Abstract
Heterocyclic compounds are of immense interest due to their extensive occurrence in nature as well as their applicability in the pharmaceutical industry. Benzothiazole and its derivatives encompass an attractive heterocyclic class that displays practical applications ranging from mevdicine to photography and agriculture, among other things. This review focuses on the synthesis and specific applications of various benzothiazole cyanine dyes. Benzothiazole-containing heterocyclic structures are prominent throughout the literature and it is very important to acknowledge their efficacy and applicability as we discuss herein.
Introduction
Structural characteristics of carbocyanine dyes
Cyanine dyes are a unique class of compounds that have a wide range of applications in numerous fields. The first member of this type was reported by Williams in 1856 [1]. The name cyano was given due to the beautiful blue (blue – kyano in Greek) color of the dye. This dye was obtained by treatment of quinoline and 4-methylquinoline with amyl iodide followed by reaction with ammonia. Vogel in 1873 found that cyanine dyes can be used to increase sensitivity of the photographic plate [2]. It was the turning point in the history of the cyanine dyes.
Cyanine dyes are a subclass of polymethine dyes. As shown in Figure 1, polymethine dyes consist of two nitrogen centers joined by a conjugated chain of odd number of methine carbons or a conjugated system of double bonds. This polymethine bridge connects an electron acceptor group at one end and an electron donor group at the other. Conjugation between the electron donor and acceptor groups results in delocalization of π electrons and hence positive charge over the two nitrogen atoms.

General structure of polymethine dyes.
Examples of related cyanine dyes are shown in Figure 2. Common names of the cyanine dyes are based on the number of methine carbons present in the polymethine chain. In Figure 2, the polymethine cyanine dyes are designated as mono-, tri-, penta-, and heptamethine cyanines for n = 0, 1, 2, and 3, respectively. The absorption/emission wavelengths of the cyanine dyes depend upon the length of the polymethine chain and the nature of the terminal groups. The monomethine and trimethine cyanine dyes usually absorb in the visible region (500–600 nm) of the electronic spectrum with each added (CH=CH) methine unit inducing a bathochromic shift of approximately 100 nm in the electronic spectrum resulting in an absorption wavelength of 700–800 nm for penta- and heptamethine cyanines. The 4-pyrilium, 4-thiopyrilium, and benz[c,d]indole heterocyclic end groups extend absorption/emission wavelength well into the near-infrared region, whereas the presence of the benzoxazole end group results in a hypsochromic shift in the electronic spectrum. Polymethine cyanine dyes are generally classi fied based on the nature of the end groups present on the polymethine chain. Dyes with two heterocyclic terminal groups are referred to as closed chain cyanine dyes or generally referred to as cyanines. The two heterocycles in the cyanines can either be identical or different. Hemicyanines are characterized by the presence of one heterocyclic and another non-cyclic end group. Dyes without a terminal heterocyclic moiety are defined as streptocyanines or open chain cyanines as shown in Figure 2.

Classification of cyanine dyes.
Cyanine dyes are structurally classified as symmetric or asymmetric cyanine dyes as depicted in Figure 3. These symmetric and asymmetric cyanine dyes are quite different with respect to spectral characteristics and nucleic acid binding behavior [3].

Classification of cyanine dyes.
In the literature, it is recognized that in 1926, Koenig identified the chromophoric nature of the polymethine structure of the cyanine dye and reported the synthesis of the first chiral polymethine dye [4, 5]. Since then many different types of cyanine dyes have been synthesized. The first bridged cyanine dye synthesis was published in 1933 where trimethine chain formed a part of cyclopentadiene ring [4]. In a review by Behera et al., it was discussed that some naturally occurring cyanine dyes have been isolated from Beta vulgaris and Amanita muscaria [5]. Cyanine dyes possess some characteristic properties which include structure-dependent photochemical stability, narrow absorption band, high molar absorption coefficients (~105 m-1 cm-1), tendency to form aggregates in solution, and relatively high fluorescence intensity. A large number of cyanine dyes have been synthesized using different heterocycles such as indolenine, quinoline, benzoxazole, and benzothiazole.
Benzothiazoles have a planar structure, which is an essential criterion for nucleic acid binding and hence for their applications as an effective biological marker [6, 7]. The use of benzothiazole compounds as in vivo imaging agents for Alzheimer’s disease is considered to be a major breakthrough for benzothiazole studies [8].
Synthesis and applications of benzothiazole cyanine dyes
The synthesis of the first benzothiazole cyanine dye was reported in the late 18th century [9]. It was synthesized by heating N-amylbenzothiazolium iodide and 2-methylbenzothiazole in the presence of ammonia. To date, a large number of symmetric and asymmetric benzothiazole cyanine dyes have been synthesized [10–12].
Benzothiazole cyanine dyes are commonly classified into four categories of mono-, tri-, penta-, and heptamethine cyanine dyes. The spectral range for these dyes extends approximately between 450 and 750 nm in the electronic spectrum. In the past few years, many synthetic routes to benzothiazole cyanine dyes have been developed.
Monomethine cyanine dyes
Monomethine benzothiazole cyanine dyes typically absorb in the visible region (450–470 nm) of the electronic spectrum depending on the substituents attached to the benzothiazole core structure. These dyes are characterized by a narrow absorption peak and high fluorescence intensity. They are best known for their nucleic acid binding properties. The oldest method for the synthesis of the monomethine cyanine dyes involves condensation of an N-alkyl-2-(methylthio)benzothiazolium salt with another alkylated heterocycle with an activated methyl group [13]. This method was adopted for the synthesis of β-cyclodextrin functionalized benzothiazole cyanine dye 1 (Equation 1) [14]. β-Cyclodextrin possesses both a hydrophobic cavity and hydrophilic surface and is used for molecular recognition as a drug delivery agent in pharmaceutics [15]. The medicines containing vitamins are generally unstable to light, heat, and oxygen, whereas the formation of inclusion complexes of vitamins with β-cyclodextrin enhances the stability, solubility, and bioavailability of the drug [16]. Analysis of such inclusion interactions of vitamins are commonly conducted by means of spectrophotometric titration using external agents such as dyes as spectral probes. The incorporation of the dye molecule as host compound provides an option to easily recognize colorless guest molecules by direct titration. Compound 1 was used to study supramolecular interactions of β-cyclodextrin directly by visible spectroscopy, which is otherwise impossible due to an insufficient chromophore system.

The synthetic procedure shown above involves a major drawback of producing methyl mercaptan, which is a toxic pollutant. Researchers have overcome this problem by developing a new procedure that uses 2-iminobenzothiazoline instead of 2-methylthiobenzothiazolium salt [17]. This method can be used to synthesize symmetric and asymmetric cyanine dyes. In this procedure, cyanine dyes are prepared by melting 2-iminobenzothiazoline with quaternary heterocyclic salt containing 2- or 4-methyl groups, as depicted in Equation 2.

Deligeorgiev et al. described a novel procedure for the synthesis of benzothiazole monocyanines, which involves heating a sulfobetaine salt of N-alkylbenzothiazolium compound with the quaternary salt of a heterocyclic compound containing a reactive methyl group, as illustrated in Equation 3 [18]. These reactions are usually carried out without solvent by heating the mixture to approximately 150–200°C. An alternative route to less thermostable compounds involves heating a solution in polar solvent. This synthetic procedure is characterized by high yields and short reaction times.

Another approach towards the synthesis of monomethine cyanine dyes involves condensation of N-alkyl-2-methylbenzothiazolium salt with 2- or 4-chloropyridinium or quinolinium substrate in a basic medium [19]. This synthetic procedure has been used to synthesize dicationic and tricationic benzothiazole cyanine dyes (Equation 4). In this particular case, the increased cationic charge helps increase water solubility of the dye.

Several homodimeric benzothiazole cyanine dyes with polycationic charge on the overall cyanine dye have been published (Equation 5) [20].

Compounds 2–4 are structural analogs of the thiazole orange dye which is used extensively for the purpose of nucleic acid detection, flow cytometry, and gel electrophoresis [17]. Typically, monomethine cyanine dyes display almost negligible fluorescence in aqueous solutions but show multifold fluorescence enhancement upon binding to nucleic acids. This phenomenon can be used to detect nucleic acids in the solution up to certain nanomolar concentrations. The cationic monomethine cyanine dye molecules with planar aromatic rings typically intercalate within the two adjacent base pairs of DNA. It has been observed that increasing cationic charges on the dye molecule results in improved binding to the negatively charged nucleic acids. Homodimeric cyanine dye 5 interacts with nucleic acids by bis-intercalation and electrostatic interaction, which provides extra stabilization to the dye-nucleic acid complex [20]. Remarkably, cyanine dye 5 exhibits approximately a 40-nm difference between fluorescence maxima in the presence of single-stranded DNA and double-stranded DNA, which could make it useful to differentiate between both species in solution.
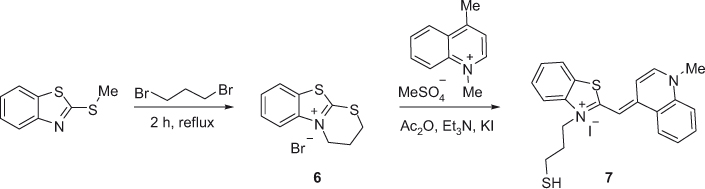
A novel method for the synthesis of monomeric asymmetric benzothiazole derivatives, such as 7 (Scheme 1), containing mercapto and thioacetyl substituents has been reported [21]. Compound 7 could be further modified as suggested by Ishiguro et al. by linking the mercapto group to oligonucleotides [22]. This method would facilitate in vitro transcription and gene expression studies. The synthetic methodology is depicted in Scheme 1. The preparation of the dye intermediate 6 was carried out by heating under reflux a mixture of 2-methylmercaptobenzothiazole with 1,3-dibromopropane for 2 h. Compound 6 was then allowed to react with 1,4-dimethylquinolinium iodide to furnish dye 7 in 95% overall yield.
A new method for the synthesis of benzothiazole monocyanines was developed making use of solid phase resin support [23]. The schematic representation of the solid phase synthesis is presented in Scheme 2. The first step involves the attachment of 2-mercaptobenzothiazole to the Merrifield resin. This resin-attached benzothiazole is then reacted in the next step with 4-methylbenzenesulfonate to yield N-methylbenzothiazolium salt 8. In the final step, condensation of N-methylbenzothiazolium salt 8 with carboxylic acid derivative of lepidine affords cyanine dye 9. Traceless cleavage of Merrifield resin is normally carried out during the course of the synthesis of the cyanine dye. The presence of a carboxylic acid side chain attached to the quinoline ring makes the dye available for post-synthetic modification. Attaching an amino acid to the carboxylic end group and modifying it with folic acid can be used to identify cancer cells that have a folacin acceptor on the cellular surface [23]. The most important feature of the solid phase synthesis is the elimination of the lengthy purification step of the conventional liquid phase method, which allows the use of synthesized dyes directly for biological screening.

Adding another vinyl unit to the central linking chain gives rise to trimethine cyanines. This class of dyes is discussed in the following section.
Trimethine cyanine dyes
Benzothiazole trimethine cyanine dyes absorb in the visible region (550–570 nm) of the electronic spectrum. The classical ortho ester method described by Koenig is the oldest known method for the synthesis of symmetric trimethine cyanine dyes [24]. The general synthetic procedure involves condensation of a quaternary benzothiazolium salt containing an activated methyl group with an ortho ester under basic conditions [25]. The ortho ester approach is illustrated in Scheme 3 by the synthesis of a dumbbell-type [60]-fullerene dimer containing a benzothiazole trimethine cyanine dye 11 [26]. The first step of the synthesis involves attachment of [60]-fullerene to the benzothiazole system, which is achieved by heating a solution of [60]-fullerene, sarcosine, and 5-formyl-2- methylbenzothiazole under a nitrogen atmosphere. The resulting compound 10 is then treated with dimethyl sulfate to generate a quaternary salt, which undergoes the coupling reaction with triethyl orthoformate in the next step to yield cyanine dye 11. Fullerene chemistry is a rapidly growing division of material science. C60 dyads have been the focus of much attention in recent years due to their applicability in artificial photosynthesis and molecular electronic devices [26]. The natural photosynthesis involves multistep electron transfer reactions achieved through various electron mediators present in the matrix which accomplish long lifetime of the final charge separated state [27]. Fullerenes have been widely employed as donor/acceptor molecules in dyads to accelerate photo-induced charge separation to mimic natural photosynthesis. The presence of two or more C60 units in the molecule could result in a longer lifetime of the final charge separated state, which is similar to the natural phenomenon.

A new class of phosphonate labeled trimethine benzothiazole cyanine dyes has been published using the ortho ester approach [28]. Synthetic methodology for the phosphonate labeled cyanine dye starts with the synthesis of compound 12 which is obtained by heating under reflux 2-methylbenzothiazole with diethyl 3-bromopropylphosphonate in acetonitrile (Scheme 4). Phosphonate salt 12 is then converted to cyanine dye 14 using triethyl orthoformate under basic conditions. Conversion of phosphonate salt to phosphonic acid salt is achieved by acidic hydrolysis, which is used in a further step to synthesize dye 13. The presence of phosphonic acid side chain improves the water solubility of the dye. In the deprotonated form, cyanine dyes 13 and 14 are pH sensitive calcium probes. Employment of the phosphonate group on the side chain makes these dyes potent fluorescent labels in complexation with biological systems [28].

Another approach to the synthesis of benzothiazole trimethine cyanine dyes involves the use of N,N-diphenylformamidine or iodoform as a condensing agent. This synthetic method also allows the synthesis of asymmetric cyanine dyes [29]. Scheme 5 shows the use of N,N-diphenylformamidine for the synthesis of benzothiazole trimethine cyanine dye 16. Alkylated benzoxazole derivative is first treated with N,N-diphenylformamidine in the presence of acetic anhydride to furnish half dye 15. Compound 15 in the next step is condensed with 2,3-dimethylbenozthiazolium iodide to yield final asymmetric dye 16. Different solvents can be used for the synthesis of asymmetric cyanine dyes using the N,N-diphenylformamidine approach. The reactions are usually carried out in acetic anhydride but can also be conducted in n-propanol or DMSO at high temperatures for several hours.

Park et al. developed novel triazacarbocyanine intramolecular charge transfer dye sensor 17 (Equation 6) [30]. This dye sensor shows reversible pH induced color switching effects. Here, a methoxy group acts as a donor group, whereas the nitro group acts as an acceptor group.

Equation 7 depicts the synthesis of trimethine cyanine dye with meso substitution in the trimethine chain. The meso methyl group undergoes condensation reaction with 9-julolidine carboxaldehyde to furnish benzothiazole derivative 18 [31].

The pentamethine cyanines are prepared using a similar approach. This class of dyes is discussed next.
Pentamethine cyanine dyes
Pentamethine cyanine dyes generally absorb in the visible region (650–670 nm) but fluoresce in the near-infrared region (690–710 nm) of the electronic spectrum. The classical synthetic procedure involves use of bis(phenylimine) hydrochloride of malonaldehyde under basic conditions, as exemplified in Equation 8 [32, 33].

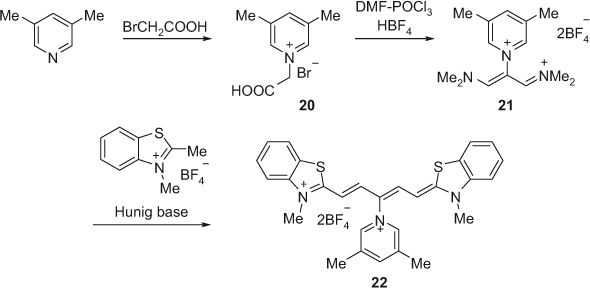
Modification of the trimethine chain in the malonaldehyde-derived reagent for the use in synthesis of γ-substituted pentamethine cyanine dye has been reported, as shown in Scheme 6 [34]. The new γ-substituted pentamethine cyanine dyes were synthesized using a three-step procedure. The first step involves synthesis of N-pyridinium acetic acid salt 20 by reaction between 3,5-dimethylpyridine and bromoacetic acid. In the second step, compound 20 was formylated in the presence of N,N-dimethylformamide and phosphorus oxychloride to obtain compound 21. The synthesis of cyanine dye 22 was carried out by condensation reaction between compound 21 and 2,3-dimethyl benzothiazole tetrafluoroborate in the presence of sterically hindered Hunig base. The presence of the pyridine ring in the pentamethine chain makes the dye more rigid and the cationic charge makes it water soluble. The presence of the pyridinium moiety affects electron density distribution of the dye molecule.
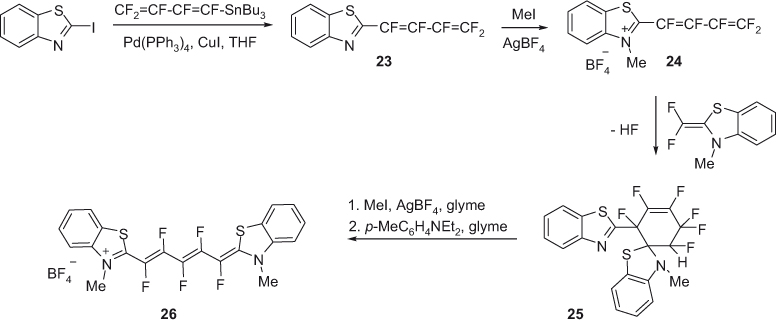
Benzothiazole cyanine dyes with fluorine substituted pentamethine chain have been reported (Scheme 7) [35]. The Stille reaction was employed in the first step for coupling of 2-iodobenzothiazole with a fluorinated tin reagent in the presence of copper iodide and tetrakis(triphenylphosphine) palladium(0) to afford compound 23. This product was alkylated by reaction with methyl iodide in the presence of silver tetrafluoroborate to obtain compound 24, which on subsequent reaction with 2-fluoromethylbenzothiazole methylene base was transformed into cyclic compound 25. Alkylation of 25 followed by reaction with p-dimethylaminotoluidine furnished pentamethine cyanine dye 26. It is noteworthy that the electronegative fluorine atoms in the pentamethine chain act as electron donating substituents resulting in the shift of absorption maximum for compound 26 (691 nm) of 41 nm relative to the non-fluorinated dye.
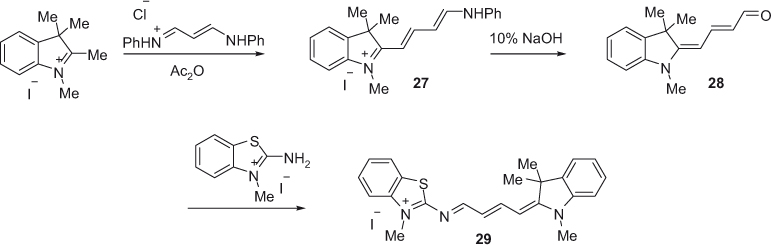
The synthesis of asymmetric pentamethine benzothiazole cyanine dyes can be accomplished utilizing an aldehyde analog of the benzothiazole moiety [36]. Meguellati et al. adapted the aldehyde strategy to synthesize the imino pentamethine cyanine dye, as depicted in Scheme 8. These dyes remain stable in the solid state, whereas in solution within an hour aldehyde and imine are regenerated [37]. Compound 29 was synthesized by reacting malonaldehyde bis(phenylimine) hydrochloride with a 1,2,3,3-tetramethylindolenine iodide salt. Basic hydrolysis of the activated hemicyanine derivative 27 in sodium hydroxide solution afforded compound 28 in excellent yield. In the last step, compound 28 was treated with N-methyl-2-aminobenzothiazolium salt to yield cyanine dye 29. The cyanine dye 29 is accessible through reversible and thermodynamically controlled reaction of amine and aldehyde.
A series of benzothiazole fluorophores with substituted cyclohexene and cyclopentene in the pentamethine chain has been described [38]. These compounds were studied as fluorescent probes for nucleic acid and proteins. It is believed that they act as nucleic acid groove binders. Synthesis of pentamethine cyanine dyes 34 and 35 containing a cyclopentene ring in the pentmethine chain was accomplished by condensing 2-methylcylcopentane-1,3-diones 30, 31 with quaternary salts of 2-methylbenzothiazolium 32, 33 at 210°C in the presence of triethylamine (Equation 9). Compound 35 exhibits a 15-fold increase and compound 34 displays an 8-fold increase in the fluorescence intensity in the presence of DNA. Cyanine dyes 34 and 35 can be further modified to increase their binding interactions with DNA.

Synthesis of pentamethine dyes with a cyclohexene ring in the pentamethine chain was achieved by condensation of quaternary salts of benzothiazole containing an active methyl group with 1,5-dimethoxy-1,4-cyclohexadienes or with 1,3-diethoxy-5,5-dimethyl- or 1,3-diethoxy-2,5,5-trimethyl-1,3-cyclohexadienes either by melting the two substrates or by heating in benzonitrile at 120–130°C, as illustrated in Equation 10. Compound 36 displays an 8-fold increase in the fluorescence intensity in the presence of RNA, but almost negligible fluorescence changes in the presence of DNA. Remarkably, compound 36 exhibits an approximately 26-fold increase in the fluorescence intensity in the presence of bovine serum albumin [38]. It has been suggested that compound 36 could be used as a potential fluorescent probe for other proteins.

Heptamethine cyanine dyes
Heptamethine cyanine dyes typically absorb and fluoresce in the near-infrared region (750–1100 nm) of the electronic spectrum. The general synthetic method for the synthesis of benzothiazole heptacyanines with a flexible polymethine chain involves use of glutaconaldehyde dianil monohydrochloride as a precursor to the heptamethine linker [39]. As shown in Equation 11, a number of benzothiazole heptamethine derivatives with different substituents at the benzothiazole nitrogen atom have been synthesized [40, 41].

Synthesis of another class of benzothiazole heptamethine dyes containing five- or six-membered cyclic systems as a part of the heptamethine linker makes use of the Vilsmeier-Haack reagents [42, 43]. The presence of a carbocyclic ring in the heptamethine chain makes dye more rigid, which helps to increase fluorescence quantum yield and decrease aggregation of the dye in solution. Vilsmeier-Haack reagents are derived from cyclopentanone or cyclohexanone in a two-step procedure, as shown in Equation 12.

The general methodology for the synthesis of heptamethine cyanines with a rigid polymethine chain involves the condensation of Vilsmeier-Haack reagents 38 or 39 with a heterocycle containing an activated methyl group in ethanol in the presence of sodium acetate as catalyst [44]. Another approach for the synthesis of heptamethine cyanine dyes involves heating under reflux a quaternary salt of benzothiazole and bis-aldehyde in butanol/benzene solvent mixture with continuous removal of the water from the reaction mixture using a Dean-Stark apparatus [45]. The major advantage of using this method of synthesis is the elimination of a catalyst and the formation of final dye 40 in high yield (Equation 13). Benzothiazole cyanine dyes similar to compound 40 can be additionally functionalized at the meso position by reaction with various nucleophiles. The products have been examined as sensitizers for zinc oxide solar cells [43].

Applications of benzothiazole cyanine dyes
Several applications of cyanine dyes have already been mentioned above. After the discovery of cyanine dyes, their first uses were limited to photographic sensitizers [46]. Other than photography, cyanine dyes now also find applications in several other fields such as recording media [47], laser materials [48], solar cells [49–51], and semiconductors [52, 53].
Cyanine dyes have often been regarded as good silver halide photography sensitizers. A silver halide solution possesses limited sensitivity up to 550 nm but when it is doped with dye its sensitivity extends up to 650 nm and sometimes even into near-infrared region of the electronic spectrum. Sensitizer dyes show a bathochromic shift when added to the silver halide solution by up to 20–80 nm. Benzothiazole dyes 41 and 42 are typical examples of photographic sensitizers [54].

Another important application of benzothiazole dyes is their use in recording media. At the beginning, most available CD-Rs used indolenine based cyanines as the recording dyes. With the discovery of the modern DVD-R system, which requires a shorter wavelength laser beam, benzothiazole trimethine cyanine dyes were found to be better dyes to obtain appropriate reflection and modulation of the DVD-R. An important advantage of using trimethine cyanine dyes is their good chemical stability and photostability. Compound 43 is an example of a practical benzothiazole cyanine dye that is used for the purpose of recording media [55].

Dye-sensitized solar cells have attracted tremendous attention within the past decade; this includes cyanine dyes substituted with heavy metal based complexes as sensitizers in solar cells. Advantages of using cyanine dyes as solar cell sensitizers include cost efficiency and easy recycling of solar cells. Several benzothiazole cyanine dyes such as compound 44 have been synthesized and studied for their efficiency towards solar sensitization [56].

References
[1] Williams, C. Cyanine dyes. Trans. R. Soc. Edinb. 1856, 21, 377.Search in Google Scholar
[2] Hamer, F. Mainly introductory. In Chemistry of Heterocyclic Compounds; Hamer, Frances M., Ed. John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2008; pp. 1–31.Search in Google Scholar
[3] Armitage, B. Cyanine dye-nucleic acid interactions. In Heterocyclic Polymethine Dyes. Strekowski, L., Ed. Springer: Berlin, 2008; Vol. 14, pp. 11–29.10.1007/7081_2007_109Search in Google Scholar
[4] Katritzky, A.; Fan, W.-Q.; Li, Q.-L. Bridged cyanine dyes. Part 1. 1-(N-methyl-4-pyridinio)-3-(N-methylpyridylene)cyclopenta-1,4-dienes. J. Heterocycl. Chem. 1988, 25, 1311–1314.Search in Google Scholar
[5] Behera, G.; Behera, P.; Mishra B. Cyanine dyes: self aggregation and behaviour in surfactants. A review. J. Surface Sci. Technol. 2007, 23, 1–31.Search in Google Scholar
[6] Kovalska, V.; Volkova, K.; Losytskyy, M.; Tolmachev, O.; Balanda, A.; Yarmoluk, S. 6,6′-Disubstituted benzothiazole trimethine cyanines – new fluorescent dyes for DNA detection. Spectrochim. Acta A Mol. Biomol. Spectr. 2006, 65, 271–277.Search in Google Scholar
[7] Kaloyanova, S.; Trusova, V.; Gorbenko, G.; Deligeorgiev, T. Synthesis and fluorescence characteristics of novel asymmetric cyanine dyes for DNA detection. J. Photochem. Photobiol. A Chem. 2011, 217, 147–156.Search in Google Scholar
[8] Klunk, W.; Engler, H.; Nordberg, A.; Wang, Y.; Blomqvist, G.; Holt, D.; Bergström, M.; Savitcheva, I.; Huang, G.-F.; Estrada, S.; et al. Imaging brain amyloid in Alzheimer’s disease with Pittsburgh compound-B. Ann. Neurol. 2004, 55, 306–319.Search in Google Scholar
[9] Mills, W. LIV. – The cyanine dyes. Part IV. Cyanine dyes of the benzothiazole series. J. Chem. Soc. Trans. 1922, 121, 455–466.Search in Google Scholar
[10] Ogul’chansky, T.; Losytskyy, M.; Kovalska, V.; Yashchuk, V.; Yarmoluk, S. Interactions of cyanine dyes with nucleic acids. XXIV. Aggregation of monomethine cyanine dyes in presence of DNA and its manifestation in absorption and fluorescence spectra. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2001, 57, 1525–1532.Search in Google Scholar
[11] Volkova, K.; Kovalska, V.; Balanda, A.; Losytskyy, M.; Golub, A.; Vermeij, R.; Subramaniam, V.; Tolmachev, O.; Yarmoluk, S. Specific fluorescent detection of fibrillar α-synuclein using mono- and trimethine cyanine dyes. Bioorg. Med. Chem. 2008, 16, 1452–1459.Search in Google Scholar
[12] Deligeorgiev, T.; Kaloyanova, S.; Vaquero J. Intercalating cyanine dyes for nucleic acid detection. Recent Patents Mater. Sci. 2009, 2, 1–26.10.2174/1874464810902010001Search in Google Scholar
[13] Brooker, L.; Keyes, G.; Williams, W. Color and constitution. V. The absorption of unsymmetrical cyanines. Resonance as a basis for a classification of dyes. J. Am. Chem. Soc. 1942, 64, 199–210.Search in Google Scholar
[14] Zhao, J.-L.; Lv, Y.; Ren, H.-J.; Sun, W.; Liu, Q.; Fu, Y.-L.; Wang, L.-Y. Synthesis, spectral properties of cyanine dyes-β-cyclodextrin and their application as the supramolecular host with spectroscopic probe. Dyes Pigments 2013, 96, 180–188.Search in Google Scholar
[15] Del Valle, E. Cyclodextrins and their uses: a review. Proc. Biochem. 2004, 39, 1033–1046.Search in Google Scholar
[16] Zhu, X.; Sun, J.; Wu, J. Study on the inclusion interactions of β-cyclodextrin and its derivative with dyes by spectrofluorimetry and its analytical application. Talanta 2007, 72, 237–242.Search in Google Scholar
[17] Deligeorgiev, T.; Gadjev, N.; Drexhage, K.-H.; Sabnis, R. Preparation of intercalating dye thiazole orange and derivatives. Dyes Pigments 1995, 29, 315–322.Search in Google Scholar
[18] Deligeorgiev, T.; Zaneva, D.; Katerinopoulos, H.; Kolev, V. A novel method for the preparation of monomethine cyanine dyes. Dyes Pigments 1999, 41, 49–54.Search in Google Scholar
[19] Deligeorgiev, T.; Zaneva, D.; Kim, S.; Sabnis, R. Preparation of monomethine cyanine dyes for nucleic acid detection. Dyes Pigments 1998, 37, 205–211.Search in Google Scholar
[20] Timtcheva, I.; Maximova, V.; Deligeorgiev, T.; Gadjev, N.; Drexhage, K.; Petkova, I. Homodimeric monomethine cyanine dyes as fluorescent probes of biopolymers. J. Photochem. Photobiol. B Biol. 2000, 58, 130–135.Search in Google Scholar
[21] Deligeorgiev, T.; Gadjev, N.; Vasilev, A.; Drexhage, K.-H.; Yarmoluk, S. Synthesis of novel monomeric cyanine dyes containing mercapto and thioacetyl substituents for nucleic acid detection. Dyes Pigments 2006, 70, 185–191.Search in Google Scholar
[22] Ishiguro, T.; Saitoh, J.; Yawata, H.; Otsuka, M.; Inoue, T.; Sugiura, Y. Fluorescence detection of specific sequence of nucleic acids by oxazole yellow-linked oligonucleotides. Homogeneous quantitative monitoring of in vitro transcription. Nucleic Acids Res. 1996, 24, 4992–4997.Search in Google Scholar
[23] Fei, X.; Yang, S.; Zhang, B.; Liu, Z.; Gu, Y. Solid-phase synthesis and modification of thiazole orange and its derivatives and their spectral properties. J. Comb. Chem. 2007, 9, 943–950.Search in Google Scholar
[24] Koenig, W. Über Indolenino-cyanine. Ber. Dtsch. Chem. Ges. 1924, 685.10.1002/cber.19240570420Search in Google Scholar
[25] Kabatc, J.; Pączkowski, J. Three-cationic carbocyanine dyes as sensitizers in very efficient photo-initiating systems for multifunctional monomer polymerization. J. Polymer Sci. A Polymer Chem. 2009, 47, 4636–4654.Search in Google Scholar
[26] Xiao, S.; Xu, J.-H.; Li, Y.-S.; Du, C.-M.; Li, Y.-L.; Jiang, L.; Zhu, D. Preparation and characterization of a novel dumbbell-type [60] fullerene dimer containing a cyanine dye. New J. Chem. 2001, 25, 1610–1612.Search in Google Scholar
[27] Ohkubo, K.; Kotani, H.; Shao, J.; Ou, Z.; Kadish, K.; Li, G.; Pandey, R.; Fujitsuka, M.; Ito, O.; Imahori, H.; et al. Production of an ultra-long-lived charge-separated state in a zinc chlorin-C60 dyad by one-step photo-induced electron transfer. Angew. Chem. Int. Ed. 2004, 43, 853–856.Search in Google Scholar
[28] Mazières, M.; Duprat, C.; Bellan, J.; Wolf, J. Synthesis and characterisation of new phosphonate labelled cyanines. Dyes Pigments 2007, 74, 404–409.Search in Google Scholar
[29] Shi, Q.-Q.; Sun, R.; Ge, J.-F.; Xu, Q.-F.; Li, N.-J.; Lu, J.-M. A comparative study of symmetrical and unsymmetrical trimethine cyanine dyes bearing benzoxazolyl and benzothiazolyl groups. Dyes Pigments 2012, 93, 1506–1511.Search in Google Scholar
[30] Park, J.-Y.; Gwon, S.-Y.; Yoon, N.-S.; Son, Y.-A.; Kim, S.-H. Novel triazacarbocyanine dye sensor synthesis: pH switching effect. Fibers Polym. 2011, 12, 976–978.Search in Google Scholar
[31] Mitekura, H.; No, T.; Suzuki, K.; Satake, K.; Kimura, M. Spectroscopic properties of meso-substituted cyanine dyes: evidences for intramolecular charge transfer from a julolidine moiety as a meso-substituent to the cyanine chromophore. Dyes Pigments 2002, 54, 113–120.Search in Google Scholar
[32] Renikuntla, B.; Rose, H.; Eldo, J.; Waggoner, A.; Armitage, B. Improved photostability and fluorescence properties through polyfluorination of a cyanine dye. Org. Lett. 2004, 6, 909–912.Search in Google Scholar
[33] Kaur, G.; Zhan, W.; Wang, C.; Barnhill, H.; Tian, H.; Wang, Q. Crosslinking of viral nanoparticles with “clickable” fluorescent crosslinkers at the interface. Sci. China Chem. 2010, 53, 1287.Search in Google Scholar
[34] Mehranpour, A.; Hashemnia, S.; Maghamifar, R. Synthesis and characterization of new γ-substituted pentamethine cyanine dyes. Synth. Commun. 2010, 40, 3594–3602.Search in Google Scholar
[35] Yagupolskii, L.; Chernega, О.; Kondratenko, N.; Chernega, A.; Vlasenko, Y.; Nedelkov, R.; Yagupolskii, Y. Synthesis of the first representative of dicarbothiacyanine dyes with completely fluorinated polymethine chain. J. Fluorine Chem. 2010, 131, 165–171.Search in Google Scholar
[36] Nesterenko, Y.; Briks, Y.; Kachkovskii, A.; Tolmachev, A. Synthesis and spectral properties of pyran-based heterocyclic polyene vinylogs. Khim. Geterotsikl. Soedin. 1990, 2, 252–255.Search in Google Scholar
[37] Meguellati, K.; Spichty, M.; Ladame, S. Reversible synthesis and characterization of dynamic imino analogues of trimethine and pentamethine cyanine dyes. Org. Lett. 2009, 11, 1123–1126.Search in Google Scholar
[38] Losytskyy, M.; Volkova, K.; Kovalska, V.; Makovenko, I.; Slominskii, Y.; Tolmachev, O.; Yarmoluk, S. Fluorescent properties of pentamethine cyanine dyes with cyclopentene and cyclohexene group in presence of biological molecules. J. Fluoresc. 2005, 15, 849–857.Search in Google Scholar
[39] Kharitonova, A. Cyanine dyes. J. Org. Chem. USSR 1992, 28, 2159–2164.Search in Google Scholar
[40] Brooker, L.; Smith, L. 1,1′-Diaralkyl-4,4′-carbocyanine salts. Patent A.l. 2,233,511 1941.Search in Google Scholar
[41] Vompe, A.; Ivanova, L.; Meskhi, L.; Monich, N.; Raikhina, R. Reactions of polymethine dyes. I. Synthesis of pseudobases of polymethine dyes and their reactions. Zh. Org. Khim. 1985, 21, 584–594.Search in Google Scholar
[42] Ramos, S.; Santos, P.; Reis, L.; Almeida, P. Some new symmetric rigidified triheterocyclic heptamethinecyanine dyes absorbing in the near infrared. Dyes Pigments 2002, 53, 143–152.Search in Google Scholar
[43] Matsui, M.; Hashimoto, Y.; Funabiki, K.; Jin, J.-Y.; Yoshida, T.; Minoura, H. Application of near-infrared absorbing heptamethine cyanine dyes as sensitizers for zinc oxide solar cell. Synth. Metals 2005, 148, 147–153.Search in Google Scholar
[44] Makin, V. Cyanine dyes. J. Org. Chem. USSR 1977, 13, 2440–2441.Search in Google Scholar
[45] Narayanan, N.; Strekowski, L.; Lipowska, M.; Patonay, G. A new method for the synthesis of heptamethine cyanine dyes: synthesis of new near-infrared labels. J. Org. Chem. 1997, 62, 9387.Search in Google Scholar
[46] Tani, T. The present status and future prospects of silver halide photography. J. Imag. Sci. Technol. 1995, 39, 31–40.Search in Google Scholar
[47] Dai, Z.; Qun, L.; Peng, B. Synthesis and characterization of some stabilized indocarbocyanine dye complex colorants for optical recording media. Dyes Pigments 1998, 36, 243–248.Search in Google Scholar
[48] Reisfeld, R.; Weiss, A.; Saraidarov, T.; Yariv, E.; Ishchenko, A. Solid-state lasers based on inorganic-organic hybrid materials obtained by combined sol-gel polymer technology. Polymers Adv. Technol. 2004, 15, 291–301.Search in Google Scholar
[49] Zhang, Q.; Dandeneau, C.; Zhou, X.; Cao, G. ZnO nanostructures for dye-sensitized solar cells. Adv. Mater. 2009, 21, 4087–4108.Search in Google Scholar
[50] Liang, M.; Xu, W.; Cai, F.; Chen, P.; Peng, B.; Chen, J.; Li, Z. New triphenylamine-based organic dyes for efficient dye-sensitized solar cells. J. Phys. Chem. C 2007, 111, 4465–4472.Search in Google Scholar
[51] Ehret, A.; Stuhl, L.; Spitler, M. Spectral sensitization of TiO2 nanocrystalline electrodes with aggregated cyanine dyes. J. Phys. Chem. B 2001, 105, 9960–9965.Search in Google Scholar
[52] Nasr, C.; Hotchandani, S.; Kamat, P.; Das, S.; Thomas, K.; George, M. Electrochemical and photoelectrochemical properties of monoaza-15-crown ether linked cyanine dyes: photosensitization of nanocrystalline SnO2 films. Langmuir 1995, 11, 1777–1783.Search in Google Scholar
[53] Tang, X.; Gan, F. Optical storage performance of cyanine-doped ormosil film. Proc. SPIE Int. Soc. Opt. Eng. 1997, 3136, 210–214.Search in Google Scholar
[54] Li, Q.; Lin, G.-L.; Peng B.-X.; Li, Z.-X. Synthesis, characterization and photographic properties of some new styryl cyanine dyes. Dyes Pigments 1998, 38, 211–218.Search in Google Scholar
[55] Tian, H.; Meng, F. Cyanine dyes for solar cells and optical data storage. In Functional Dyes; Sung-Hoon, K., Ed. Elsevier Science: Amsterdam, 2006, pp. 47–84.10.1016/B978-044452176-7/50003-6Search in Google Scholar
[56] Ehret, A.; Stuhl, L.; Spitler, M. Variation of carboxylate-functionalized cyanine dyes to produce efficient spectral sensitization of nanocrystalline solar cells. Electrochim. Acta 2000, 45, 4553–4557.Search in Google Scholar
©2013 by Walter de Gruyter Berlin Boston
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Articles in the same Issue
- Masthead
- Masthead
- Reviews
- Synthesis and applications of benzothiazole containing cyanine dyes
- Synthesis and chemistry of structurally unique hexasubstituted pyrazolines
- Research Articles
- Synthesis and characterization of heteroarylthio derivatives of 5,17-di-tert-butyl-11,23-diamido-25, 27-diprotected calix[4]arene
- New heterocyclic chalcones. Part 6. Synthesis and cytotoxic activities of 5- or 6-(3-aryl- 2-propenoyl)-2(3H)-benzoxazolones
- Novel benzofuran derivatives: synthesis and antitumor activity
- Synthesis, characterization and in vitro antimicrobial assessment of some novel 4H-1, 4-benzothiazines and their sulfone derivatives
- Efficient synthesis, X-ray diffraction study and antimicrobial activity of some novel thiazolidin-4-ones and perhydro-1,3-thiazin-4-ones
- A simple and efficient synthesis of novel naphthyridine-1-H-pyrazole-4-carboxylic acid esters/carbaldehydes using Vilsmeier-Haack reagent
- Melamine-formaldehyde resin supported H+-catalyzed three-component synthesis of 1,8-dioxo-decahydroacridine derivatives in water and under solvent-free conditions
- A simple and efficient procedure for synthesis of symmetrical bis(4-amino-4H-1,2,4-triazole-5-thiols)
- Poly(ethylene)glycol/AlCl3 as a new and efficient system for multicomponent Biginelli-type synthesis of pyrimidinone derivatives
- Synthesis, crystal structure, and bioactivity of N-dichloroacetyl diazabicyclo compounds
Articles in the same Issue
- Masthead
- Masthead
- Reviews
- Synthesis and applications of benzothiazole containing cyanine dyes
- Synthesis and chemistry of structurally unique hexasubstituted pyrazolines
- Research Articles
- Synthesis and characterization of heteroarylthio derivatives of 5,17-di-tert-butyl-11,23-diamido-25, 27-diprotected calix[4]arene
- New heterocyclic chalcones. Part 6. Synthesis and cytotoxic activities of 5- or 6-(3-aryl- 2-propenoyl)-2(3H)-benzoxazolones
- Novel benzofuran derivatives: synthesis and antitumor activity
- Synthesis, characterization and in vitro antimicrobial assessment of some novel 4H-1, 4-benzothiazines and their sulfone derivatives
- Efficient synthesis, X-ray diffraction study and antimicrobial activity of some novel thiazolidin-4-ones and perhydro-1,3-thiazin-4-ones
- A simple and efficient synthesis of novel naphthyridine-1-H-pyrazole-4-carboxylic acid esters/carbaldehydes using Vilsmeier-Haack reagent
- Melamine-formaldehyde resin supported H+-catalyzed three-component synthesis of 1,8-dioxo-decahydroacridine derivatives in water and under solvent-free conditions
- A simple and efficient procedure for synthesis of symmetrical bis(4-amino-4H-1,2,4-triazole-5-thiols)
- Poly(ethylene)glycol/AlCl3 as a new and efficient system for multicomponent Biginelli-type synthesis of pyrimidinone derivatives
- Synthesis, crystal structure, and bioactivity of N-dichloroacetyl diazabicyclo compounds

