Abstract
In analytical chemistry literature, the term isobar is frequently used to describe small molecules with identical molecular formulas, often interchangeably used with isomer. This usage conflicts with the historical and International Union of Pure and Applied Chemistry (IUPAC) definition of isobar, which refers to nuclides of different elements with equal mass numbers but different proton-to-neutron ratios. In small-molecule LC-MS/MS analysis, this terminological overlap leads to confusion, particularly when distinguishing between compounds with identical exact masses (isomers) and those with different elemental compositions but nearly identical masses (molecular isobars). This article explores the origin and definitions of both terms and provides examples highlighting that the term isobar can be used for molecules but not for isomers, thereby emphasizing the importance of precise nomenclature for accurate molecular identification and interpretation in mass spectrometry.
Introduction
The term isobar has become increasingly used in analytical chemistry literature, particularly in the context of small-molecule mass spectrometry. In many cases, it is used interchangeably with the term isomer to describe compounds sharing the same molecular formula. 1 , 2 , 3 This can be observed in studies involving steroid assays, 4 hexose monosaccharides such as galactose, glucose, and mannose, 5 and more recently if the interference is less clear like fatty acid analyses involving different acyl compositions but identical molecular weights. 6
Historically, the term isomer was introduced in 1837 by J.J Berzelius, meaning “put together from the same pieces”. 7 Subsequently, Van’t Hoff introduced the theory of the three-dimensional structure and the idea of stereoisomers, thereby laying the foundation for the modern understanding of isomers. 8
Much later, in 1918, the term isobar was introduced by A. W. Stewart. 9 Derived from the Greek words isos (equal) and baros (weight), the term was originally used not to describe molecules, but to describe nuclides (nuclei of different chemical elements) that have the same total mass number (A), which is the sum of the protons and neutrons in the nucleus. At the time, the internal structure of the atomic nucleus was not yet fully understood. Two years later, in 1920, Ernest Rutherford proposed the existence of a positively charged subatomic particle, the proton and also speculated about the existence of a neutral particle of similar mass, which would eventually be discovered in 1932 by James Chadwick and called neutron. 10 In 1933, it became evident through the work of the Joliot-Curies and others that there is a slight mass difference between protons and neutrons, the neutron being slightly heavier. 11 As a result, the term isobar refers to nuclides of different elements that share the same mass number, meaning they have the same total number of nucleons, but differ slightly in exact atomic mass. This revised definition is, in essence, a practical convention rather than a reflection of true mass equality. Thus, the concept of isobars is not based on exact mass parity, but instead reflects a historically developed and functionally convenient framework centred around nominal mass values.
Although the term ‘isobar’ remains a useful classification in nuclear chemistry, it found its way into mass spectrometry as well, where its meaning and practical relevance, especially for small-molecule analysis, is less clear. While in textbooks related to analytical chemistry and/or mass spectrometry, the definition of isomers is clear, 12 , 13 the definition of isobars is not referenced, 12 or is constrained by the description of “same nominal mass (integral)”. 13
While the International Union of Pure and Applied Chemistry (IUPAC) still defines isobars as “different nuclides of equal mass number,”this definition originating from the Physical Chemistry division of IUPAC, and thus still limiting the term to elements. 14 In addition, Murray et al. published recommendations for terminology in mass spectrometry, including isobars as “Atomic or molecular species with the same nominal mass but different exact masses”. 15 This description has also been adopted by the Royal Society of Chemistry (RSC) Analytical Methods Committee. 16 Similarly, a recent graphical review regarding isotopes, isomers and isobars defined isobars as “same mass, different molecular formula”. 17
However two molecules may meet the definition of “molecules with the same nominal mass but different exact masses,” this does not inherently make them isobars, at least not when applying the term as used in nuclear chemistry. The fact remains that mass spectrometrists across different subfields whether in ICP-MS (geochemistry, elemental analysis) or ESI-MS (biochemistry, macromolecular studies) use the same terminology with distinct definitions. Given that mass spectrometry spans multiple disciplines, its terminology should ideally be self-consistent to avoid ambiguity.
Hence, the term isobar is often used to describe compounds with similar masses, particularly in the context of interfering peaks in a mass spectrum. However, the degree to which their exact masses correspond is far more determinative than merely sharing the same nominal mass. This distinction becomes even more pronounced as the molecular mass increases. Next, there are many practical examples of isomers in the literature, and when it comes to isomers, using this term provides the most clarity. Using the term ‘isobar’ for isomers ignores the essential reason why the term ‘isobars’ was introduced in the first place, namely, to name different elements with equal nucleons but different proton to neutron (P:N) ratios. When ‘isobars’ is used for molecules, it should refer to different molecular formulas with equal nucleons, different P:N ratios, and therefore similar masses. Moreover, practical examples of molecular isobars, which may or may not interfere with mass spectrometry, illustrate that an isobar cannot be an isomer and vice versa.
In practice for isomers and molecule isobars
For isomers, which have an identical molecular formula and consequently an equal P:N ratio but different structures, distinguishing between them in mass spectrometry can be challenging. Depending on their structural (dis)similarity, they can be separated chromatographically, as is often the case with steroids and the ratios of the MS fragments ions can vary greatly. 18
In contrast, with isomers with a very similar structure, such as C6 sugars, this can be very difficult, depending on the column used and also the MS fragments ions are very similar to each other. These characteristics are typical for isomers and overall the term isomer is generally used correctly when it concerns to these molecules with identical molecular formula.
Isomers are common in practice, but there are also practical examples of isobaric molecules. Isobaric molecules can be divided into two groups. In both cases, there is a difference in the molecular formula, but in the first group, the P:N ratio between the two molecules differs (like elements), and in the second group, the P:N ratio between the two is the equal.
Regarding to the first group, if an analyte or a matrix compound has the same number of nucleons as the target analyte, but differs in the P:N ratio, this will result in a chemically different compound and/or a difference in the functional group. A simplified example is shown in Fig. 1, where an aldehyde group has the same nominal mass as an ethyl group. In these cases, the term isobar is appropriate due to difference in P:N 15:14 for the aldehyde group and 17:12 for the ethyl group. However, a difference in the functional group almost always leads to a different interaction with the stationary phase of the analytical column and also to a different fragmentation pattern with the MS/MS compared to the target analyte.

Mass spectrum by the national institute of standard and technology (NIST) of butane, C4H10 with monoisotopic mass 58.078 and propanal, C3H6O with monoisotopic mass 58.042. 19
For the second group, there is a practical example that slightly differs from the isobar example above, where the authors correctly used the term ‘isobar’. 20 In this example, glucose-6-sulfate and glucose-6-phosphate differ in their functional groups (phosphate and sulfate), but the P:N ratio is identical for both molecules. However, there remains a small mass difference of approximately 0.009 Da, which arises from the difference in nuclear binding energy between the combination of 31P + 1H and 32S. In practice, the similarity results in insufficient interaction with the stationary phase of the column, causing the two analytes to co-elute. Furthermore, the fragmentation pattern is also similar for both analytes (Fig. 2), making this example very challenging to separate both chromatographically and by mass spectrometry. Unlike the first group, this example highlights the difficulties in separation between isobars.

Sulfated monosaccharide with monoisotopic mass 259.012, P:N 135:124 and phosphorylated monosaccharide with monoisotopic mass 259.022, P:N 135:124, figures adapted and simplified from Zang et al. 20
Conclusion
Clearly, it remains crucial to specify the distinction between molecules with an equal number of nucleons and to clarify whether it involves interference of the same molecular formula and exact mass (isomers) or interference with a different molecular formula and a possible difference P:N ratio (isobaric). If the term ‘isobar’ is exclusively employed for the latter purpose, similar to categorizing the difference between two elements with an equal sum of nucleons, only then does ‘isobar’ serve as a valuable addition to the nomenclature in analytical chemistry.
When the mass interference is unknown, it serves no purpose to substitute the notion of equal or similar weight with the Greek-derived term isobar. The term ‘isobar’ has a clear meaning in element chemistry, which is closely connected to analytical chemistry. Given the practical examples of molecules that can be explained as isobars of each other, as indicated by the provided examples, it is illogical to use ‘isobars’ interchangeably for ‘isomers’ in analytical chemistry, or for nominal mass interference in general. The diagram (Fig. 3) can be used for clarification of similar/equal weight molecules.
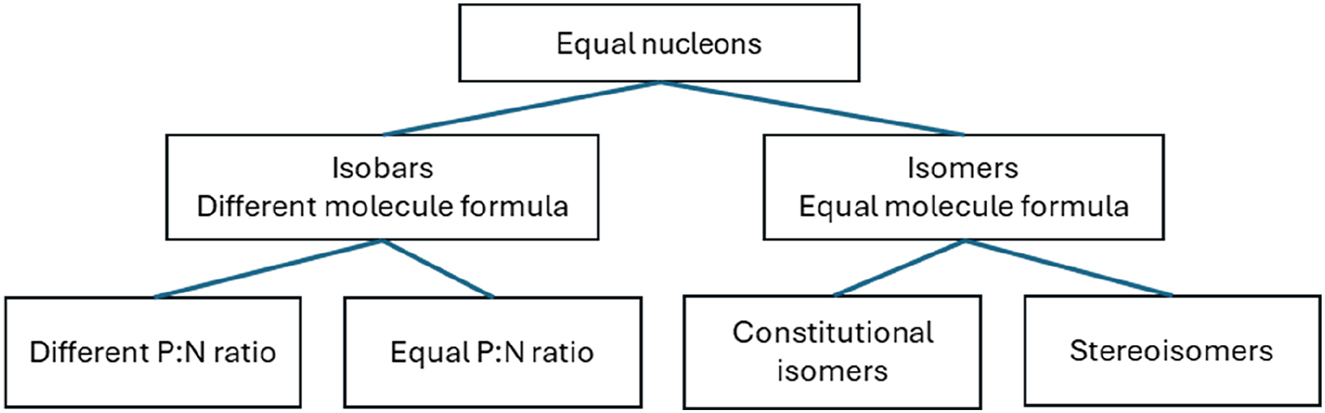
Schematic overview of different types for equal nucleons/similar weight molecules.
-
Research ethics: Not applicable.
-
Informed consent: Not applicable.
-
Author contributions: All authors have accepted responsibility for the entire content of this manuscript and approved its submission.
-
Use of Large Language Models, AI and Machine Learning Tools: During the preparation of this work the author(s) used Le Chat (Mistral AI) in order to improve the clarity and fluency of the text. After using this tool/service, the author(s) reviewed and edited the content as needed and take(s) full responsibility for the content of the publication.
-
Conflict of interest: The authors state no conflict of interest.
-
Research funding: None declared.
-
Data availability: Not applicable.
References
1. Volmer, D. A.; Stokes, C. S. Analytical Considerations for Accurately Capturing the Relevant Species Contributing to Vitamin D Status in Liquid chromatography-Tandem Mass Spectrometry Assays. Anal. Sci. Adv. 2022, 3, 14–20. https://doi.org/10.1002/ansa.202100057.Search in Google Scholar PubMed PubMed Central
2. Keevil, B. G. LC–MS/MS Analysis of Steroids in the Clinical Laboratory. Clin. Biochem. 2016, 49, 989–997. https://doi.org/10.1016/j.clinbiochem.2016.04.009.Search in Google Scholar PubMed
3. Ponzetto, F.; Parasiliti-Caprino, M.; Leoni, L.; Marinelli, L.; Nonnato, A.; Nicoli, R.; Kuuranne, T.; Ghigo, E.; Mengozzi, G.; Settanni, F. LC-MS/MS Measurement of Endogenous Steroid Hormones and Phase II Metabolites in Blood Volumetric Absorptive Microsampling (VAMS) for Doping Control Purposes. Clin. Chim. Acta 2024, 557, 117890. https://doi.org/10.1016/j.cca.2024.117890.Search in Google Scholar PubMed
4. Šimková, M.; Kolátorová, L.; Drašar, P.; Vítků, J. An LC-MS/MS Method for the Simultaneous Quantification of 32 Steroids in Human Plasma. J. Chromatogr., B: Anal. Technol. Biomed. Life Sci. 2022, 1201–1202. https://doi.org/10.1016/j.jchromb.2022.123294.Search in Google Scholar PubMed
5. Harvey, D. J. Matrix-Assisted Laser Desorption/Ionization Mass Spectrometry of Carbohydrates and Glycoconjugates. Int. J. Mass Spectrom. 2003, 226, 1–35. https://doi.org/10.1016/S1387-3806(02)00968-5.Search in Google Scholar
6. Kozlov, O.; Lísa, M.; Riecan, M.; Kuda, O. Chiral Supercritical Fluid Chromatography-Mass Spectrometry with Liquid Chromatography Fractionation for the Characterization of Enantiomeric Composition of Fatty Acid Esters of Hydroxy Fatty Acids. Anal. Chim. Acta 2025, 1345. https://doi.org/10.1016/j.aca.2025.343735.Search in Google Scholar PubMed
7. Berzelius, J. J. On the Composition of Tartaric Acid and Racemic Acid (John’s Acid from the Vosges Mountains), on the Atomic Weight of Lead Oxide, Together with General Remarks on those Substances with Have the Same Composition but Different Properties. Poggendorf’s Annalen Der Physik Und Chemie 1830, 19, 305.Search in Google Scholar
8. J. H. van ’t Hoff, Voorstel tot uitbreiding der tegenwoordig in de scheikunde gebruikte structuur-formules in de ruimte renevens een daarmee samenhangende opmerking omtrent het verband tusschen optisch actief vermogen en chemische constitutie van organische verbindingen., in: 1874: pp. 1–16.Search in Google Scholar
9. Stewart, A. W. Atomic Structure from the Physico Chemical Standpoint.Pdf. Philos. Mag. J. Sci. 1918, 36, 326–337.10.1080/14786441008635836Search in Google Scholar
10. Rutherford, E. Nuclear Constitution of Atoms. Nature 1920, 105, 500–501; https://doi.org/10.1038/105500b0.Search in Google Scholar
11. Leone, M.; Robotti, N. Frédéric Joliot, Irène Curie and the Early History of the Positron (1932–33). Eur. J. Phys. 2010, 31, 975–987. https://doi.org/10.1088/0143-0807/31/4/027.Search in Google Scholar
12. Watson, J. T.; Sparkman, O. D. Introduction to Mass Spectrometry : Instrumentation, Applications and Strategies for Data Interpretation; John Wiley & Sons, Ltd: Chichester, 2008.10.1002/9780470516898Search in Google Scholar
13. de Hoffmann, E.; Stroobant, V. Mass Spectrometry Principles and Applications, 3rd ed.; John Wiley & Sons, Ltd.: Chichester, 2007.Search in Google Scholar
14. Isobars” in IUPAC Compendium of Chemical Terminology. In of Pure and Applied Chemistry; 2025. Online version 5.0.0, 5th ed.; International Union: Research Triangle Park, 2025; p 2025.Search in Google Scholar
15. Murray, K. K.; Boyd, R. K.; Eberlin, M. N.; Langley, G. J.; Li, L.; Naito, Y. Definitions of Terms Relating to Mass Spectrometry (IUPAC Recommendations 2013)* 2013, 85, 1515–1609; https://doi.org/10.1351/pac-rec-06-04-06.Search in Google Scholar
16. Sargent, M. Ed., Guide to achieving reliable quantitative LC-MS measurements, RSC Analytical Methods Committee, 2013. ISBN 978-0-948926-27-3.Search in Google Scholar
17. Habler, K.; Rexhaj, A.; Adling-Ehrhardt, M.; Vogeser, M. Understanding Isotopes, Isomers, and Isobars in Mass Spectrometry. J. Mass Spectrom. Adv. Clin. Lab. 2024, 33, 49–54. https://doi.org/10.1016/j.jmsacl.2024.08.002.Search in Google Scholar PubMed PubMed Central
18. Ionita, I. A.; Fast, D. M.; Akhlaghi, F. Development of a Sensitive and Selective Method for the Quantitative Analysis of Cortisol, Cortisone, Prednisolone and Prednisone in Human Plasma. J. Chromatogr., B: Anal. Technol. Biomed. Life Sci. 2009, 877, 765–772. https://doi.org/10.1016/j.jchromb.2009.02.019.Search in Google Scholar PubMed
19. Nist MS number 18940 Butane, and Nist MS Number 291282, Propanal; NIST Mass Spectrometry Data Center Collection; U.S. Department of Commerce: Gaithersburg, MD, 2014.Search in Google Scholar
20. Zhang, Y.; Go, E. P.; Jiang, H.; Desaire, H. A Novel Mass Spectrometric Method to Distinguish Isobaric Monosaccharides that are Phosphorylated or Sulfated Using ion-pairing Reagents. J. Am. Soc. Mass Spectrom. 2005, 16, 1827–1839. https://doi.org/10.1016/j.jasms.2005.07.010.Search in Google Scholar PubMed
© 2025 the author(s), published by De Gruyter, Berlin/Boston
This work is licensed under the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.

