Abstract
The reaction of dimethyl tin(IV) dichloride with 1,2-diphenylethane-1,2-dione dihydrazone in the presence of metal ions in a 2:2:1 molar ratio resulted in the isolation of macrocyclic complexes of the types [MLX2Sn(CH3)2] and [CuLSn(CH3)2]X2 (M=Mn(II), Co(II), Ni(II) and Zn(II); X=Cl or NO3). In both the cases, complexes exhibit identical patterns suggesting them to be isostructural. The cyclization has been identified on the basis of IR, 1H NMR and 119Sn NMR studies, and the overall geometry of the complexes has been assigned by the data obtained from elemental analyses, electron paramagnetic resonance and electronic spectral studies, and conductivity and magnetic susceptibility measurements. Except for the complexes of copper, which are square planar, all other complexes have octahedral structures. All the synthesized tin(IV) macrocyclic metal complexes were tested for their in vitro anti-fungal activity against some fungal strains, namely Aspergillus fumigatus and Trichoderma harzianum.
Introduction
The relevant transition metal complexes are of great interest in coordination chemistry. Although this subject has been extensively studied (Keypour et al., 2015a,b), the synthesis and complexing properties of the macrocycles deserve special attention as in some cases these macrocycles have been used for the construction of the corresponding polyaza macrocyclic complexes (Romain et al., 2015). The importance of macrocyclic complexes has provided a motivation for the investigation of the metal ion chemistry of biological systems as well as of cyclic ligand systems (Zhang et al., 2013). Schiff-base ligands contain an N=C=N structural unit and form a strong chelate ring with different metal ions or anionic species, with significant potential in areas such as catalysis, modeling of metalloenzymes and molecular recognition (Khan et al., 1990; Chandra and Sangeetika, 2004; Dong et al., 2014) which may affect the nature of the complex formed (Keypour et al., 2015a,b). Recently, the template synthesis of various imine macrocyclic complexes has been reported (Adams et al., 1999; Hong et al., 2005; Daszkiewicza et al., 2010) and has a long-term interest in producing imine macrocyclic complexes of a wide variety. The template condensation reaction lies at the heart of macrocyclic chemistry and has been widely used for the synthesis of macrocyclic complexes. Many of the synthetic routes to macrocyclic ligands involve the use of a metal ion template to orient the reacting groups of the ligand in the desired conformation for optimum ring closure and thereby promote the cyclization reaction (Curtis, 1968; Niasari and Davar, 2006). The extensive developments over recent decades in the use of organotin compounds as reagents or intermediates in organic synthesis prompted the preparation of many new organotin compounds (Ghani and Deo, 2014). A considerable number of organotin macrocyclic complexes with nitrogen donors have been reported over the past few decades (Chaudhary et al., 2006; Zhang et al., 2016). Herein, we report a new series of tetraimine macrocyclic complexes of the types [MLX2Sn(CH3)2] and [CuLSn(CH3)2]X2 [M=Mn(II), Co(II), Ni(II) and Zn(II); X=Cl or NO3] obtained by the template condensation of dimethyl tin(IV) dichloride with 1,2-diphenylethane-1,2-dione dihydrazone (DPEDDH) in the presence of metal ions in a 2:2:1 molar ratio.
Results and discussion
The tertaiminetetraamide macrocyclic complexes [MLX2] and [CuL]X2 (M=Mn(II), Co(II), Ni(II) and Zn(II); X=Cl or NO3) have been prepared by the condensation reaction of DPEDDH with dimethyl tin(IV) dichloride followed by metal templation in a 2:2:1 molar ratio (Scheme 1).
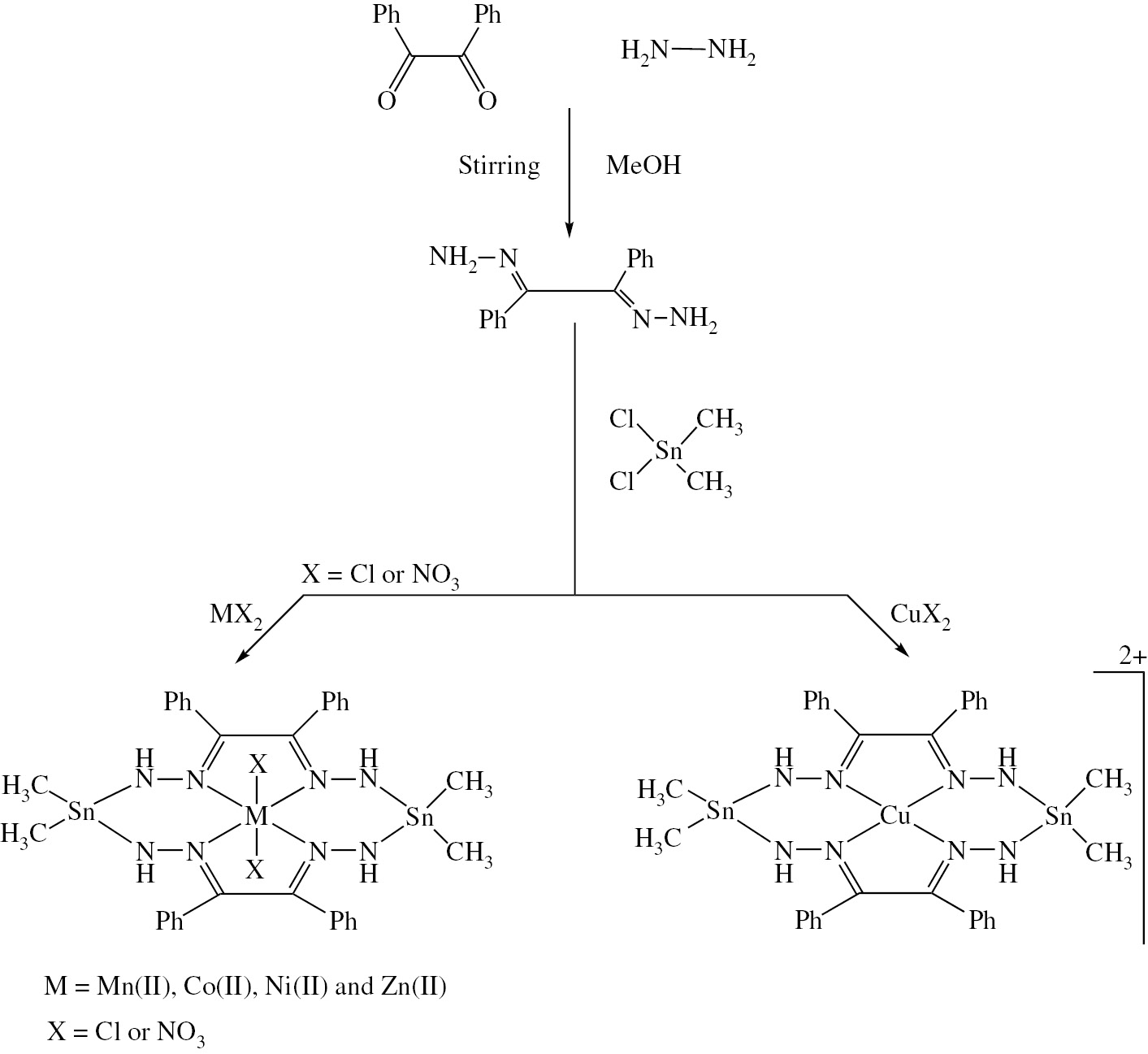
Formation and suggested structure of tin(IV) macrocyclic complexes.
The complexes examined show poor solubility in solvents such as chloroform, carbon tetrachloride and water. However, they are freely soluble in dimethylsulfoxide (DMSO) and warm dimethylformamide. The purity of the product was monitored by thin-layer chromatography using silica gel. The elemental analysis results agree well with the mononuclear structure. The molar conductivities in DMSO indicate (Geary, 1971; Khan et al., 2010) that copper(II) complexes are electrolytic while others are non-electrolytic in nature. The IR spectra of all the complexes are similar but strikingly different from the spectra of the free ligand. The spectra of the free ligand (DPEDDH) show two strong, medium-intensity prominent bands at 3290–3210 cm−1, assignable to ν(N-H) vibrations of amino groups which occur at slightly lower frequencies than are usually associated with ν(N-H) vibrations of primary amino groups (Khan and Ghani, 2005). This result is expected because of the high degree of conjugation in the ligand as the interaction of the amino group lone pair with conjugated groups weakens the N-H bond. The ν(C=N) band vibration appeared at 1615 cm−1, which is found to be slightly negatively shifted than that usually found for the azomethine linkage, further confirms the delocalization of electrons in the free ligand.
The reported macrocyclic complexes exhibit a strong intensity band in the range of 1580–1595 cm−1 assignable (Nelson et al., 1988; Thoonen et al., 2006) to the coordinated ν(C=N). There is an observed increase in the ν(C=N) band intensity which may be due to the possible transformation of the trans structure of the free ligand (DPEDDH) into cis configuration upon complexation. The cis structure is stabilized by the chelate ring, lowering the symmetry which consequently enhances the ν(C=N) band intensity. The bands at 425–470 cm−1 originate from (M-N) vibration. In addition, the IR spectra of nitrato complexes exhibit bands at 1280, 1040 and 850 cm−1 which are consistent with the monodentate nature (Ball and Blake, 1969) of this group. However, the phenyl ring vibrations appear at their expected positions.
The 1H NMR spectra of benzildihydrazone (DPEDDH) and the corresponding zinc complexes have been recorded and compared. Benzildihydrazone shows a multiplet at 7.18 ppm and a slightly broad signal at 4.92 ppm assignable (Chaudhary and Singh, 2001) to the phenyl ring (C6H5, 10 H) and amino (N-NH2, 4 H) protons of the hydrazine moiety. Both the zinc complexes showed the appearance of a band at 8.42 and 8.45 ppm, reasonably ascribed (Parker et al., 1994) to the (N-NH, 4 H) protons. A multiplet at 6.90 and 6.97 ppm in both complexes is characteristic of phenyl ring (C6H5, 20 H) protons. The absence of characteristic peaks corresponding to the protons of hydrazone moieties further confirms that cyclization has indeed taken place.
The complexes gave a sharp signal at δ −578 ppm and δ −576 ppm in the 119Sn NMR spectra which is at par with the reported values for a four-coordinated geometry (Ghani and Deo, 2014).
The room temperature electron paramagnetic resonance (EPR) spectra of both polycrystalline Cu(II) complexes exhibit a similar single broad absorption band. However, no complex is found to give hyperfine splitting. The calculated g║ and g⊥ values appeared in the region 2.31, 2.34 and 2.08, 2.09, respectively, which support the contention that dx2−dy2 may be the ground state of the unpaired electron. Both the complexes show g║>2.3, indicating that they exhibit appreciable ionic character (Khan et al., 2006). The g values are related by the expression G=(g║−2)/(g⊥−2), and the present complexes [CuLSn(CH3)2]X2 (X=Cl or NO3) gave the axial symmetry parameter G in the 3.875, 3.775 region, indicating (Proctor et al., 1968) very small exchange interaction among copper(II) ions in these complexes, as the G values are < 4.
The electronic spectra of manganese gave two bands in the 22 400–22 550 cm−1 and 18 650–18 850 cm−1 regions which may be assigned to the 6A1g→4T2g(F) and 6A1g→4T1g(P) transitions, respectively, corresponding to an octahedral environment around the Mn(II) ion. The cobalt complexes exhibit two ligand field bands in the 21 400–21 700 and 15 950–16 200 cm−1 regions which may reasonably correspond to the 4T1g(F)→4T1g(P) and 4T1g(F)→4A2g(F) transitions, respectively, consistent with octahedral geometry (Lever, 1984) around the Co(II) ion. All the nickel complexes exhibit two electronic bands in the 21 200–24 350 and 18 200–18 750 cm−1 regions which may be ascribed to the 3A2g(F)→3T1g(P) and 3A2g(F)→3T1g(F) transitions, respectively, suggesting (Kimura, 1986) an octahedral geometry around the metal ion. Magnetic moment values are in close agreement with their electronic spectral data which further support the proposed geometry.
The observed magnetic moments of Cu(II) complexes in the range 1.87–1.92 Bohr Magneton (B.M.) are consistent with the square planar Cu(II) system (Inba et al., 2013). The electronic spectra of these complexes show a broad band maximum at 16 050 cm−1 which may be assigned to the 2B1g → 2A1g transition. However, two weak shoulders appearing in the regions 21 500–21 750 and 11 750–12 450 cm−1 may be attributed to the 2B1g→2Eg and 2B1g→2B2g transitions, respectively, suggesting (Jain et al., 1977) a square-planar geometry around the Cu(II) ion.
The antifungal activities of the synthesized complexes were tested against Aspergillus fumigatus and Trichoderma harzianum by the radial growth method (Bansal and Singh, 2000) using Czapek’s agar medium. The complexes were directly mixed with the medium in 50, 100 and 200 ppm concentrations in methanol. The controls were run and three replicates were used in each case. The linear growth of the fungus was obtained by measuring the diameter of the fungal colony after 5 days. The amount of growth inhibition in all the replicates was calculated by the following equation: percent inhibition=(C−T)/C×100, where C is the diameter of the fungal colony in the control plate and T is the diameter of the fungal colony in the test plate. The experimental data of antifungal screening reveal that the metal complexes are more fungi toxic than the starting materials. The fungi toxicity of precursors and their complexes decreases on lowering the concentration. The results recorded from the biocidal activity were also compared with the standard fungicide Folicur (Table 1). The increased fungi toxicity of the complexes may be due to the chelation as the polarity of the metal ion in the complexes which reduces considerably due to the partial sharing of its positive charge with the donor groups and possible π-electron delocalization over the whole chelate ring system. This in turn increases its permeation through the lipoid layers of the fungal membrane (Sharma et al., 2001). The enhanced activities of the metal complexes relative to free macrocycles can be attributed to the increased lipophilic nature of these complexes arising due to chelation (Ibrahim et al., 2006).
Antifungal activity in μg/mL of the complexes and starting materials after 5 days at 25±2°C.
| Test compounds | Aspergillus fumigatus | Trichoderma harzianum | ||||
|---|---|---|---|---|---|---|
| 50 ppm | 100 ppm | 200 ppm | 50 ppm | 100 ppm | 200 ppm | |
| DPEDDH | 25 | 38 | 64 | 31 | 41 | 58 |
| [MnLSn(CH3)2(NO3)2] | 32 | 65 | 71 | 27 | 45 | 82 |
| [MnLSn(CH3)2Cl2] | 38 | 63 | 80 | 43 | 70 | 79 |
| [CoLSn(CH3)2(NO3)2] | 44 | 72 | 85 | 48 | 72 | 83 |
| [CoLSn(CH3)2Cl2] | 37 | 61 | 79 | 35 | 59 | 59 |
| NiLSn(CH3)2(NO3)2] | 36 | 78 | 84 | 39 | 64 | 76 |
| [NiLSn(CH3)2Cl2] | 42 | 63 | 72 | 48 | 71 | 65 |
| [CuLSn(CH3)2](NO3)2 | 39 | 68 | 77 | 36 | 48 | 78 |
| [CuLSn(CH3)2]Cl2 | 34 | 62 | 74 | 40 | 51 | 74 |
| [ZnLSn(CH3)2(NO3)2] | 40 | 67 | 82 | 44 | 73 | 85 |
| [ZnLSn(CH3)2Cl2] | 35 | 60 | 78 | 37 | 67 | 81 |
| Standard Folicur | 89 | 93 | 101 | 90 | 93 | 102 |
It is rational to assume that more lipophilic compounds are more active simply because they enter the lipoid layers of cell membranes more briskly.
Conclusions
The tertaimine macrocyclic complexes of Mn(II), Co(II), Ni(II) and Zn(II) have been synthesized by the condensation reaction of DPEDDH with dimethyl tin(IV) dichloride followed by metal templation in an appropriate molar ratio which directs the course of the reaction. The attempts to prepare metal-free macrocyclic ligands were not successful as there was formation of oily products which could not be isolated and characterized. Analytical data, electronic spectra, magnetic susceptibility, IR, 1H NMR and EPR data reveal mononuclear nature of all the complexes. Except for the copper(II) complexes which are electrolytic, the others are non-electrolytic in nature. The complexes exhibit moderate antifungal activity on Aspergillus fumigatus and Trichoderma harzianum.
Experimental section
All chemicals were purchased from Merck (Kenilworth, NJ, USA) and were used without further purification. Benzildihydrazone (DPEDDH) was prepared by the reaction of benzyl and hydrazinehydrate in methanol in 1:2 molar ratios. Elemental analyses were carried out on a Perkin-Elmer 240 elemental analyzer (Waltham, MA, USA). 1H NMR spectra in DMSO-d6 (Sigma-Aldrich, St. Louis, MO, USA) were recorded on a JEOL-FX-100-FT-NMR spectrometer (Cranford, NJ, USA), with MeSi4 (Sigma-Aldrich, St. Louis, MO, USA) as an internal standard, and 119Sn spectra were recorded on Bruker ACF300 (Billerica, MA, USA) (at 300 MHz) with SnMe4 as an external standard. The estimation of halogen was done gravimetrically (Vogel, 1961) and the metals were estimated by titrating with standard EDTA solution (Reilley et al., 1959). The FT-IR spectra (4000–400 cm−1) were recorded as KBr disks on an IR 408 Shimadzu spectrophotometer (Columbia, MD, USA). The electronic spectra of the compounds were recorded on a Pye-Unicam 8800 spectrophotometer (Cambridge, UK) at room temperature. The EPR spectra of solid complexes were recorded on a JEOL JES RE2X EPR spectrometer (Arvada, CO, USA). Magnetic susceptibility measurements were carried out using a Faraday balance at 25°C. Conductance measurements were performed using a Hanna HI 8820 conductivity meter (Carrollton, TX, USA).
Synthesis of complexes
A gradual color change was observed when a sample (1.191 g, 20 mmol) of DPEDDH, suspended in CH2Cl2 (150 cm3), was treated with a methanolic (65 cm3) solution of metal salt (10 mmol) and dimethyltin(IV) dichloride (0.68 cm3, 20 mmol) simultaneously. The reaction mixture was stirred for 8 h and then it was refluxed for 13 h. The product obtained was filtered off, washed with CH2Cl2 and dried over fused CaCl2 kept in a desiccator.
Dinitrato(6,13-tetramethyl-2,3,9,10-tetraphenyl-1,4,5,7,8,11,12,14-octaazacyclotetraadecane-1,3,8,10-tetraene) Mn(II)-Sn(IV): Pink; mp 191–193°C; Λm 21.5 cm−1 ohm−1 mol−1; μeff 5.75 B.M.; IR: 3250, 1595, 440, 1230, 1015, 860 cm−1; Anal. Calcd for C32H38N10O6MnSn2: C, 40.4; H, 4.0; N, 14.7; Mn, 5.8. Found: C, 40.8; H, 4.4; N, 14.9; Mn, 5.8.
Dichloro(6,13-tetramethyl-2,3,9,10-tetraphenyl-1,4,5,7,8,11,12,14-octaazacyclotetraadecane-1,3,8,10-tetraene) Mn(II)-Sn(IV): Light pink; mp 185°C; Λm 18.6 cm−1 ohm−1 mol−1; μeff 5.70 B.M.; IR: 3270, 1580, 425, 1235, 1010, 865 cm−1; Anal. Calcd for C32H38N8O6Cl2MnSn2: C, 42.8; H, 4.3; N, 12.5; Cl, 7.9; Mn, 6.1. Found: C, 43.1; H, 4.7; N, 12.9; Cl, 8.3; Mn, 6.4.
Dinitrato(6,13-tetramethyl-2,3,9,10-tetraphenyl-1,4,5,7,8,11,12,14-octaazacyclotetraadecane-1,3,8,10-tetraene) Co(II)-Sn(IV): Bright pink; mp 210–212°C; Λm 19.9 cm−1 ohm−1 mol−1; μeff 4.65 B.M.; IR: 3275, 1585, 460, 1240, 1025, 865 cm−1; Anal. Calcd for C32H38N10O6CoSn2: C, 40.2; H, 4.0; N, 14.7; Co, 6.2. Found: C, 40.6; H, 4.2; N, 15.0; Co, 6.6.
Dichloro(6,13-tetramethyl-2,3,9,10-tetraphenyl-1,4,5,7,8,11,12,14-octaazacyclotetraadecane-1,3,8,10-tetraene) Co(II)-Sn(IV): Light pink; mp 221–223°C; Λm 17.5 cm−1 ohm−1 mol−1; μeff 4.57 B.M.; IR: 3275, 1590, 470, 1225, 1030, 870 cm−1; Anal. Calcd for C32H38N8O6Cl2CoSn2: C, 42.6; H, 4.2; N, 12.4; Cl, 7.8; Co, 6.5. Found: C, 45.0; H, 3.9; N, 12.1; Cl, 8.3; Co, 6.9.
Dinitrato(6,13-tetramethyl-2,3,9,10-tetraphenyl-1,4,5,7,8,11,12,14-octaazacyclotetraadecane-1,3,8,10-tetraene) Ni(II)-Sn(IV): Green; mp 194–196°C; Λm 20.5 cm−1 ohm−1 mol−1; μeff 3.18 B.M.; IR: 3265, 1595, 450, 1240, 1020, 845 cm−1; Anal. Calcd for C32H38N10O6NiSn2: C, 40.2; H, 4.0; N, 14.7; Ni, 6.1. Found: C, 40.6; H, 4.4; N, 15.1; Ni, 6.1.
Dichloro(6,13-tetramethyl-2,3,9,10-tetraphenyl-1,4,5,7,8,11,12,14-octaazacyclotetraadecane-1,3,8,10-tetraene) Ni(II)-Sn(IV): Dim green; mp 204–206°C; Λm 21.0 cm−1 ohm−1 mol−1; μeff 3.12 B.M.; IR: 3240, 1585, 435, 1230, 1025, 860 cm−1; Anal. Calcd for C32H38N8O6Cl2NiSn2: C, 42.6; H, 4.3; N, 12.4; Cl, 7.9; Ni, 6.5. Found: C, 42.9; H, 4.1; N, 12.1; Cl, 7.4; Ni, 6.1.
(6,13-Tetramethyl-2,3,9,10-tetraphenyl-1,4,5,7,8,11,12,14-octaazacyclotetraadecane-1,3,8,10-tetraene) Cu(II)-Sn(IV): Brown; mp 214–216°C; Λm 107.0 cm−1 ohm−1 mol−1; μeff 1.92 B.M.; IR: 3250, 1580, 455, 1235, 1010, 840 cm−1; Anal. Calcd for C32H38N8O6CuSn2: C, 40.0; H, 3.1; N, 14.6; Cu, 6.6. Found: C, 40.5; H, 3.4; N, 14.9; Cu, 6.9.
(6,13-Tetramethyl-2,3,9,10-tetraphenyl-1,4,5,7,8,11,12,14-octaazacyclotetraadecane-1,3,8,10-tetraene) Cu(II)-Sn(IV): Dark brown; mp 220–222°C; Λm 98.0 cm−1 ohm−1 mol−1; μeff 1.87 B.M.; IR: 3245, 1590, 450, 1225, 1000, 875 cm−1; Anal. Calcd for C32H38N8O6CuSn2: C, 42.4; H, 4.2; N, 12.4; Cu, 7.0. Found: C, 42.9; H, 3.9; N, 12.1; Cu, 7.8.
Dinitrato(6,13-tetramethyl-2,3,9,10-tetraphenyl-1,4,5,7,8,11,12,14-octaazacyclotetraadecane-1,3,8,10-tetraene) Zn(II)-Sn(IV): White; mp 189–191°C; Λm 19.4 cm−1 ohm−1 mol−1; IR: 3250, 1595, 450, 1230, 1015, 855 cm−1; Anal. Calcd for C32H38N10O6ZnSn2: C, 39.9; H, 4.0; N, 14.6; Zn, 6.8. Found: C, 39.7; H, 4.4; N, 15.1; Zn, 6.5.
Dichloro(6,13-tetramethyl-2,3,9,10-tetraphenyl-1,4,5,7,8,11,12,14-octaazacyclotetraadecane-1,3,8,10-tetraene) Zn(II)-Sn(IV): Off white; mp 196–198°C; Λm 18.7 cm−1 ohm−1 mol−1; IR: 3265, 1585, 445, 1240, 1010, 850 cm−1; Anal. Calcd for C32H38N8O6Cl2ZnSn2: C, 42.3; H, 4.2; N, 12.3; Cl, 7.8; Zn, 7.2. Found: C, 42.7; H, 4.6; N, 11.9; Cl, 7.4; Zn, 7.5.
Acknowledgment
The author is grateful to HoD, Department of Chemistry, Arba Minch University, Arba Minch, Ethiopia, for providing necessary research facilities.
References
Adams, H.; Elsegood, M. R. J.; Fenton, D. E.; Heath, S. L.; Ryan, S. J. Dinuclear silver(I) complexes of bibracchial tetraimine Schiff base macrocycles derived from pyrrole-2,5-dicarbaldehyde. J. Chem. Soc., Dalton Trans.1999, 12, 2031–2038.10.1039/a901992hSearch in Google Scholar
Ball, P. W.; Blake, A. B. Magnetic properties of polynuclear complexes. Part I: Superexchange in some binuclear nickel(II) complexes. J. Chem. Soc. A.1969, 1415–1422.10.1039/j19690001415Search in Google Scholar
Bansal, A.; Singh, R. V. Template synthesis, structural and biological studies of new tetraaza macrocyclic complexes of lead(II). Bol. Soc. Chil. Quim. 2000, 45, 479–486.Search in Google Scholar
Chandra, S.; Sangeetika, X. EPR, magnetic and spectral studies of copper(II) and nickel(II) complexes of Schiff base macrocyclic ligand derived from thiosemicarbazide and glyoxal. Spectrochim. Acta2004, 60A, 147–153.10.1016/S1386-1425(03)00220-8Search in Google Scholar
Chaudhary, A.; Singh, R. V. Synthesis and characterization of 18-to 22-membered tetraazamacrocycles and their tin complexes. Ind. J. Chem.2001, 40A, 1330–1333.Search in Google Scholar
Chaudhary, A.; Phor, A.; Singh, R. V. Studies on potentially biodynamic heterocyclic organotin(II) macrocyclic complexes. Heterocycl. Commun.2006, 12, 53–60.10.1515/HC.2006.12.1.53Search in Google Scholar
Curtis, N. F. Macrocyclic coordination compounds formed by condensation of metalamine complexes with aliphatic carbonyl compounds. Coord. Chem. Rev.1968, 3, 3–47.10.1016/S0010-8545(00)80104-6Search in Google Scholar
Daszkiewicza, B. K.; Bilewiczb, R.; Woźniakb, K. Tetraimine macrocyclic transition meta complexes as building blocks for molecular devices. Coord. Chem. Rev.2010, 254, 1637–1660.10.1016/j.ccr.2009.12.004Search in Google Scholar
Dong, S.; Zheng, B.; Wang, F.; Huang, F. Supramolecular polymers constructed from macrocycle-based host-guest molecular recognition motifs. Acc. Chem. Res. 2014, 47, 1982–1994.10.1021/ar5000456Search in Google Scholar PubMed
Geary, W. J. The use of conductivity measurements in organic solvents for the characterisation of coordination compounds. Coord. Chem. Rev.1971, 7, 81–122.10.1016/S0010-8545(00)80009-0Search in Google Scholar
Ghani, S. S.; Deo, A. Synthesis and characterization of N2S2-tin macrocyclic complexes of Co(II), Ni(II), Cu(II) and Zn(II). Main Group Met. Chem. 2014, 37, 137–142.10.1515/mgmc-2014-0032Search in Google Scholar
Hong, Z.; Zheng, H. P.; Zhi, Q. P.; Bo, L.; Xue, L. H.; Yong, Q. L. Novel heterodinuclear transition metal macrocyclic complexes: syntheses, characterization and crystal structures. J. Coord. Chem. 2005, 58, 443–451.10.1080/00958970412331336286Search in Google Scholar
Ibrahim, A. S.; John, E. E. J.; Fu, Y.; Spellberg, B. Deferiprone iron chelation as a novel therapy for experimental mucormycosis. J. Antimicrob. Chemother.2006, 58, 1070–1073.10.1093/jac/dkl350Search in Google Scholar PubMed
Inba, P. J. K.; Annaraj, B.; Thalamuthu, S.; Neelakantan, M. A. Cu(II), Ni(II), and Zn(II) complexes of salan-type ligand containing ester groups: synthesis, characterization, electrochemical properties, and in vitro biological activities. Bioinorg. Chem. Appl.2013, 2013, 1–11.Search in Google Scholar
Jain, M. C.; Srivastava, A. K.; Jain, P. C. Some tetragonally distorted copper (II) complexes of 4-benzylamidothiosemicarbazide and its thiosemicarbazone. Inorg. Chim. Acta1977, 23, 199–203.10.1016/S0020-1693(00)94761-3Search in Google Scholar
Keypour, H.; Rezaeivala, M.; Monsef, M. M.; Sayin, K.; Dilek, N.; Unver, H. Synthesis and characterization of Co(II), Ni(II), Cu(II) and Zn(II) complexes with a new homopiperazine macrocyclic Schiff base ligand. Inorg. Chim. Acta2015a, 432, 243–249.10.1016/j.ica.2015.04.017Search in Google Scholar
Keypour, H.; Shooshtari, A.; Rezaeivala, M.; Kup, F. O.; Rudbari, H. A. Synthesis of two new N2O4 macroacyclic Schiff base ligands and their mononuclear complexes: spectral, X- ray crystal structural, antibacterial and DNA cleavage activity. Polyhedron. 2015b, 97, 75–82.10.1016/j.poly.2015.02.029Search in Google Scholar
Khan, T. A.; Ghani, S. S. Synthesis and physico-chemical studies of 14-membered tetraaza macrocyclic complexes of Co(II), Ni(II), Cu(II) and Zn(II) derived from 3,4-diaminobenzophenone. Polish J. Chem. 2005, 79, 817–823.Search in Google Scholar
Khan, M. M. T.; Halligudi, S. B.; Shukla, S.; Shaikh, Z.A. Reductive carbonylation of nitrobenzene to phenylurethane catalyzed by Ru(III)-schiff base complexes. J. Mol. Catal.1990, 57, 301–305.10.1016/0304-5102(90)85004-2Search in Google Scholar
Khan, T. A.; Ghani, S. S.; Tabassum, S. Template synthesis of 16-membered octaazamacrocyclic complexes of Fe(II), Co(II), Ni(II), Cu(II) and Zn(II) ions. Pol. J. Chem.2006, 80, 1449–1455.Search in Google Scholar
Khan, T. A.; Ghani, S. S.; Naseem, S. Interaction of Co(II), Ni(II), Cu(II), and Zn(II) with 12- and 14-membered macrocycles containing O2N2 donors. J. Coord. Chem.2010, 63, 4411–4420.10.1080/00958972.2010.535524Search in Google Scholar
Kimura, E. Distinctive coordination chemistry and biological relevance of complexes with macrocyclic oxo polyamines. J. Coord. Chem.1986, 15, 1–28.10.1080/00958978608075853Search in Google Scholar
Lever, A. B. P. Inorganic Electronic Spectroscopy; Elsevier: Amsterdam, 1984.Search in Google Scholar
Nelson, J.; Murphy, B.P.; Drew, M. G. B.; Yates, P. C.; Nelson, S. M. Binucleating N6 24-and 26-membered macrocyclic ligands. Part 2: Transition-metal homo- and hetero-binuclear complexes: X-ray crystallographic structure determination of a lead–manganese heterobinuclear complex. J. Chem. Soc. Dalton Trans.1988, 4, 1001–1010.10.1002/chin.198830263Search in Google Scholar
Niasari, M. S.; Davar, F. In situ one-pot template synthesis (IOPTS) and characterization of copper(II) complexes of 14-membered hexaaza macrocyclic ligand 3,10-dialkyl-dibenzo-1,3,5,8,10,12-hexaazacyclotetradecane. Inorg. Chem. Commun. 2006, 9, 175–179.10.1016/j.inoche.2005.10.028Search in Google Scholar
Parker, D.; Pulukkody, K.; Smith, F. C.; Batsanov, A.; Howard, J. A. K. Structures of the yttrium complexes of 1,4,7,10-tetraazacyclododecane-N,N′,N″,N‴-tetraacetic acid (H4dota) and N,N″-bis(benzylcarbamoylmethyl)diethylenetriamine-N,N′,N″-triacetic acid and the solution structure of a zirconium complex of H4dota. J. Chem. Soc. Dalton Trans.1994, 5, 698–693.10.1039/DT9940000689Search in Google Scholar
Proctor, I. M.; Hathaway, B. J.; Nicholls, P. The electronic properties and stereochemistry of the copper(II) ion. Part I: Bis(ethylenediamine) copper(II) complexes. J. Chem. Soc. A.1968, 1678–1684.10.1039/j19680001678Search in Google Scholar
Romain, C.; Bennington, M. S.; White, A. J. P.; Williams, C. K.; Brooker, S. Macrocyclic dizinc(II) alkyl and alkoxide complexes: reversible CO2 uptake and polymerization catalysis testing. Inorg. Chem.2015, 54, 11842–11851.10.1021/acs.inorgchem.5b02038Search in Google Scholar PubMed
Reilley, C. N.; Schmid, R. W.; Sadek, F. A. Chelon approach to analysis (I) survey of theory and application. J. Chem. Educ. 1959, 36, 555–564.10.1021/ed036p555Search in Google Scholar
Sharma, K.; Fahmi, N.; Singh, R. V. Synthesis, characterization and toxicity of new heterobimetallic complexes of platinum(II) and palladium(II). Appl. Organomet. Chem.2001, 15, 221–226.10.1002/aoc.132Search in Google Scholar
Thoonen, S. H. L.; Hoek, H. V.; Wolf, E. D.; Lutz, M.; Spek, A. L.; Deelman, B. J.; Koten, G. V. Synthesis of novel terdentate N,C,N′-coordinated butyltin (IV) complexes and their redistribution reactions with SnCl4. J. Organomet. Chem.2006, 691, 1544–1553.10.1016/j.jorganchem.2005.11.063Search in Google Scholar
Vogel, A. I. Text Book of Quantitative Inorganic Analysis; Longmans: London, 1961.Search in Google Scholar
Zhang, L.; Mu, J.; Jiang, Z.; Zhang, H.; Yue, X. Fully aromatic macrocycle-terminated polyimide: synthesis and cross-linking. Polym. Adv. Technol. 2013, 24, 415–420.10.1002/pat.3097Search in Google Scholar
Zhang, R.; Wang, F.; Li, Q.; Zhang, S.; Ma, C. Syntheses and structural characterization of organotin complexes derived from 2-trifluoromethyl benzeneseleninic acid: tetranuclear macrocycle, 1-D polymeric chain, helical double-chain. J. Coord. Chem. 2016, 69, 704–713.10.1080/00958972.2015.1124989Search in Google Scholar
©2017 Walter de Gruyter GmbH, Berlin/Boston
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Articles in the same Issue
- Frontmatter
- Research Articles
- Lead acetate toxicity on human lymphocytes at non-cytotoxic concentrations detected in human blood
- Synthesis, characterization and biological activity of tin(IV) macrocyclic complexes derived from 1,2-diphenylethane-1,2-dione dihydrazone
- Synthesis and characterization of a new tin(IV) complex with anthracene-9-carboxylic acid as a precursor in the preparation of an organic light-emitting diode
- Synthesis of propargylamines catalyzed by nano-colloidal silica-tethered polyhedral oligomeric silsesquioxanes with eight branches of 3-aminopropyltriethoxysilane as an efficient catalyst
- Short Communication
- Synthesis, characterization, and thermal properties of N,N,N′,N′-tetramethyl guanidinium tribromidocadmate(II) exhibiting an unusual coordination geometry
Articles in the same Issue
- Frontmatter
- Research Articles
- Lead acetate toxicity on human lymphocytes at non-cytotoxic concentrations detected in human blood
- Synthesis, characterization and biological activity of tin(IV) macrocyclic complexes derived from 1,2-diphenylethane-1,2-dione dihydrazone
- Synthesis and characterization of a new tin(IV) complex with anthracene-9-carboxylic acid as a precursor in the preparation of an organic light-emitting diode
- Synthesis of propargylamines catalyzed by nano-colloidal silica-tethered polyhedral oligomeric silsesquioxanes with eight branches of 3-aminopropyltriethoxysilane as an efficient catalyst
- Short Communication
- Synthesis, characterization, and thermal properties of N,N,N′,N′-tetramethyl guanidinium tribromidocadmate(II) exhibiting an unusual coordination geometry


