condensation reaction of bio-based 1,5-pentanediamine and petrochemical-based adipic acid. 7 Compared to traditional petrochemical-derived polyamides, its monomer sources significantly reduce the dependence on petrochemical resources and its carbon footprint is notably lower. Therefore, PA56 is considered a promising green material in the “low-carbon economy” era, with tremendous market potential in various industries such as automotive, electronics, household appliances, and textiles. 8 , 9 , 10 Although PA56 has great potential in terms of its green and renewable properties, its unique molecular structure also presents certain challenges. Due to the special arrangement of even–odd carbon atoms, PA56 has fewer hydrogen bonds in its molecular chain, which affects intermolecular interactions and, to some extent, impacts its mechanical properties and limits its processability. 11 , 12 , 13 Furthermore, this molecular structure may result in insufficient durability and stability in complex application environments. Therefore, the processability and applicability of PA56 require further enhancement.
To overcome these limitations, researchers are exploring various modification strategies, such as composite technology, copolymerization, and crosslinking, to enhance the intermolecular interactions within PA56, thereby improving its mechanical strength, thermal stability, and chemical resistance. 14 Consequently, enhancing the performance of PA56 through modification technologies and optimizing its applicability in practical applications has become a key area of research. These studies will not only accelerate the industrialization of PA56 but also provide valuable insights for the development of other bio-based materials.
2 Synthesis of PA56
PA56 refers to a polyamide where at least one of the monomers used in its synthesis is derived from biobased feedstocks through fermentation processes. Currently, the commonly available PA56 employs biobased 1,5-pentanediamine as a monomer. This 1,5-pentanediamine is produced through microbial fermentation of biomass, such as corn, to produce L-lysine. The L-lysine is subsequently converted into 1,5-pentanediamine via microbial fermentation or whole-cell catalysis. 15 Subsequently, biobased 1,5-pentanediamine is reacted with petroleum-derived adipic acid to prepare a polyamide 56 salt solution with a mass concentration of approximately 50 %. The salt solution is then transferred to a high-pressure polymerization reactor, where air is displaced with nitrogen 3–5 times. The temperature within the reactor is gradually increased, and the salt solution is subjected to low-temperature concentration. When the material temperature reaches 230 °C and the pressure inside the reactor increases to 2.0 MPa, venting and depressurization are initiated. Once the material temperature reaches 265 °C and the pressure inside the reactor gradually decreases to 0.2 MPa, followed by the application of vacuum to reach −0.06 MPa (gauge vacuum pressure). The product is then discharged through melting, cast into ribbons, pelletized, dried, and packaged to obtain PA56 pellets. A crucial aspect of the polymerization process is the removal of trace amounts of water from the system, which is essential to achieving a high molecular weight for PA56. 16 The preparation process of PA56 is shown in the following figure (Figure 1).
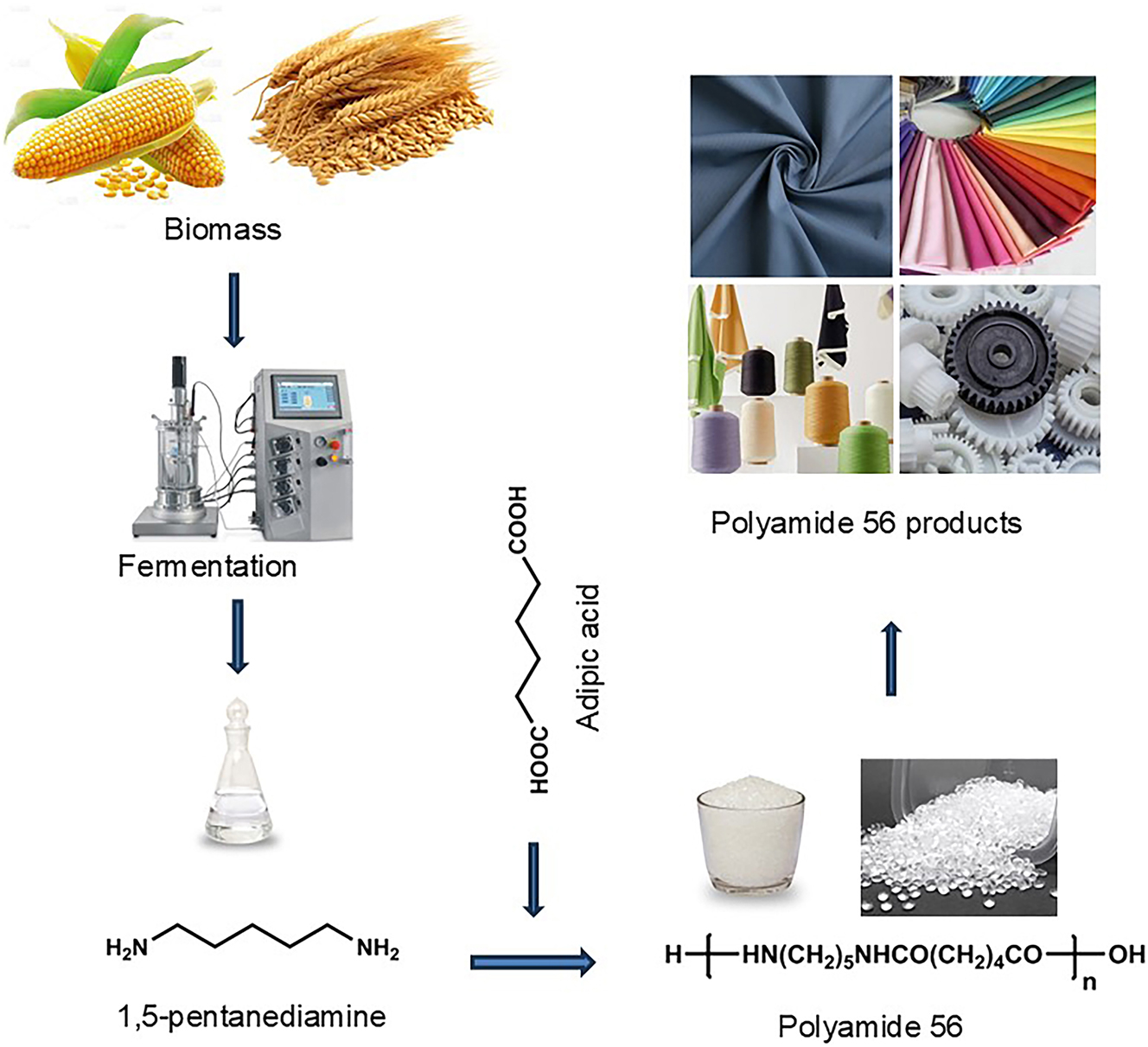
Bio-based PA56 material synthesis diagram.
3 PA56 modification methods
The modification of PA56 resin can be categorized into blending modification, copolymerization modification, and post-processing modification, based on the stage at which the modifier is introduced. Additionally, PA56 modification methods can be classified according to the functions they achieve, further dividing them into reinforcement, flame retardancy, toughening, antibacterial treatments, and other approaches. This paper reviews and discusses the progress of these various modification methods.
3.1 PA56 reinforcement modification
Fiber reinforcement is a highly effective strategy for improving the mechanical and thermal properties of plastics, including PA56. Reinforcing fibers are generally classified into two categories: one is inorganic fibers, such as glass fibers, carbon fibers, and asbestos, the other is organic fibers, including aramid, ultra-high molecular weight polyethylene (UHMWPE) fibers, and polyester fibers. Among these, glass fibers are particularly favored for their high elastic modulus, cost-effectiveness, and versatility.
PA56 can be reinforced with glass fibers using a twin-screw extruder, where PA56/glass fiber (GF) composite materials are produced through continuous fiber melt blending. A study by Zhao et al. 17 demonstrates that the incorporation of glass fibers into PA56 significantly improves its tensile strength, flexural strength, heat resistance, and impact resistance, with performance enhancements of up to 307.4 % compared to pure PA56 resin. Furthermore, glass fiber-reinforced PA56 exhibits low density and superior overall performance, making it an ideal material for demanding applications in industries such as transportation and wind energy.
This method not only broadens the range of applications for PA56 but also underscores the potential of fiber-reinforced composites as high-performance, lightweight, and durable materials, capable of meeting the demands of modern engineering challenges.
3.2 Flame retardant modification of PA56
The development of flame-retardant PA is primarily achieved through three technical approaches: blending, copolymerization, and post-finishing methods (Figure 2). In the blending method, an additive flame retardant is evenly mixed with PA resin in a mixer, followed by extrusion and pelletizing using a twin-screw extruder. 18 , 19 , 20 The copolymerization method employs reactive flame retardants as monomers during the polymerization process, allowing the reactive groups in the flame retardant structure to participate in the reaction. This introduces flame-retardant elements, such as nitrogen, phosphorus, or halogens, into the main or side chains of the polyamide, thereby imparting flame resistance to the polymer. 21 , 22 The post-finishing method involves applying surface treatment techniques, such as coating or grafting, during the textile processing stage to bestow flame-retardant properties to the fabric. 23 , 24

Flame retardant modification methods of polyamide.
3.2.1 Flame retardant PA56 blending modification method
The blending modification method is widely employed due to its simplicity, cost-effectiveness, and minimal process complexity. By merely adjusting the ratio of polymer to flame retardant, this approach allows for efficient and time-saving enhancements in material performance. 25 However, this approach typically requires the incorporation of a substantial amount of flame retardants. Additionally, the flame retardants introduced through physical blending are not strongly integrated with the polymer matrix. As a result, this blending system is prone to microphase separation between the flame retardant and the polymer matrix, leading to issues such as migration, precipitation, and degradation of the flame retardant. 26
Wang et al. 27 developed a novel flame retardant consisting of graphitic carbon nitride-modified melamine cyanurate acid (CNMAC), which was melt-mixed with PA56 using a rheological mixer. Then, PA56 with certain flame retardant properties was obtained through hot pressing and cooling. Test results demonstrated that the flame-retardant PA56 achieved a UL-94 V-0 rating, with a limiting oxygen index (LOI) value of 30.1 %. The introduction of CNMAC promoted the formation of a carbon layer on the surface of PA56, reducing the concentration of combustible gases in the air and enhancing thermal insulation. Zhang et al. 28 used ammonium polyphosphate to coat graphitic-phase carbon nitride to obtain a stable nitrogen-phosphorus flame retardant (CN@APP), which was melt-blended with PA56 and then hot-pressed. The results showed that the flame retardant system achieved UL-94 V-0 rating, with a LOI value of 31.7 %. Wang et al. 29 prepared a core–shell melamine cyanuric flame retardant (BP-MCA) through hydrogen bond self-assembly and solvent exchange strategies. PA56/BP-MCA composites were then prepared by melt blending in a twin-screw extruder. The results revealed that the introduction of 16 wt% BP-MCA enabled PA56 to achieve a UL-94 V-0 rating and a LOI value of 32.5 %. Notably, PA56/BP-MCA composites exhibited excellent mechanical properties, owing to the large aspect ratio, good dispersibility, and compatibility of the BP-MCA.
3.2.2 Flame-retardant PA56 copolymerization modification method
The copolymerization method chemically incorporates flame retardants into the molecular chain of PA56, ensuring that the flame retardant is strongly bonded and resistant to precipitation. This method requires only a small amount of flame retardant, yet delivers excellent flame retardancy with high efficiency. However, the copolymer produced by this method may exhibit a broad molecular weight distribution. Furthermore, this technology is quite complex, with the critical challenge being overcoming the technical bottleneck that prevents the transition from random copolymerization of reactive flame retardants and polyamide molecular chains to block copolymerization. Therefore, most flame-retardant polyamide 56 synthesized via copolymerization remains in the experimental research phase and has not yet been commercialized.
In Zhao’s study, flame-retardant polyamide 56(FRPA56) was synthesized through one-step copolymerization using a phosphorus-based flame retardant, adipic acid, and pentamethylenediamine as raw materials. 30 The resulting FRPA56 exhibited excellent flame retardancy, achieving a LOI of 28.4 % and a vertical burning rating of V-0 when the flame retardant content was 6 wt%. Additionally, the copolymerization process preserved the mechanical properties and thermal stability of PA56, with FRPA56 demonstrating a tensile strength of 72 MPa, a flexural strength of 115 MPa, and a melting point of 248 °C. Yang’s team 31 , 32 explored the use of two phosphorus-based flame retardant monomers, 9,10-dihydro-10-[2,3-di (hydroxycarbonyl)propyl]-10-phosphaphenanthrene-10-oxide (DDP) and 2-carboxyethyl (phenyl) phosphinic acid (CEPPA), to successfully prepare flame-retardant PA56 copolymers through melt copolymerization (Figure 3). The results indicated that with the addition of only 1.0 mol% DDP, the bio-based PA56 achieved a V-0 rating in the UL-94 vertical burning test, while PA56 containing 4.6 mol% CEPPA also achieved a V-0 rating in the same test.
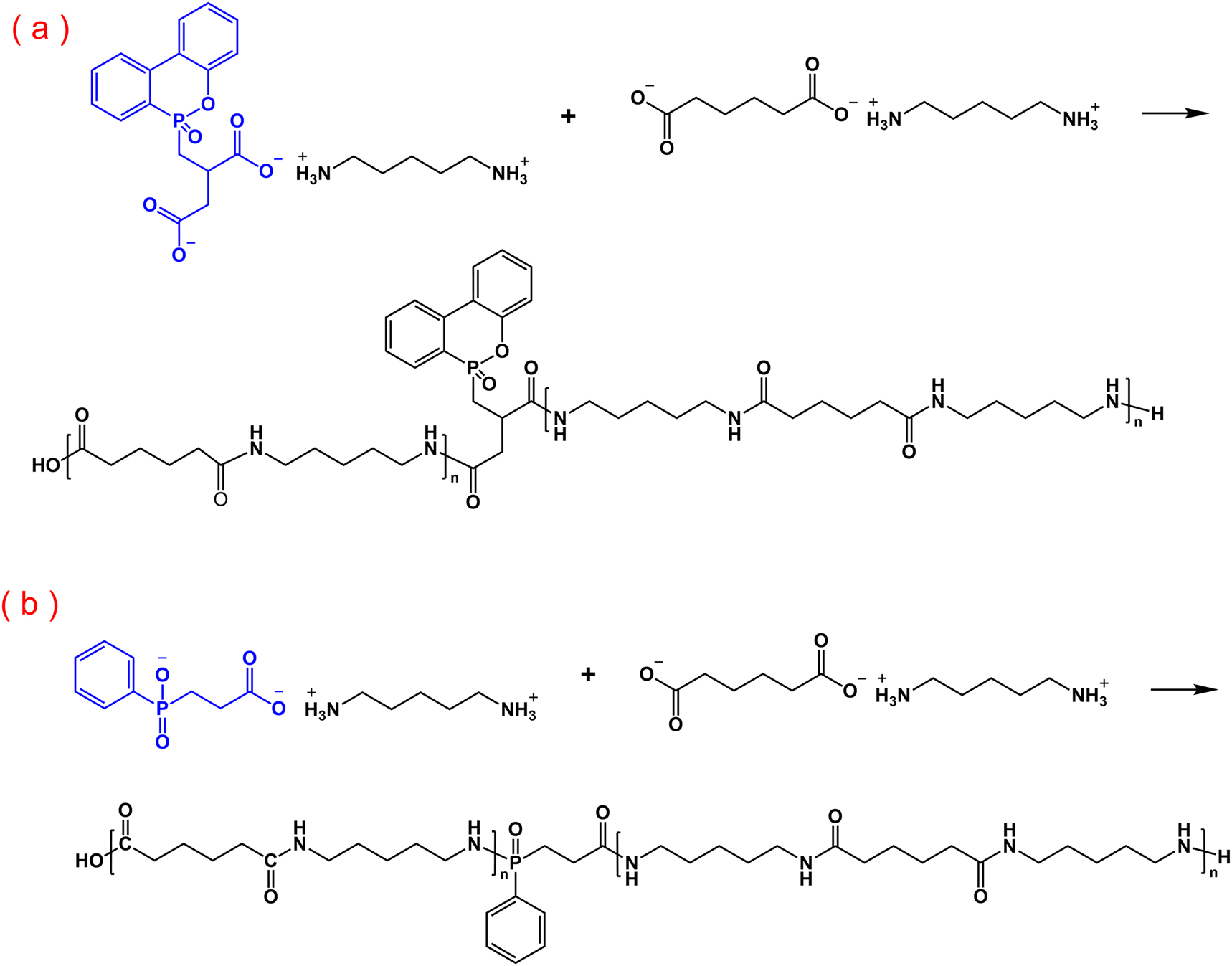
Reaction equation for polymerization of PA56 with flame retardants DDP (a); and CEPPA (b).
3.2.3 Flame-retardant PA56 post-finishing method
The post-finishing method typically involves applying a flame retardant to the surface of PA56 or introducing a flame retardant coating onto the fiber surface. This approach offers several advantages, including a short processing time, simplicity of operation, wide applicability, high usage frequency, and minimal impact on the material’s primary properties. However, the durability and flame retardant efficacy of this method are often inferior to those achieved through blending or copolymerization modification. In the case of PA56, Cui et al. 33 successfully coated polyethyleneimine, sodium tripolyphosphate, and melamine onto PA56 fabric using a simple three-step process. These compounds were cross-linked via weak interactions to form a three-dimensional, cross-linked network flame retardant coating, with melamine on the surface. The treated PA56 fabric exhibited no melt dripping during the vertical burning test. Similarly, Jin et al. 34 used 9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide (DOPO) and allylthiourea (ATU) to synthesize a green and sustainable multifunctional coating (DOPO-ATU), which was then applied to modified PA56 fabrics using a simple surface coating technique. The results demonstrated a significant improvement in flame retardancy and fire safety, with a LOI value of 30.1 %, no molten drops, and a damage length as low as 11.1 cm.
3.3 Toughening modification of PA56
The toughness of thermoplastic polymers can typically be improved through the incorporation of rigid materials, rubber, or thermoplastic elastomers (Figure 4), such as polyolefin elastomer (POE), ethylene propylene rubber (EPR), and ethylene-propylene-diene monomer (ERDM), which have been shown to significantly enhance the material’s toughness. 35 , 36 The primary preparation methods for high-toughness nylon include blending extrusion method and copolymerization.
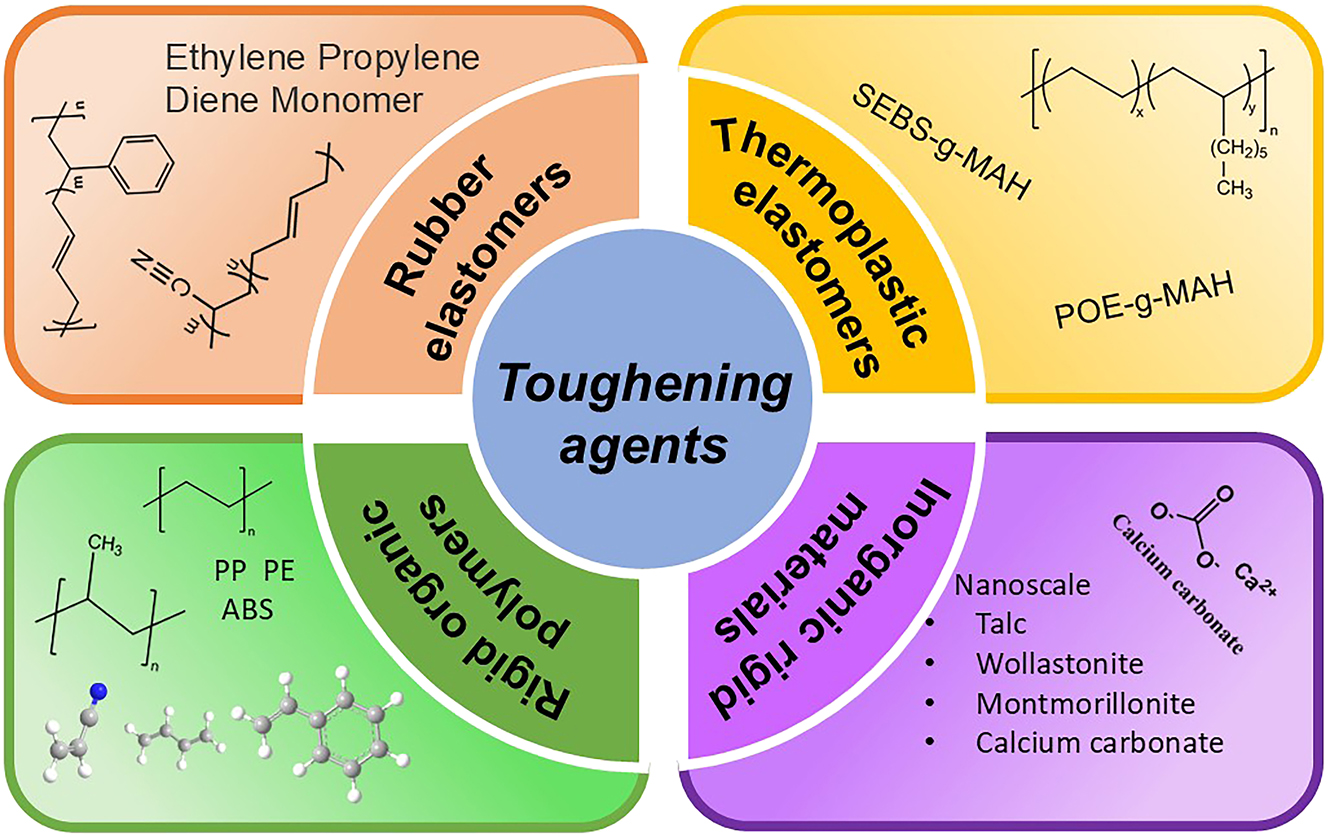
Four major types of toughening agents.
3.3.1 Blending extrusion method
Blending extrusion method is a widely used technique for toughening polyamide, primarily due to its simplicity and ease of operation. In this process, polyamide is used as the base resin, while elastomers, thermoplastic elastomers, or other high-toughness materials are employed as toughening agents. Under melt blending conditions, these components are effectively compounded, resulting in a material with significantly enhanced impact resistance and low-temperature performance. Consequently, this toughening technique expands the potential applications of PA56. According to toughening-related theories, morphological blending characteristics such as particle size and dispersed phase concentration have a notable impact on the final mechanical behavior. A finely dispersed phase at the submicron-level is typically a prerequisite for obtaining the desired notch impact response. In the case of rubber, for example, it is widely accepted that the impact strength of rubber-toughened polyamide is improved when the rubber particle size is between 0.2 and 1 μm. This is because appropriately sized phases create more interfaces and potential sites of stress concentration, which trigger mechanisms such as silver streaks and shear yielding, which help to dissipate energy and prevent crack propagation, thereby enhancing the material’s toughness. 37 However, particles with excessively large particle sizes are more likely to form defects within the polymer matrix, leading to brittle cracks and damaging the toughness of the material, while particles that are too small do not effectively absorb energy and terminate silver streaks. 38 Furthermore, the uniformity of phase distribution is crucial for toughness. A soft phase that is well-dispersed and uniformly distributed throughout the hard matrix to form a uniform “sea island structure” can provide a more effective toughening effect. In contrast, poor dispersion of the phase may lead to localized weaknesses and a decrease in toughness. Furthermore, the strength of the interface bonding between phases is also important. Strong interfacial bonding can effectively transfer stress between phases and prevent crack initiation and propagation, while weak interfacial bonding can lead to phase separation and reduced toughness.
Zhang et al. 39 used poly (octene-co-ethylene) grafted glycidyl methacrylate (POE-g-GMA) and melt-blended it with PA56 using a twin-screw extruder. The results revealed that notched impact strength of the toughened PA56 increased by 17 times. Chong et al. 40 employed a physical co-blending modification method to prepare a bio-based PA56 alloy, toughened with maleic anhydride (MAH) and glycidyl methacrylate (GMA) dual-monomer-grafted POE (POE-g-MAH/GMA), also using a twin-screw extruder. The results indicated that the notched impact strength of the alloy was 31 times and 29 times that of pure PA56 at 23 °C and −30 °C, respectively. Xu et al. 41 melt-blended maleic anhydride-grafted ethylene propylene diene monomer (EPDM-g-MAH) with bio-based PA56 in a twin-screw extruder to obtain polyamide elastomers with different formulations. The test results indicated that the composite material with a 10 % EPDM-g-MAH mass fraction exhibited the best impact resistance and thermal properties within the 0–20 % EPDM-g-MAH range. Fracture morphology analysis showed that the interface between the rubber phase and the polyamide phase was well fused, and the elastomer exhibited ductile fracture behavior. Wang et al. 10 employed maleic anhydride (MAH) as a compatibilizer to graft onto POE (POE-g-MAH), thereby improving its compatibility with bio-based PA56. Subsequently, POE-g-MAH was blended with bio-based PA56 to enhance its toughness. Mechanical strength tests indicated that the optimal toughening effect was achieved at a POE-g-MAH content of 20 %. Following toughening, the resulting super-tough nylon exhibited a significant improvement in toughness, with a notched impact strength of 88 kJ/m2 – an increase of 1,700 %. This performance meets the stringent requirements for industrial applications of super-tough polyamide.
3.3.2 Copolymerization method
The toughening of polyamide through copolymerization involves two primary technical routes, one of which is copolymerization with other polyamide varieties, such as PA6. Yang et al. 42 synthesized polyamide 56/6 copolymers using bio-based 1,5-pentanediamine, adipic acid, and caprolactam as the main raw materials through the copolymerization method. The tests showed that the tensile strength of the copolymer was similar to that of pure PA56 and PA6, but its toughness was enhanced, with the elongation at break increasing up to five times that of PA56. This improvement is likely due to the incorporation of PA6, which disrupts the molecular chain regularity of PA56, increases the disorder within the molecular chains, and reduces the hydrogen bonding density and crystallinity of PA56. However, this incorporation significantly enhances the elongation at break of PA56, thereby improving its flexibility and impact resistance. Lin et al. 43 synthesized a PA56/PA66 copolymer with an elongation at break of up to 758.1 % using pentamethyleneammonium adipate (PMA) salt and hexamethyleneammonium adipate (HMA) salt as raw materials through a one-pot polycondensation reaction.
Another promising method for toughening polyamide is the copolymerization of flexible segments into the polyamide matrix to prepare thermoplastic polyamide elastomers (TPAEs). These elastomers consist of hard and soft segments, where the hard segments include PA6, PA1010, PA1012, PA11, PA1212, etc., while the soft segments refer to flexible or low glass transition temperature chains, typically made from polyether diols such as polyethylene glycol (PEG), polypropylene glycol (PPG), and polytetramethylene glycol (PTMG). Compared to pure PA, the resulting copolymers exhibit improved toughness. 44 , 45 , 46 This is because the flexible chain segments act as plasticizers, enhancing the polymer matrix’s ability to deform plastically under stress and reducing brittle fracture. Prepared high-performance polyamide elastomer can be formed into a low-density, highly abrasion-resistant foam material suitable for high-end footwear applications after foaming treatment. Additionally, it can be processed into products for medical balloon catheter applications through extrusion-blow molding. However, the methods for synthesizing TPAEs are often complex and tend to be limited by reaction conditions such as high temperatures, long reaction times under high vacuum and special catalysts. Furthermore, achieving a strict stoichiometric balance between the –COOH and –OH groups is crucial in TPAEs; any imbalance can complicate the control of molecular structure and molecular weight during the preparation process. 47
Unfortunately, little research has been done on PA56 elastomers, which may be attributed to the complexity of the synthesis method and limited by processes such as reaction temperature, high vacuum for reaction time, and preparation techniques such as special catalysts. However, this may also provide a new research direction for researchers.
3.4 Antibacterial modification of PA56
Antibacterial fibers are specialized functional fibers developed to protect the human body and the environment from bacteria and microorganisms. In recent years, researchers have focused on incorporating substances capable of killing bacteria or inhibiting bacterial reproduction to prevent microbial growth and combat infections. Wang et al. 48 utilized ethylene-methyl acrylate-glycidyl methacrylate terpolymer (EMA) as a compatibilizer and polypentamethylene guanidine sulfate (PPGS) as an antibacterial agent to prepare antibacterial bio-based PA56 fibers through high-speed spinning. The results indicated that, compared to pure PA56 fibers, the elongation at break of the sample containing 2.0 wt% EMA and PPGS increased by 25.93 %, while the inhibition rate of the 1.0 wt% PPGS fiber against Escherichia coli and Staphylococcus aureus exceeded 99.99 %. However, this study also found that dyeing could affect the antibacterial properties of the fibers. Yang et al. 49 investigated the antibacterial properties of bio-polyamide nanofibers. PA56 nanofiber membranes were initially prepared using electrospinning technology and subsequently modified with chitosan (CS) to form PA56-CS membranes. These membranes were further enhanced with reactive dyes (reactive green 19, RG19, and reactive red 141, RR141) to produce PA56-CS-dye membranes. Poly (hexamethylene biguanide) (PHMB), a positively charged polymer, was then conjugated to these membranes to create PA56-CS-dye-PHMB nanofibrous membranes. Comprehensive physical characterization was performed using Fourier transform infrared spectroscopy (FTIR), scanning electron microscopy (SEM), and thermogravimetric analysis (TGA), while both qualitative and quantitative antibacterial analysis against E. coli was conducted. The PA56-CS-RR141-PHMB nanofibrous membranes exhibited excellent antibacterial efficiency (AE) of more than 97 %. Wen et al. 50 prepared highly efficient antibacterial nanofibrous membranes using bio-based PA56. PA56 nanofibers were chemically modified by sequential coupling with alginate (AG) and chitosan (CS) to form PA56-AG and PA56-AG-CS nanofibers. Further modification with poly (hexamethylene biguanide) (PHMB) resulted in PA56-AG-PHMB and PA56-AG-CS-PHMB antibacterial nanofiber membranes. The results demonstrated that the PA56-based membranes exhibited near 100 % antibacterial efficiency within a short period. Moreover, the PA56-AG-PHMB membrane exhibited a faster antibacterial rate and higher efficiency during repeated use compared to the PA56-AG-CS-PHMB membrane.
4 Conclusions and perspectives
The modification of bio-based polyamide 56 is of significant importance for expanding its application potential and developing a diverse range of products. As modern industries evolve, the performance requirements for PA56 are becoming increasingly stringent, necessitating innovative and targeted modification techniques.
For reinforcement modification, the primary objective is to enhance the mechanical properties of PA56, such as tensile and flexural strength, while minimizing any negative effects on toughness and melt flowability. In flame-retardant modification, the key focus is achieving halogen-free flame retardancy. Simultaneously, efforts are directed toward improving the material’s anti-melting droplet performance to ensure both robust flame-retardant properties and thermal stability. Toughening modification aims to enhance the low-temperature impact resistance and overall toughness of PA56 without significantly compromising its rigidity and hardness, thus balancing flexibility and structural integrity. For antibacterial modification, the compatibility of antibacterial agents with the PA56 matrix is critical. Further research and development of novel antibacterial modification technologies, such as the incorporation of nano-antibacterial agents or bio-based functional coatings, is essential to meet evolving application requirements.
With the growth of the bio-based PA56 industry, the demand for functionalized and differentiated products is expected to rise significantly. Future research will likely focus on advanced modification strategies in areas such as super-toughening, halogen-free flame retardancy, high-temperature resistance, enhanced strength, and multifunctional integration. These advancements are anticipated to drive the development of next-generation PA56 materials, enabling broader applications in industries such as automotive, electronics, textiles, and medical devices, while strengthening PA56’s role in promoting green and sustainable development.
Acknowledgments
The authors would like to acknowledge Dianzi Zhang and Benbin Li for inspiring the authors’ research on nylon modification.
-
Research ethics: Not applicable.
-
Informed consent: Not applicable.
-
Author contributions: All authors have accepted responsibility for the entire content of this manuscript and approved its submission.
-
Use of Large Language Models, AI and Machine Learning Tools: None declared.
-
Conflict of interest: The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
-
Research funding: None declared.
-
Data availability: None declared.
References
1. Candau, N.; Galland, S.; Cretenoud, J.; Balog, S.; Michaud, V.; Chenal, J.-M.; Lame, O.; Plummer, C. J. G.; Frauenrath, H. High-performance Polyamides with Engineered Disorder. Poly. Chem. 2021, 12 (44), 6426–6435. https://doi.org/10.1039/D1PY01225H.Search in Google Scholar
2. Chen, H.; Tang, M.; Yang, X.; Tsang, Y. F.; Wu, Y.; Wang, D.; Zhou, Y. Polyamide 6 Microplastics Facilitate Methane Production during Anaerobic Digestion of Waste Activated Sludge. Chem. Eng. J. 2021, 408, 127251. https://doi.org/10.1016/j.cej.2020.127251.Search in Google Scholar
3. Venoor, V.; Park, J. H.; Kazmer, D. O.; Sobkowicz, M. J. Understanding the Effect of Water in Polyamides: A Review. Poly. Chem. 2021, 61 (3), 598–645. https://doi.org/10.1080/15583724.2020.1855196.Search in Google Scholar
4. Froidevaux, V.; Negrell, C.; Caillol, S.; Pascault, J.-P.; Boutevin, B. Biobased Amines: From Synthesis to Polymers; Present and Future. Chem. Rev. 2016, 116 (22), 14181–14224. https://doi.org/10.1021/acs.chemrev.6b00486.Search in Google Scholar PubMed
5. Danso, D.; Chow, J.; Streit Wolfgang, R. Plastics: Environmental and Biotechnological Perspectives on Microbial Degradation. Appl. Environ. Microbiol. 2019, 85 (19), e01095–e01019. https://doi.org/10.1128/AEM.01095-19.Search in Google Scholar PubMed PubMed Central
6. Wang, Z.; Ganewatta, M. S.; Tang, C. Sustainable Polymers from Biomass: Bridging Chemistry with Materials and Processing. Prog. Polym. Sci. 2020, 101, 101197. https://doi.org/10.1016/j.progpolymsci.2019.101197.Search in Google Scholar
7. Xue, Y. J.; Zhao, Y. L.; Ji, X. L.; Yao, J. H.; Busk, P. K.; Lange, L.; Huang, Y. H.; Zhang, S. J. Advances in Bio-Nylon 5X: Discovery of New Lysine Decarboxylases for the High-Level Production of Cadaverine. Green Chem. 2020, 22 (24), 8656–8668. https://doi.org/10.1039/d0gc03100c.Search in Google Scholar
8. Yang, H.; Zhang, X.; Liu, Y.; Mao, L.; He, S.; Huang, M.; Hao, L.; Xu, W.; Liu, W. Synthesis and Comprehensive Characterization of Bio-Based Polyamide 56/6 Copolymer: Mechanical, Thermal, and Processing Properties. Eur. Polym. J. 2024, 202. https://doi.org/10.1016/j.eurpolymj.2023.112593.Search in Google Scholar
9. Kang, H.; Wang, Z.; Hao, X.; Liu, R. Thermal Induced Crystalline Transition of Bio-Based Polyamide 56. Polymer 2022, 242, 124540. https://doi.org/10.1016/j.polymer.2022.124540.Search in Google Scholar
10. Wang, Y.; Zhang, Y.; Xu, Y.; Liu, X.; Guo, W. Research on Compatibility and Surface of High Impact Bio-Based Polyamide. High Perform. Polym. 2021, 33 (8), 960–968. https://doi.org/10.1177/09540083211005511.Search in Google Scholar
11. Zhang, S.; Ma, J. Tailored Process for Spinning Fine Denier Bio-Based Polyamide 56 Fibers. J. Biobased Mater. Bioenergy 2019, 13 (1), 102–108. https://doi.org/10.1166/jbmb.2019.1822.Search in Google Scholar
12. Morales-Gámez, L.; Soto, D.; Franco, L.; Puiggalí, J. Brill Transition and Melt Crystallization of Nylon 56: An Odd–Even Polyamide with Two Hydrogen-Bonding Directions. Polymer 2010, 51 (24), 5788–5798. https://doi.org/10.1016/j.polymer.2010.09.074.Search in Google Scholar
13. Yang, T.; Gao, Y.; Wang, X.; Ma, B.; He, Y. Hydrogen Bonding and Crystalline Structure of Bio-Based PA56. Polymer 2021, 237, 124356. https://doi.org/10.1016/j.polymer.2021.124356.Search in Google Scholar
14. Zhao, S.; Gong, S.; Zhao, B.; Hou, L.; Zhang, L.; Hu, Q.; Pan, K. Mechanism Study of the Polymerization of Polyamide 56: Reaction Kinetics and Process Parameters. Macromol. Rapid Commun. 2023, 44 (24), e2300371. https://doi.org/10.1002/marc.202300371.Search in Google Scholar PubMed
15. Kim, H. T.; Baritugo, K. A.; Oh, Y. H.; Kang, K. H.; Jung, Y. J.; Jang, S.; Song, B. K.; Kim, I. K.; Lee, M. O.; Hwang, Y. T.; Park, K.; Park, S. J.; Joo, J. C. High-Level Conversion of L-Lysine into Cadaverine by Escherichia coli Whole Cell Biocatalyst Expressing Hafnia Alvei L-Lysine Decarboxylase. Polymers 2019, 11 (7). https://doi.org/10.3390/polym11071184.Search in Google Scholar PubMed PubMed Central
16. Yang, P.; Peng, X.; Wang, S.; Li, D.; Li, M.; Jiao, P.; Zhuang, W.; Wu, J.; Wen, Q.; Ying, H. Crystal Structure, Thermodynamics, and Crystallization of Bio-Based Polyamide 56 Salt. CrystEngComm 2020, 22 (18), 3234–3241. https://doi.org/10.1039/D0CE00198H.Search in Google Scholar
17. Zhao, Y.; Wang, L.; Chong, Y.; Chen, Y.; Zheng, R.; Zhang, L. Preparation and Properties of Glass Fiber Reinforced Nylon56 Composites. Polym. Compos. 2022, 43 (9), 6660–6666. https://doi.org/10.1002/pc.26982.Search in Google Scholar
18. Qiu, X.; Li, Z.; Li, X.; Zhang, Z. Flame Retardant Coatings Prepared Using Layer by Layer Assembly: A Review. Chem. Eng. J. 2018, 334, 108–122. https://doi.org/10.1016/j.cej.2017.09.194.Search in Google Scholar
19. He, W.; Song, P.; Yu, B.; Fang, Z.; Wang, H. Flame Retardant Polymeric Nanocomposites through the Combination of Nanomaterials and Conventional Flame Retardants. Prog. Mater. Sci. 2020, 114, 100687. https://doi.org/10.1016/j.pmatsci.2020.100687.Search in Google Scholar
20. Guo, S.; Xu, J.; Ni, X. Synthesis, Structures, and Properties of a New Pentaerythritol-Derived Flame Retardant Used in Polyamide 66. ACS Omega 2021, 6 (19), 12887–12897. https://doi.org/10.1021/acsomega.1c01385.Search in Google Scholar PubMed PubMed Central
21. Zhang, J.; Lian, S.; He, Y.; Cao, X.; Shang, J.; Liu, Q.; Ye, G.; Zheng, K.; Ma, Y. Intrinsically Flame-Retardant Polyamide 66 with High Flame Retardancy and Mechanical Properties. RSC Adv. 2021, 11 (1), 433–441. https://doi.org/10.1039/d0ra07822k.Search in Google Scholar PubMed PubMed Central
22. Čolović, M.; Vasiljević, J.; Štirn, Ž.; Čelan Korošin, N.; Šobak, M.; Simončič, B.; Demšar, A.; Malucelli, G.; Jerman, I. New Sustainable Flame Retardant DOPO-NH-functionalized Polyamide 6 and Filament Yarn. Chem. Eng. J. 2021, 426, 130760. https://doi.org/10.1016/j.cej.2021.130760.Search in Google Scholar
23. Mourgas, G.; Giebel, E.; Schneck, T.; Unold, J.; Buchmeiser, M. R. Syntheses of Intrinsically Flame-Retardant Polyamide 6 Fibers and Fabrics. J. Appl. Polym. Sci. 2019, 136 (31), 47829. https://doi.org/10.1002/app.47829.Search in Google Scholar
24. Liu, W.; Shi, R.; Ge, X.; Huang, H.; Chen, X.; Mu, M. A Bio-Based Flame Retardant Coating Used for Polyamide 66 Fabric. Prog. Org. Coat. 2021, 156, 106271. https://doi.org/10.1016/j.porgcoat.2021.106271.Search in Google Scholar
25. Xiong, L.; Qing, B.; Liu, X. Bio-based Flame Retardant Polyamide 56 and its Preparation Method. CN 201710879316.6, 2017.Search in Google Scholar
26. Chen, X.; Xu, D.; Zhang, H.; Feng, X.; Deng, J.; Pan, K. In Situ Polymerization of Flame Retardant Modification Polyamide 6,6 with 2-carboxy Ethyl (Phenyl) Phosphinic Acid. J. Appl. Polym. Sci. 2020, 137 (20), 48687. https://doi.org/10.1002/app.48687.Search in Google Scholar
27. Wang, Z.; Wang, J.; Zhang, J.; Guo, W. Synergistic Flame Retardant Modification of Bio-Based Nylon 56 by Graphitic Carbon Nitride and Melamine Cyanurate. J. Vinyl Addit. Technol. 2024, 30 (2), 456–469. https://doi.org/10.1002/vnl.22060.Search in Google Scholar
28. Zhang, Y.; Zhao, Y.; Wang, Z.; Jiang, X.; Liu, X.; Guo, W. Synergistic Effect of Graphitic Carbon Nitride and Ammonium Polyphosphate on the Enhanced Flame Retardant and Smoke Suppression Properties of Biobased Polyamide 56. J. Vinyl Addit. Technol. 2022, 28 (3), 615–630. https://doi.org/10.1002/vnl.21912.Search in Google Scholar
29. Wang, W.; Zhu, L.; Liu, Y.; Wang, Q. Preparation of Core-Shell Melamine Cyanuric Based Flame Retardant for Improving Comprehensive Performance of Polyamide 56. Polym. Degrad. Stab. 2024, 228, 110933. https://doi.org/10.1016/j.polymdegradstab.2024.110933.Search in Google Scholar
30. Zhao, S.; Chen, X.; Zhou, Y.; Zhao, B.; Hu, Q.; Chen, S.; Pan, K. Molecular Design of Reactive Flame Retardant for Preparing Biobased Flame Retardant Polyamide 56. Polym. Degrad. Stab. 2023, 207, 110212. https://doi.org/10.1016/j.polymdegradstab.2022.110212.Search in Google Scholar
31. Yang, T.; Gao, Y.; Liu, X.; Wang, X.; Yu, J.; Huang, S.; Ma, B.; He, Y. A Strategy to Achieve the Inherently Flame-Retardant PA56 by Copolymerization with DDP. J. Polym. Environ. 2022, 30 (9), 3802–3814. https://doi.org/10.1007/s10924-022-02466-6.Search in Google Scholar
32. Yang, T.; Gao, Y.; Liu, X.; Wang, X.; Ma, B.; He, Y. Flame-retardant Polyamide 56 with High Fire Safety and Good Thermal Performance. Polym. Adv. Technol. 2022, 33 (9), 2807–2819. https://doi.org/10.1002/pat.5734.Search in Google Scholar
33. Cui, Y.; Liu, Y.; Gu, D.; Zhu, H.; Wang, M.; Dong, M.; Guo, Y.; Sun, H.; Hao, J.; Hao, X. Three-Dimensional Cross-Linking Network Coating for the Flame Retardant of Bio-Based Polyamide 56 Fabric by Weak Bonds. Polymers 2024, 16 (8), 1044. https://doi.org/10.3390/polym16081044.Search in Google Scholar PubMed PubMed Central
34. Jin, W.-J.; Dong, S.; Guan, J.-P.; Cheng, X.-W.; Qin, C.-X.; Chen, G.-Q. Multifunctional and Sustainable DOPO-Derivative Coating for Flame-Retardant, Antibacterial and UV-Protective of Polyamide 56 Protective Biomaterials. Surf. Interfaces 2023, 42, 103513. https://doi.org/10.1016/j.surfin.2023.103513.Search in Google Scholar
35. Xu, M.; Lu, J.; Qiao, Y.; Wei, L.; Liu, T.; Lee, P. C.; Zhao, L.; Park, C. B. Toughening Mechanism of Long Chain Branched Polyamide 6. Mater. Des. 2020, 196, 109173. https://doi.org/10.1016/j.matdes.2020.109173.Search in Google Scholar
36. Wang, B.; Yang, Y.; Guo, W. Effect of EVOH on the Morphology, Mechanical and Barrier Properties of PA6/POE-g-MAH/EVOH Ternary Blends. Mater. Des. 2012, 40, 185–189. https://doi.org/10.1016/j.matdes.2012.03.017.Search in Google Scholar
37. Gallego, R.; García-López, D.; López-Quintana, S.; Gobernado-Mitre, I.; Merino, J. C.; Pastor, J. M. Toughening of PA6/mEPDM Blends by Two Methods of Compounding, Extruder and Internal Mixer: Rheological, Morphological and Mechanical Characterization. Polym. Bull. 2008, 60 (5), 665–675. https://doi.org/10.1007/s00289-008-0902-1.Search in Google Scholar
38. Xu, M. H.; Qiu, W.; Qiu, G. X. Ethylene-Propylene Elastomer Grafted Maleic Anhydride Toughened Polyamide-6 Morphology and Properties. J. Macromol. Sci. Part B-Phys. 2013, 52 (1), 155–166. https://doi.org/10.1080/00222348.2012.695644.Search in Google Scholar
39. Zhang, Y.; Wang, Y.; Xu, Y.; Liu, X.; Guo, W. Modification of Biobased Polyamide 56 to Achieve Ultra-toughening. Polym.-Plast. Technol. Mater. 2021, 60 (14), 1585–1604. https://doi.org/10.1080/25740881.2021.1924198.Search in Google Scholar
40. Chong, Y.; Zhuang, X.; Guan, C.; Wang, L.; Zheng, R.; Bie, Z. Preparation and Performance Study of POE-G-MAH/GMA Toughened Bio-Based PA56 Alloys. J. Alloys Compd. 2023, 960, 170813. https://doi.org/10.1016/j.jallcom.2023.170813.Search in Google Scholar
41. Xu, Y.; Zhao, C.; Guo, Z.; Dong, W.; Liu, X.; Guo, W. EPDM-g-MAH Toughened Bio-Based Polyamide 56 to Prepare Thermoplastic Polyamide Elastomer and the Performance Characterization. J. Appl. Polym. Sci. 2022, 139 (24), 52346. https://doi.org/10.1002/app.52346.Search in Google Scholar
42. Yang, H.; Zhang, X.; Liu, Yi.; Mao, L.; He, S.; Huang, M.; Hao, L.; Xu, W.; Liu, W. Synthesis and Comprehensive Characterization of Bio-Based Polyamide 56/6 Copolymer: Mechanical, Thermal, and Processing Properties. Eur. Polym. J. 2024, 202, 112593; https://doi.org/10.1016/j.eurpolymj.2023.112593.Search in Google Scholar
43. Lin, N.; Wang, Z.; Kang, H.; Hao, X.; Liu, R. Synthesis and Characterizations of Random Copolyamide PA(56-co-66): Excellent Toughness and Transparency. Polymer 2024, 307, 127271. https://doi.org/10.1016/j.polymer.2024.127271.Search in Google Scholar
44. Jiang, J.; Tang, Q.; Pan, X.; Xi, Z.; Zhao, L.; Yuan, W. Structure and Morphology of Thermoplastic Polyamide Elastomer Based on Long-Chain Polyamide 1212 and Renewable Poly(trimethylene Glycol). Ind. Eng. Chem. Res. 2020, 59 (39), 17502–17512. https://doi.org/10.1021/acs.iecr.0c01334.Search in Google Scholar
45. Tong, X.; Peng, W.-M.; Zhang, M.-L.; Wang, X.-J.; Zhang, G.; Long, S.-R.; Yang, J. A New Class of Poly(ether-Block-Amide)s Based on Semi-aromatic Polyamide: Synthesis, Characterization and Structure–Property Relations. Polym. Int. 2021, 70 (2), 230–241. https://doi.org/10.1002/pi.6119.Search in Google Scholar
46. Tang, J. X.; Gong, S.; Zhao, B.; Chen, K.; Deng, J. P.; Pan, K. Biobased Thermoplastic Polyamide Elastomers: Preparation, Properties, and Applications. Ind. Eng. Chem. Res. 2024, 63 (8), 3465–3482. https://doi.org/10.1021/acs.iecr.3c04554.Search in Google Scholar
47. Gong, S.; Zhao, S.; Chen, X.; Liu, H.; Deng, J.; Li, S.; Feng, X.; Li, Y.; Wu, X.; Pan, K. Thermoplastic Polyamide Elastomers: Synthesis, Structures/Properties, and Applications. Macromol. Mater. Eng. 2021, 306 (12), 2100568. https://doi.org/10.1002/mame.202100568.Search in Google Scholar
48. Wang, L.; Zhou, B.; Du, Y.; Bai, M.; Xu, X.; Guan, Y.; Liu, X. Guanidine Derivatives Leverage the Antibacterial Performance of Bio-Based Polyamide PA56 Fibres. Polymers 2024, 16 (19), 2707. https://doi.org/10.3390/polym16192707.Search in Google Scholar PubMed PubMed Central
49. Yang, J.-H.; Le, Q.-V.; Liu, B.-L.; Srinophakun, P.; Wang, C.-Y.; Chiu, C. Y.; Ng, I. S.; Chen, K.-H.; Chang, Y.-K. Enhanced Antimicrobial Performance of Single-Use Polyamide 56 Nanofiber Membranes Modified with Chitosan, Reactive Dyes, and Poly(hexamethylene Biguanide) (PHMB) for Biological Waste Treatments. Biochem. Eng. J. 2025, 213, 109538. https://doi.org/10.1016/j.bej.2024.109538.Search in Google Scholar
50. Wen, H.-Y.; Le, Q.-V.; Liu, B.-L.; Srinophakun, P.; Chiu, C.-Y.; Wang, C.-Y.; Ng, I. S.; Chen, K.-H.; Chang, Y.-K. Alginate and Chitosan-Based Polyamide 56 Modified Nanofiber Membrane for Highly Effective Capture of Escherichia coli: Antibacterial and Cytotoxicity Studies. Int. J. Biol. Macromol. 2024, 279, 135464. https://doi.org/10.1016/j.ijbiomac.2024.135464.Search in Google Scholar PubMed
© 2025 Walter de Gruyter GmbH, Berlin/Boston
Articles in the same Issue
- Frontmatter
- Material Properties
- Nonisothermal crystallization and compatibility performances of poly(d-lactide) and poly(l-lactide) blends modified with poly(butylene succinate)
- Preparation and Assembly
- Research on modification strategies of bio-based polyamide 56
- Cross-linked hydrophilic polymers: universal sensors for analyzing solutions containing polar organic compounds
- β-Cyclodextrin modified PSF bifunctional blood purification membrane for bilirubin removal
- Radiochromic film dosimeter based on povidone-iodine for gamma dosimetric applications
- Engineering and Processing
- Blade coating process using hyperbolic tangent model: a perturbation method
- Dynamic excellence in magnetorheological shear stiffening gel: fabrication and formulation of a diagonal and distributional particle chain model
Articles in the same Issue
- Frontmatter
- Material Properties
- Nonisothermal crystallization and compatibility performances of poly(d-lactide) and poly(l-lactide) blends modified with poly(butylene succinate)
- Preparation and Assembly
- Research on modification strategies of bio-based polyamide 56
- Cross-linked hydrophilic polymers: universal sensors for analyzing solutions containing polar organic compounds
- β-Cyclodextrin modified PSF bifunctional blood purification membrane for bilirubin removal
- Radiochromic film dosimeter based on povidone-iodine for gamma dosimetric applications
- Engineering and Processing
- Blade coating process using hyperbolic tangent model: a perturbation method
- Dynamic excellence in magnetorheological shear stiffening gel: fabrication and formulation of a diagonal and distributional particle chain model

