Abstract
Objectives
The aims of this retrospective study were to evaluate the clinical applicability of the latest International Society for the Study of Vulvovaginal Disease (ISSVD) and International Federation for Cervical Pathology and Colposcopy (IFCPC) terminology for vulvar diseases, and to explore a new evaluation flow to optimize decision-making on diagnosis.
Methods
A total of 1,068 patients with 5,340 qualified vulvar images were evaluated by observers using 2011 ISSVD and 2011 IFCPC terminology systems. The sensitivity, specificity, positive predictive value, negative predictive value, Youden Index and Overall Diagnostic Value (ODV) were calculated for each finding in the two systems. Then the disease diagnosis order and a diagnosis flow draft (DFD) were obtained.
Results
A total of 15 kinds of vulvar diseases were diagnosed. The proportion of patients accompanied with cervical or vaginal intraepithelial neoplasia was highest (83.3 %) in vulvar Paget’s disease group (p<0.001). Total area of lesions was larger in vulvar Paget’s disease, lichen simplex chronicus and lichen sclerosus group (p<0.001). Among the top five findings of ODV, some findings inferred several (≥6) kinds of diseases, while some findings only exist in a certain disease. When the DFD was used, the agreement between the initial impression and histopathology diagnosis was 68.8 %, higher than those when ISSVD an IFCPC terminology systems used (p=0.028), and it didn’t change with the experience of the observer (p=0.178).
Conclusions
Based on the findings in ISSVD and IFCPC terminology systems, we explored a DFD for observers with different experience on the detection of vulvar disease.
Introduction
Vulvar disease represents an area of particular diagnostic difficulty for clinicians, since it lies at the cross-section of gynecology, dermatology, and women’s health, and the diagnosis of vulvar disease depends on the visual sense and experience of the observers [1]. Clinicians from multiple specialties, such as gynecologists, dermatologists, pathologists, physical therapists, psychologists, and sex therapists, cannot reach a consensus on the appropriate names or classification of vulvar dermatological disorders, which may be attributable to inflammatory dermatoses, infections, neoplasms, hormonal changes, neuropathies, psychologic comorbidities, or multifactor. In addition, despite serving as the primary provider for patients with vulvar disease, both gynecologists and dermatologists typically receive limited training on vulvar diseases, which is an underserved category and not commonly a focus within training programs [2].
To define and promulgate an international nomenclature for vulvar disease, the International Society for the Study of Vulvovaginal Disease (ISSVD) was founded in 1971 and revised vulvar disease terminology and classification system constantly on the basis of consensus among experts in multiple disciplines [3]. In 2006, the ISSVD Classification of Vulvar Dermatoses was formulated based on the most common histological patterns with standard dermatological terminology [4]. In 2011, the ISSVD published a new terminology and classification system to assist clinicians in different disciplines in formulating the description of a lesion and clustering diseases with similar clinical presentations into groups [5]. However, the terminology for colposcopic patterns of the vulva was not included in the 2011 ISSVD system. Many female patients with vulvar dermatological disorders are referred to colposcopy clinics for care, but reports of vulvar findings have been inconsistent due to the lack of colposcopic terminology for vulvar patterns [6, 7]. The International Federation for Cervical Pathology and Colposcopy (IFCPC), founded in 1972, provides standardized interpretations of colposcopic findings and organizes a comprehensive classification by revising the versions in 1975, 1990, and 2002 [8]. In 2011, the IFCPC presented vulvar (including anal) clinical and colposcopic terminology, which was based on the new ISSVD vulvar terminology and added other specific terminology for colposcopic examination at the 2011 World Congress in Rio de Janeiro [6]. However, owing to limited reports, clinical evaluation studies on the applicability of the latest 2011 ISSVD and 2011 IFCPC terminology for vulvar diseases are extremely scant. An operable and relatively simplified diagnostic approach, which can combine the characteristics of the two terminology systems, is required [9].
In this study, we utilized the latest 2011 ISSVD and 2011 IFCPC terminology systems to describe the appearance impression of vulvar diseases and explored a diagnosis flow draft (DFD) based on the two systems. Finally, we evaluated the clinical application of the diagnosis flow draft and provided a new idea for the diagnosis of vulvar diseases before invasive detection.
Materials and methods
Study design and population
In this retrospective study, from January 2020 to December 2022, the population of patients with clinically suspected vulvar disease who underwent colposcopy-guided punch biopsy of the vulva at the Obstetrics and Gynaecology Hospital of Fudan University were enrolled, with a total of 2,724 patients. The exclusion criteria were as follows: (1) patients with unqualified vulvar images; (2) patients with undefined histopathological diagnosis; (3) patients with incomplete history records. Finally, a total of 1,068 patients with 5,340 qualified vulvar images and defined histopathological diagnosis were evaluated for the study. All patients signed informed consent forms before the study, and Institutional Review Board approval was obtained properly (2023–25).
In this study, the 5,340 qualified vulvar images were evaluated by observers using different terminology systems. The observers, who had qualifications in colposcopy and diagnosis of vulva diseases after training, were blinded to the vulvar histological diagnosis, and divided into five groups (Group A, B, C, D, E) based on work experience in colposcopy. All the data were documented in the hospital database.
Detection of vulvar disease using ISSVD terminology and classification system
Three observers (Group A) with more than 10 years of experience in colposcopy evaluated all the vulvar images in five steps, which were listed in the 2011 ISSVD terminology and classification [5]. Step 1. Lesions were defined by choosing one or more of the following nouns: blister, bulla (>0.5 cm), cyst, edema, erosion, excoriation, fissure, lesion, macule (<1.0 cm), nodule (>1.0 cm), papule (<1.0 cm), patch (>1.0 cm), plaque (>1.0 cm), pustule, rash, ulcer, vesicle (<0.5 cm), eczema, surface disruption, and lichenification. Step 2. Choose appropriate adjectives to modify the noun(s) previously chosen: color, surface, margination, and configuration. Step 3. Formulate a list of differential diagnoses. Step 4. Reduce the number of diagnoses in the list of differential diagnoses. Step 5. Confirm a clinical diagnosis. If there was disagreement among the observers, a consensus was reached through discussion. According to the 2013 Lower Anogenital Squamous Terminology, vulvar low-grade squamous intraepithelial lesion (LSIL) is the equivalent of vulvar intraepithelial neoplasia 1 (VIN 1), and vulvar high-grade squamous intraepithelial lesion (HSIL) includes VIN 2 and VIN 3 [3]. The terms LSIL and HSIL were used to describe human papillomavirus-associated squamous lesions of vulva in this study.
Detection of vulvar disease using IFCPC terminology system
Another three observers (Group B) with more than 10 years of experience in colposcopy evaluated all the vulvar images based on six sections, which were included in the 2011 IFCPC terminology system [6]. Section 1. Basic definitions: various anatomical structures and composition (squamous epithelium: hairy/non-hairy, mucosa). Section 2. Normal findings included micropapillomatosis, sebaceous glands (fordyce spots), and vestibular redness. Section 3. Abnormal findings including size in centimeters and location: macule (<1.5 cm), patch (>1.5 cm), papule (<1.5 cm), plaque (>1.5 cm), nodule (>1.5 cm), cyst, vesicle (<0.5 cm), bulla (>0.5 cm), pustule, lesion color, eczema, lichenification, excoriation, purpura, scarring, ulcer, erosion, fissure, wart. Section 4. Miscellaneous findings: trauma and malformation. Section 5. Suspicion of malignancy: gross neoplasm, ulceration, necrosis, bleeding, exophytic lesion, and hyperkeratosis with or without white, gray, red, or brown discoloration. Section 6. Abnormal colposcopic/other magnification findings: (thin or dense) acetowhite epithelium, (fine or coarse) punctuation, (fine or coarse) mosaic, atypical vessels, surface irregularities. Finally, the observers recorded the description of the vulvar appearance findings and made an initial impression through discussion.
Developing a DFD
Calculate the diagnostic and predictive parameters of each findings for the disease
The sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and Youden Index (YI, sensitivity+specificity−1) were used to assess the accuracy and predictability of findings in the ISSVD and IFCPC systems, respectively. Some of the findings in the two systems overlap, some are different, and some have the same name but different definitions, such as the spot and papule (the diameters are different in the two systems). We also considered and analyzed some new features such as exudation, atrophy, thickening, and lesion texture (softness, medium hardness, hardness, and fragility), although they were not included in the ISSVD or IFCPC system.
Calculate the overall diagnostic value (ODV) of findings and obtain the top five in diseases
According to the sensitivity, specificity, PPV, NPV, and YI, all findings were ranked from high to low. The first, second, and third top ranking findings were scored as three, two and one, respectively. Sensitivity, specificity, PPV, NPV, and YI were given different coefficients, and the ODV of each finding in the ISSVD and IFCPC systems was calculated as follows: ODV=ranking score (in sensitivity)*1+ranking score (in specificity)*1+ ranking score (in PPV)*1+ranking score (in NPV)*1+ranking score (in YI)*2. Finally, for each disease, the top five ODV findings were obtained for the two systems.
Obtain the disease diagnosis order for each finding
Based on the above findings, which were considered to have high diagnostic value, vulvar diseases were ranked from high to low based on the ODV. A possible disease diagnosis order can be obtained for each finding in the ISSVD and IFCPC systems.
Draw a DFD based on the results of the two systems
Based on the possible disease diagnosis order of each finding in the ISSVD and IFCPC systems, we drew a DFD. The diseases in the diagnosis order at the first, second, third, and fourth positions are marked with numbers and blue arrows of different thicknesses and brightness. The higher the sort, the thicker and darker is the arrow. The flow paths in the ISSVD are marked in yellow, those in the IFCPC systems are marked in green, and the overlapping paths are marked in purple.
Assessment of the DFD on clinical application
Based on the DFD, all the vulvar images were evaluated by another nine observers respectively, who were divided into three groups according to their work experience in colposcopy: those with more than 10 years (3 observers, Group C), those with 5–10 years (3 observers, Group D), and those with less than 5 years (3 observers, Group E). The agreement between the initial impression and histopathological diagnosis was compared among groups A, B, and C, and among Groups C, D, and E, respectively.
Statistical analysis
Statistical analysis was performed using Statistical Package for Social Sciences Version 29.0 Software (SPSS 29.0). Age data were expressed as mean±standard deviation (SD). If the variance was homogeneous, a one-way analysis of variance was used to compare the differences in quantitative variables. If the variance was not homogeneous, Welch’s test was used. The chi-squared test or Kruskal–Wallis test was used to compare the differences between categorical variables. A p-value less than 0.05 was considered to indicate a statistically significant result.
Results
Analysis of basic characteristics of patients
A total of 1,068 patients with vulvar diseases diagnosed by histopathology at the Obstetrics and Gynecology Hospital of Fudan University between January 2020 and December 2022 were included in this study. There were 15 types of vulvar diseases with the proportions of patients as follows: squamous cell carcinoma (SCC, 14/1,068, 1.3 %), HSIL (78/1,068, 7.3 %), vulvar Paget’s disease (12/1,068, 1.1 %), LSIL (397/1,068, 37.2 %), condyloma acuminatum (40/1,068, 3.7 %), vulvitis (252/1,068, 23.6 %), papillomatosis (39/1,068, 3.7 %), lichen simplex chronicus (28/1,068, 2.6 %), lichen sclerosus (55/1,068, 5.1 %), molluscum contagiosum (19/1,068, 1.8 %), epithelia keratinization (46/1,068, 4.3 %), pigment incontinence (25/1,068, 2.3 %), syringoma (16/1,068, 1.5 %), nevus (26/1,068, 2.4 %) and fibroepithelial polyp (21/1,068, 2.0 %). The mean age of all patients was 42.6±15.4 years (range: 16–85 years). As shown in Figure 1A, the differences in the mean age of patients between the SCC group (60.2±16.5 years) and condyloma acuminatum group (36.2±15.1 years), lichen sclerosus group (53.6±15.5 years) and HSIL (43.5±15.0 years), LSIL (39.9±15.0 years) (p<0.05), condyloma acuminatum (36.2±15.1 years), and papillomatosis groups (37.7±11.9 years) were significantly different (p<0.01).

The basic characteristics of patients. (A) The age of patients in the 15 groups. Data are expressed as the means±SD. *p<0.05, compared with SCC group; #p<0.05, compared with lichen sclerosus group. (B) The proportion of patients accompanied with CIN or VaIN. The percentages above the stacked bars mean the proportions of patients accompanied CIN or VaIN in each group. (C) The proportions of patients with mucosa lesion, patients with skin lesion, and patients with both lesions. The sum of white and gray bars represents the number of patients with lesions involved the mucosa. The sum of gray and black bars represents the number of patients with lesions involved the skin. SCC, squamous cell carcinoma; HSIL, high-grade squamous intraepithelial lesion; LSIL, low-grade squamous intraepithelial lesion; CIN, cervical intraepithelial neoplasia; VaIN, vaginal intraepithelial neoplasia.
Regarding the proportion of patients with cervical or vaginal intraepithelial neoplasia, the differences among the 15 groups were significant (p<0.001), as shown in Figure 1B. The proportion (accompanied CIN or VaIN patients/group patients) in groups decreased from high to low: the vulvar Paget’s disease (10/12, 83.3 %), LSIL (210/397, 52.9 %), HSIL (38/78, 48.7 %), papillomatosis (13/39, 33.3 %), condyloma acuminatum (12/40, 30.0 %), vulvitis (63/252, 25.0 %), nevus (6/26, 23.1 %), SCC (3/14, 21.4 %), syringoma (3/16, 18.8 %), pigment incontinence (4/25, 16.0 %), molluscum contagiosum (3/19, 15.8 %), epithelia keratinization (5/46, 10.9 %), lichen simplex chronicus (3/28, 10.7 %), fibroepithelial polyp (1/21, 4.8 %) and lichen sclerosus (2/55, 3.6 %).
To determine which vulvar lesions are likely to involve the mucosa or skin, the numbers of patients with mucosal lesions, patients with skin lesions, and patients with both lesions were counted, and the differences in these proportions among the 15 groups were significant (p<0.001). As shown in Figure 1C, the highest proportion of mucosa involved [(patients with mucosal lesion+patients with both lesions)/group patients] was condyloma acuminatum (82.5 %), SCC (64.3 %), LSIL (64.2 %), and HSIL (53.8 %), whereas the lesions of lichen simplex chronicus, lichen sclerosus, molluscum contagiosum, syringoma, and fibroepithelial polyp were only found in the skin.
Diagnostic and predictive parameters of each finding in ISSVD and IFCPC systems
The sensitivity, specificity, PPV, NPV, and YI of each finding in the ISSVD and IFCPC systems were calculated and the details are listed in Supplementary Table 1. These data will be used as the basis for the follow-up calculation and analysis of ODV, disease diagnosis order, and DFD.
We then measured the total lesion size and maximum size of a single lesion in the 15 groups (Figure 2A). The total area of lesions in vulvar Paget’s disease, lichen simplex chronicus, lichen sclerosus, papillomatosis, syringoma, and nevus was significantly different from those in the other multiple groups (>10 groups) (p<0.001). The largest total area of lesions was found in vulvar Paget’s disease, and the nevus was the smallest.

The total lesion size, the maximum size of single lesion and the mean number of lesions in the 15 groups. (A) The total lesion size in the 15 groups. Data are expressed as the means±SD. (B) The differences in the total lesion size between each two groups. p≥0.05, no straight line; 0.01≤p<0.05, blue straight line; 0.001≤p<0.01, red thin straight line; p<0.001, red thick straight line. (C) The maximum size of single lesion in the 15 groups. Data are expressed as the means±SD. (D) The differences in the maximum size of single lesion between each two groups. p≥0.05, no straight line; 0.01≤p<0.05, blue straight line; 0.001≤p<0.01, red thin straight line; p<0.001, red thick straight line. (E) The mean number of lesions in the 15 groups. Data are expressed as the means±SD. (F) The differences in the mean number of lesions between each two groups. p≥0.05, no straight line; 0.01≤p<0.05, blue straight line; 0.001≤p<0.01, red thin straight line; p<0.001, red thick straight line. SCC, squamous cell carcinoma; HSIL, high-grade squamous intraepithelial lesion; LSIL, low-grade squamous intraepithelial lesion.
The largest maximum size of a single lesion was in the vulvar Paget’s disease group and was significantly different from those of the other 10 groups (p<0.05). The smallest was in the molluscum contagiosum which was significantly different from the other 12 groups (p<0.05), as shown in Figure 2B.
The mean number of lesions in the syringoma group was the largest, with an average of 11.4 lesions per patient, which was different from that in the other 14 groups (p<0.05). The number of SCC lesions was the smallest, with an average of 1.08, which was different from the other six groups (p<0.05), as shown in Figure 2C.
A DFD obtained from ODV of each finding in ISSVD and IFCPC systems
To assess the ODV of findings in the ISSVD and IFCPC systems, the ODV composited comprehensively for sensitivity, specificity, PPV, NPV, and YI for each finding was calculated. A total of 35 findings in the ISSVD system and 33 findings in the IFCPC system were included; the top five findings of ODV for each vulvar disease are listed in Table 1. The highest value of ODV obtained was 18, which exist in the following cases: exudation for diagnosis of vulvar Paget’s disease, round for diagnosis of molluscum contagiosum, brown for diagnosis of syringoma, atypical vessels/exotic region/necross bleeding/red gross neoplasm/fragility for diagnosis of SCC.
The top five findings of overall diagnostic value in the 15 vulvar disease groups.
| Rank | SCC | HSIL | Vulvar Paget’s disease | LSIL | Condyloma acuminatum | Vulvitis | Papillomatosis | Lichen simplex chronicus | Lichen sclerosus | Molluscum contagiosum | Epithelia keratinization | Pigment incontinence | Syringoma | Nevus | Fibroepithelial polyp | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Finding | ODV | Finding | ODV | Finding | ODV | Finding | ODV | Finding | ODV | Finding | ODV | Finding | ODV | Finding | ODV | Finding | ODV | Finding | ODV | Finding | ODV | Finding | ODV | Finding | ODV | Finding | ODV | Finding | ODV | |
| (A) According to ISSVD system | ||||||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||||||
| 1 | Fragility | 18 | Rough | 12 | Exudation | 18 | Rough | 10 | Cristate | 14 | Smooth | 10 | Rough | 12 | Medium hardness | 14 | Medium hardness | 15 | Round | 18 | Irregular shape | 11 | Smooth | 10 | Brown | 18 | Round | 11 | Papule | 14 |
| 2 | Hardness | 14 | Sharply marginated | 10 | Erosion | 12 | Softness | 6 | Rough | 8 | Macule | 6 | Papule | 8 | Lichenification | 11 | Lichenification | 10 | Pink | 14 | Poorly marginated | 10 | White | 6 | Round | 14 | Smooth | 10 | Smooth | 11 |
| 3 | Rough | 6 | Softness | 6 | Red | 8 | Villiform | 6 | Sharply marginated | 6 | Nodule | 6 | Sharply marginated | 6 | Thickening | 6 | White | 4 | Papule | 10 | Spherical | 6 | Softness | 6 | Poorly marginated | 9 | Black | 7 | Irregular round | 8 |
| 4 | Sharply marginated | 6 | Gray | 4 | Ulcer | 7 | Papule | 4 | Softness | 6 | Softness | 6 | Softness | 6 | Smooth | 6 | Thickening | 3 | Smooth | 7 | Verrucosa | 6 | Strip-type | 6 | Papule | 6 | Sharply marginated | 6 | Irregular shape | 8 |
| 5 | Ulcer | 4 | Brown | 4 | Plaque | 6 | Fingerlike | 4 | Pink | 5 | Patch | 3 | Cauliflower-like | 6 | Strip-type | 6 | Erosion | 3 | Sharply marginated | 6 | Purpura | 6 | Macule | 4 | Smooth | 6 | Softness | 6 | Softness | 6 |
|
|
||||||||||||||||||||||||||||||
| (B) According to IFCPC system | ||||||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||||||
| 1 | Atypical vessels | 18 | Dense acetowhite epithelium | 15 | Exudation | 18 | Thin acetowhite epithelium | 15 | Thin acetowhite epithelium | 15 | Macule | 13 | Papule | 12 | Medium hardness | 15 | Medium hardness | 15 | Pink | 18 | Thickening | 8 | White | 13 | Brown | 18 | Black | 14 | Papule | 15 |
| 2 | Exophytic lesion | 18 | Coarse punctation | 7 | Erosion | 12 | Papule | 9 | Papule | 9 | Patch | 8 | Gray | 11 | Lichenification | 12 | Lichenification | 10 | Papule | 14 | Softness | 6 | Macule | 8 | Papule | 14 | Papule | 13 | Skin-colored | 14 |
| 3 | Necrosis bleeding | 18 | Softness | 6 | Red | 8 | Fine punctation | 7 | Pink | 6 | Softness | 7 | Softness | 9 | White | 5 | White | 4 | Softness | 10 | Purpura | 6 | Softness | 6 | Softness | 10 | Brown | 10 | Softness | 9 |
| 4 | Red gross neoplasm | 18 | Fine mosaic | 6 | Surface irregularities | 7 | Softness | 6 | Softness | 6 | Nodule | 5 | Plaque | 3 | Thickening | 5 | Thickening | 3 | Lichenification | 4 | Patch | 3 | Softness | 6 | Pink | 5 | ||||
| 5 | Fragility | 18 | Papule | 3 | Ulcer | 7 | Plaque | 3 | Red | 3 | Black | 3 | White | 1 | Fissure | 3 | Erosion | 3 | Excoriation | 4 | Brown | 3 | Gray | 3 | ||||||
-
ISSVD, International Society for the Study of Vulvovaginal Disease; SCC, squamous cell carcinoma; HSIL, high-grade squamous intraepithelial lesion; LSIL, low-grade squamous intraepithelial lesion; ODV, overall diagnostic value; IFCPC, International Federation for Cervical Pathology and Colposcopy.
The possible disease diagnosis order was then obtained according to the ODV, as shown in Table 2. We then created a DFD based on the disease diagnosis order (Figure 3). Some findings inferred several kinds of diseases with high diagnostic value, such as smooth, sharply marginated, soft, and papular, which indicated ≥6 kinds of diseases. In contrast, some findings had high diagnostic value only for certain diseases, such as nodules, exudation, petechia, fingerlike, cristate, villiform, black, fragility, hardness, ulcer, coarse punctation, fine mosaic, and atypical vessels, which only indicated a certain disease. To summarize the two systems, the DFD includes all the findings in the two systems, in which the DFD for all findings can be found. The thicker and darker the arrow, the more directional the diagnosis.
The possible disease diagnosis order for each finding according to the overall diagnostic value.
| Finding | Disease ranking (ODV) | ||||||
|---|---|---|---|---|---|---|---|
| No. 1 | No. 2 | No. 3 | No. 4 | No. 5 | No. 6 | ||
| (A) In ISSVD system | |||||||
|
|
|||||||
| Primary lesion | Macule | Vulvitis (6) | Pigment incontinence (4) | ||||
| Patch | Vulvitis (3) | ||||||
| Papule | Fibroepithelial polyp (14) | Molluscum contagiosum (10) | Papillomatosis (8) | Syringoma (6) | LSIL (4) | ||
| Plaque | Vulvar Paget’s disease (6) | ||||||
| Nodule | Vulvitis (6) | ||||||
| Secondary morphology | Ulcer | Vulvar Paget’s disease (7) | SCC (4) | ||||
| Erosion | Vulvar Paget’s disease (12) | Lichen sclerosus (3) | |||||
| Lichenification | Lichen simplex chronicus (11) | Lichen sclerosus (10) | |||||
| Purpura | Epithelia keratinization (6) | ||||||
| Surface | Rough | HSIL/Papillomatosis (12) | LSIL (10) | Condyloma acuminatum (8) | SCC (6) | ||
| Smooth | Vulvitis/nevus/pigment incontinence (10) | Molluscum contagiosum (7) | Lichen simplex chronicus/Syringoma (6) | ||||
| Margination | Sharply marginated | HSIL (10) | SCC/Condyloma acuminatum/Papillomatosis/Molluscum contagiosum/Nevus (6) | ||||
| Poorly marginated | Epithelia keratinization (10) | Syringoma (9) | |||||
| Appearance | Thickening | Lichen simplex chronicus (6) | Lichen sclerosus (3) | ||||
| Exudation | Vulvar Paget’s disease (18) | ||||||
| Configuration | Fingerlike | LSIL (4) | |||||
| Cristate | Condyloma acuminatum (14) | ||||||
| Villiform | LSIL (6) | ||||||
| Cauliflower-like | Condyloma acuminatum (6) | SCC (5) | |||||
| Strip-type | Lichen simplex chronicus/pigment incontinence (6) | ||||||
| Spherical | Molluscum contagiosum (18) | Syringoma (14) | Nevus (11) | ||||
| Irregular shape | Epithelia keratinization (11) | Fibroepithelial polyp (8) | |||||
| Round | Epithelia keratinization (6) | ||||||
| Verrucosa | Epithelia keratinization (6) | ||||||
| Irregular round | Fibroepithelial polyp (8) | ||||||
| Color | Red | Vulvar Paget’s disease (8) | |||||
| Gray | HSIL (4) | ||||||
| Pink | Molluscum contagiosum (14) | Condyloma acuminatum (5) | |||||
| Brown | Syringoma (18) | HSIL (4) | |||||
| White | Pigment incontinence (6) | Lichen sclerosus (4) | |||||
| Black | Nevus (7) | ||||||
| Lesion texture | Fragility | SCC (18) | |||||
| Hardness | SCC (14) | ||||||
| Medium hardness | Lichen sclerosus (15) | Lichen simplex chronicus (14) | |||||
| Softness | HSIL/LSIL/Condyloma acuminatum/vulvitis/Papillomatosis/pigment incontinence/nevus/fibroepithelial polyp (6) | ||||||
|
|
|||||||
| (B) In IFCPC system | |||||||
|
|
|||||||
| Primary lesion | Macule | Vulvitis (13) | Pigment incontinence (8) | ||||
| Patch | Vulvitis (8) | Pigment incontinence (3) | |||||
| Papule | Fibroepithelial polyp (15) | Syringoma/Molluscum contagiosum (14) | Nevus (13) | Papillomatosis (12) | LSIL/Vulvitis(9) | HSIL(3) | |
| Plaque | LSIL/Papillomatosis (3) | ||||||
| Nodule | Vulvitis (5) | ||||||
| Secondary morphology | Erosion | Vulvar Paget’s disease (12) | Lichen sclerosus (3) | ||||
| Ulcer | Vulvar Paget’s disease (7) | ||||||
| LLichenification | Lichen simplex chronicus (12) | Lichen sclerosus (10) | Epithelia keratinization (4) | ||||
| Fissure | Lichen simplex chronicus (3) | ||||||
| Purpura | Epithelia keratinization (6) | ||||||
| Excoriation | Epithelia keratinization (4) | ||||||
| Appearance | Thickening | Epithelia keratinization (8) | Lichen simplex chronicus (5) | Lichen sclerosus (3) | |||
| Exudation | Vulvar Paget’s disease (18) | ||||||
| Lesion texture | Medium hardness | Lichen simplex chronicus/Lichen sclerosus (15) | |||||
| Softness | Molluscum contagiosum/Syringoma (10) | Papillomatosis/fibroepithelial polyp (9) | Vulvitis (7) | HSIL/LSIL/Condyloma acuminatum/epithelia keratinization/pigment incontinence/nevus (6) | |||
| Color | Gray | Papillomatosis (11) | Nevus (3) | ||||
| Red | Vulvar Paget’s disease (8) | Condyloma acuminatum (3) | |||||
| Pink | Molluscum contagiosum (18) | Condyloma acuminatum (6) | Fibroepithelial polyp (5) | ||||
| Black | Nevus (14) | Vulvitis (3) | |||||
| Brown | Syringoma (18) | Nevus (10) | Pigment incontinence (3) | ||||
| Skin-colored | Fibroepithelial polyp (14) | ||||||
| White | Pigment incontinence (13) | Lichen simplex chronicus (5) | Lichen sclerosus (4) | Papillomatosis (1) | |||
| Colposcopic findings | Dense AWE | HSIL (15) | |||||
| Coarse punctation | HSIL (7) | ||||||
| Fine mosaic | HSIL (6) | ||||||
| Thin AWE | LSIL/Condyloma acuminatum (15) | ||||||
| Fine punctation | LSIL (7) | ||||||
| Surface irregularities | Vulvar Paget’s disease (7) | ||||||
| Atypical vessels | SCC (18) | ||||||
| Exophytic lesion | SCC (18) | ||||||
| Necrosis bleeding | SCC (18) | ||||||
| Red gross neoplasm | SCC (18) | ||||||
| Fragility | SCC (18) | ||||||
-
The italic value means the overall diagnostic value of the finding for a certain vulvar disease. ISSVD, International Society for the Study of Vulvovaginal Disease; ODV, overall diagnostic value; No., number; LSIL, low-grade squamous intraepithelial lesion; SCC, squamous cell carcinoma; HSIL, high-grade squamous intraepithelial lesion; IFCPC, International Federation for Cervical Pathology and Colposcopy; AWE, acetowhite epithelium.
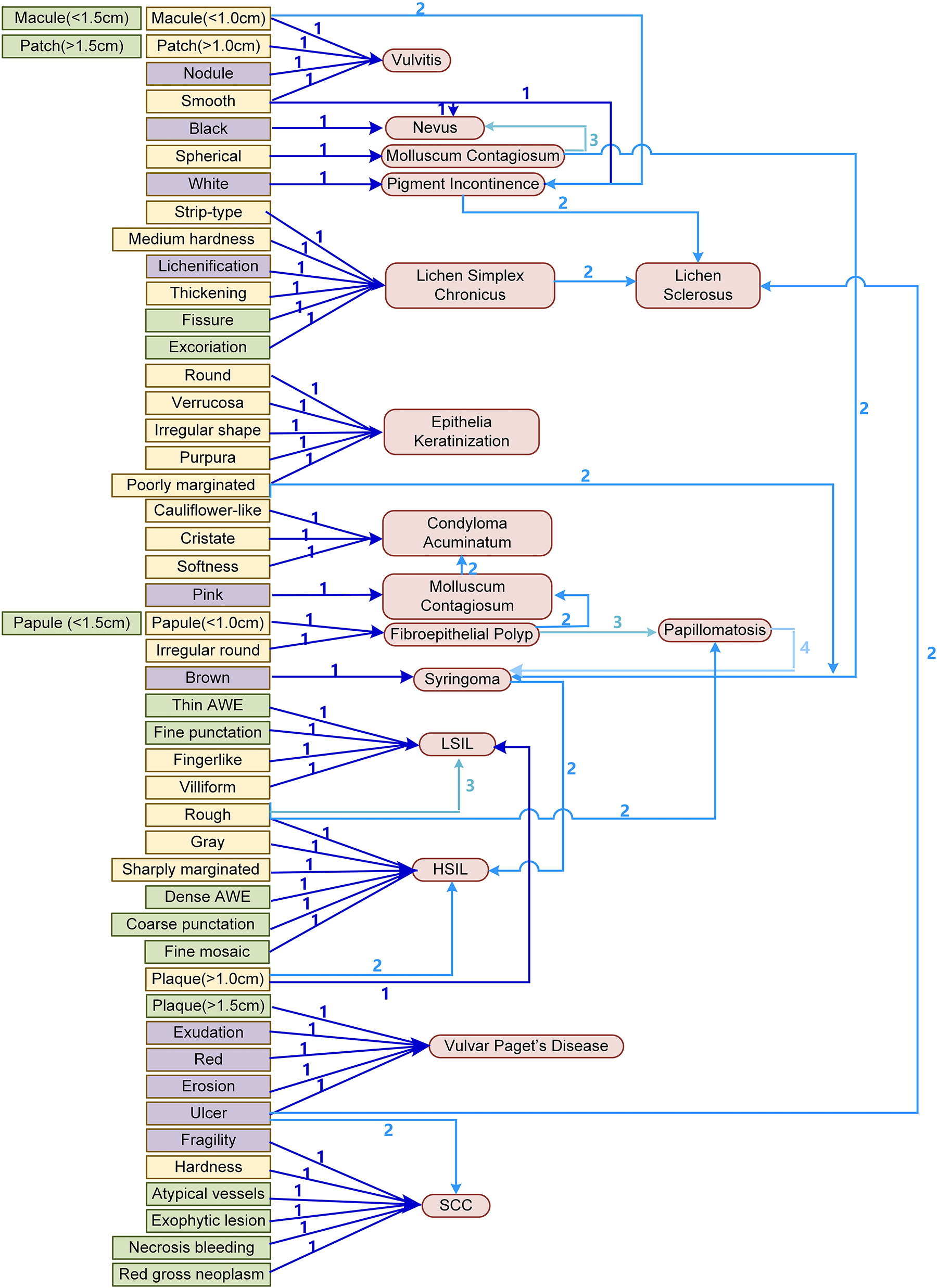
The diagnosis flow draft (DFD). The diseases in the diagnosis order at the first, second, third, and fourth positions are marked with numbers and blue arrows of different thicknesses and brightness. The higher the sort, the thicker and darker is the arrow. The flow paths in ISSVD are marked in yellow, those in IFCPC systems are marked in green, and overlapping ones are marked in purple. SCC, squamous cell carcinoma; HSIL, high-grade squamous intraepithelial lesion; LSIL, low-grade squamous intraepithelial lesion; AWE, acetowhite epithelium.
Validation on the concordance between initial impression based on DFD and histopathologic diagnosis
The concordance rate of the diagnosis between initial impression based on DFD and histopathologic diagnosis was compared among observers with different experiences. As shown in Table 3A, the agreements between the initial impression and histopathology diagnosis for observers with more than 10 years of experience, 5–10 years of experience and less than 5 years of experience were 68.8 , 65.6 and 65.4 %, respectively, and there was no significant difference among them (χ 2=3.448, p=0.178).
Agreement between the initial impression and histopathology diagnosis in observers with (A) different working experience according to diagnosis flow draft; (B) more than 10 years of working experience according to different terminology systems.
| Initial impression and histopathology diagnosis, n | ||||
|---|---|---|---|---|
| Matched | Unmatched | Total | Agreement, % | |
| (A) Working experience of observers | ||||
|
|
||||
| More than 10 years (Group C) | 735 | 333 | 1,068 | 68.8 |
| 5–10 years (Group D) | 701 | 367 | 1,068 | 65.6 |
| Less than 5 years (Group E) | 699 | 369 | 1,068 | 65.4 |
| Total | 2,135 | 1,069 | 3,204 | |
|
|
||||
| (B) Terminology systems | ||||
|
|
||||
| ISSVD (Group A) | 678 | 390 | 1,068 | 63.5 |
| IFCPC (Group B) | 695 | 373 | 1,068 | 65.1 |
| DFD (Group C) | 735 | 333 | 1,068 | 68.8 |
| Total | 2,108 | 1,096 | 3,204 | |
-
ISSVD, International Society for the Study of Vulvovaginal Disease; IFCPC, International Federation for Cervical Pathology and Colposcopy; DFD: diagnosis flow draft.
Relative to the different terminology systems, as shown in Table 3B, the concordance rate for observers with more than 10 years of experience in the ISSVD group, IFCPC group and DFD group were 63.5 , 65.1 and 68.8 %, respectively, where the differences were significant among the three groups (χ 2=7.125, p=0.028).
Discussion
For years, the 2011 ISSVD and 2011 IFCPC terminology and classification systems have been used to describe the findings and lesions in the vulva. The ISSVD terminology system represents a classification system according to the pathophysiology of the disease, but it does not include pattern recognition [9]. In contrast, the IFCPC terminology system can be very useful for describing clinical findings in the vulva, but its classification was not as detailed as that of ISSVD’s, which makes it difficult to compare the clinical findings with the final histological results from the punch biopsy [9]. In addition, clinical evaluation studies on the applicability of these two classification systems for vulvar diseases are extremely scarce. In the present study, we analyzed the clinical diagnostic value of findings in the two systems and developed a DFD based on vulvar appearance impression of the two terminology systems. We also evaluated the clinical application of the DFD in the diagnosis of vulvar diseases.
In this study, 15 types of vulvar diseases were included, including noninfectious inflammation, viral infection, pigmented lesions of the vulva, and neoplasms. Our data showed that patients with SCC (60.2±16.5) and lichen sclerosus (53.6±15.5 years) were generally older, while those with condyloma acuminatum (36.2±15.1 years) were younger. Similar to our results, the median age of patients with vulvar malignancy was 68 years in Virarkar’s report, and the mean age of lichen sclerosus was 55.1 years in women in Torres et al. [10, 11]. This suggests that for vulvar malignancy and lichen sclerosus, particular attention should be paid to older and postmenopausal women [12]. Diţescu et al. reported the onset ages of condyloma acuminatum range from 17 to 33 years in 80 % of cases, which was similar to our results [13]. Condyloma acuminatum associated with HPV infection may be more likely to occur in sexually active young patients [14]. There was a high proportion (accompanied by CIN or VaIN patients/group patients) in the vulvar Paget’s disease (83.3 %), LSIL (52.9 %), and HSIL (48.7 %) groups, indicating that HPV-related disease can manifest throughout the anogenital region rather than being confined to a particular organ [15]. The association between HPV infection and vulvar Paget’s disease remains controversial [16, 17]. Our data showed a close association between vulvar Paget’s disease and lower genital tract intraepithelial neoplasia with HPV infection, and we suggest making a complete evaluation of the lower genital tract (with cytology, HPV testing, and colposcopy) for patients with vulvar Paget’s disease [16]. According to our results, vulvar dermatosis, molluscum contagiosum, syringoma, and fibroepithelial polyps mainly affect the vulvar skin, while vulvar diseases associated with HPV infection usually involve both the skin and mucosa.
In this study, the largest total area of lesions and the largest maximum size of a single lesion were found in the vulvar Paget’s disease group. This is in line with other reports that extramammary Paget disease has the main characteristics of malignant potential, multiple sites involved, and large size of the lesion [18], [19], [20]. As chronic inflammatory dermatosis without unknown etiology, lichen simplex chronicus and lichen sclerosus currently have no cure and high relapse rate, which may cause the lesion to expand gradually [21, 22]. The main characteristics of molluscum contagiosum are dome-shaped, smooth-surfaced, pearly, firm, skin-colored, pink, yellow, or white papules, 2–5 mm in diameter with central umbilication, and the lesions should be differentiated from those in genital warts, herpes simplex, and herpes zoster [23]. Similar to previous reports, we found that syringomas are frequently multiple with tiny, firm, and skin-colored subcutaneous papules [24, 25].
The application of IFCPC terminology, which provides standardized interpretations and classifications of colposcopic findings in the lower genital tract, including the cervix, vagina, vulva, and anus, has been a concern. Some studies reported the application of the IFCPC system on diagnosis of lower genital tract neoplasia [9, 26, 27]. In some studies, the clinical application of the 2011 IFCPC terminology for the detection of CIN and VaIN were explored, and the diagnostic value of some findings were evaluated [8, 28]. Therefore, we utilized the latest 2011 ISSVD and 2011 IFCPC terminology systems to describe vulvar diseases in this study. Findings that were considered to have high diagnostic value were screened out based on the ODV and combined with both systems. Furthermore, we draw a DFD on the basis of the possible disease diagnosis order, in which the findings and diseases are listed and connected by arrows. Based on vulvar appearance impression, initial impression diagnosis can be soon suggested. We used the ODV, which was composed of sensitivity, specificity, PPV, NPV, and YI, to comprehensively assess the diagnostic value of each finding in the ISSVD and IFCPC systems. The finding with the highest ODV (value=18) was considered to have high diagnostic value for certain vulvar diseases, such as exudation for vulvar Paget’s disease, round for molluscum contagiosum, brown for syringoma, and atypical vessels for SCC. Among the top five findings for each disease, some findings (such as smoothness and softness) were found to infer several (≥6) kinds of diseases, which probably indicated that although they had high diagnostic value for diseases, they were not conducive to differential diagnosis. In contrast, some findings (such as exudation and atypical vessels) only exist in certain diseases, showing that these findings are of high value in distinguishing other diseases.
The aim of the IFCPC was to create terminology that would take into account the 2011 ISSVD vulvar terminology, as well as add terminologies specific to other regions and compatible with colposcopy, so some of the findings in the two systems overlap [6]. For example, the names (such as macules and patches) and color of the lesions are both described in the two systems. Although macule, patch, papule, plaque and nodule are defined differently in diameter in the two systems, this may not affect the identification of the lesion, since measurements are approximate and overlap may occur between “small” and “large” lesions [5]. Some of the findings for the two systems were different. The ISSVD includes the description of the surface (such as smooth and rough), margination (such as sharply marginated and poorly marginated), and configuration (the shape of the lesion), while structures and composition of the vulva, lesion size and location, and colposcopic terminology are added to the IFCPC. Some new features (such as exudation and lesion texture), which were not included in the ISSVD or IFCPC system, were also considered because of their high frequency and diagnostic value in some vulvar diseases.
It is challenging for observers to make an accurate diagnosis of vulvar disease, and one of the most common reasons is lack of training and experience [29]. In this study, the differences in agreements between the initial impression and histopathology diagnosis among the observers with differing amounts of experience in colposcopy were not significant when the DFD was used. Stuebs et al. reported that the overall concordance rate in detecting early vulvar neoplasia using the new classification was 53.9 %, which was lower than that in our study [9]. Furthermore, when different terminology systems were used by examiners with similar experience, the concordance rate in the DFD group was higher than those in the ISSVD and IFCPC groups. For a long time, it has been considered that the judgement and evaluation of vulvar disease are subjective and dependent on the visual sense and experience of observers. The present study showed that observers could obtain a high diagnostic rate even with less experience when using an objectively reliable assistant diagnostic method [8].
However, vulvar diseases are so complex and heterogeneous that trying to simplify them into an algorithm is difficult and can lead to unnecessary biopsies and erroneous treatments. To achieve good management of vulvar diseases, it is still recommended that the specialist be the one to carry out the diagnosis and treatment. Despite the large number of patients in total in this study, the contribution of some diseases is small in numbers, so comparisons can be biased. Thus, more kinds of vulvar disease are needed to be considered. The DFD could be useful for observers with little training in vulvar diseases. However, more precise and refined classification is warranted to be improved. In addition, emphasis should be put on alternative assessments or adjunct technologies (such as immunohistochemical findings, fluorescence labeling and artificial intelligence), which could benefit objective evaluation of the vulva.
Conclusions
In summary, we explored a diagnosis flow draft based on vulvar appearance impression of the two terminology systems for detection of vulvar disease, in which initial impression diagnosis can be soon suggested. The diagnosis flow draft may be of the potential benefit to the diagnosis of vulvar disease in the fields of gynecology, dermatology, venereology, and urogenital departments.
Funding source: Shanghai Nature Science Foundation
Award Identifier / Grant number: 21ZR1410400
Award Identifier / Grant number: 22ZR1408800
Funding source: Science and Technology Commission of Shanghai Municipality
Award Identifier / Grant number: 21Y11906500
Funding source: National Natural Science Foundation of China
Award Identifier / Grant number: 82272970
Acknowledgments
The authors thank the Shanghai Key Laboratory of Female Reproductive Endocrine Related Diseases. All participants in this study are appreciated.
-
Research ethics: Institutional Review Board approval was obtained properly (2023–25).
-
Informed consent: Informed consent was obtained from all individuals included in this study.
-
Author contributions: All authors have accepted responsibility for the entire content of this manuscript and approved its submission.
-
Competing interests: The authors state no conflict of interest.
-
Research funding: This study was supported by the Science and Technology Commission of Shanghai Municipality (No. 21Y11906500), the National Natural Science Foundation of China (Grant No. 82272970) and Shanghai Nature Science Foundation (Grant No. 21ZR1410400 and No. 22ZR1408800).
-
Data availability: The raw data can be obtained on request from the corresponding author.
References
1. Sally, R, Shaw, KS, Pomeranz, MK. Benign “lumps and bumps” of the vulva: a review. Int J Womens Dermatol 2021;7:383–90, https://doi.org/10.1016/j.ijwd.2021.04.007.Search in Google Scholar PubMed PubMed Central
2. Mauskar, MM, Marathe, K, Venkatesan, A, Schlosser, BJ, Edwards, L. Vulvar diseases: approach to the patient. J Am Acad Dermatol 2020;82:1277–84, https://doi.org/10.1016/j.jaad.2019.07.115.Search in Google Scholar PubMed
3. Bornstein, J, Bogliatto, F, Haefner, HK, Stockdale, CK, Preti, M, Bohl, TG, et al.. The 2015 International Society for the Study of Vulvovaginal Disease (ISSVD) terminology of vulvar squamous intraepithelial lesions. Obstet Gynecol 2016;127:264–8, https://doi.org/10.1097/aog.0000000000001285.Search in Google Scholar PubMed
4. Lynch, PJ, Moyal-Barracco, M, Moyal-Barrocco, M, Bogliatto, F, Scurry, J. 2006 ISSVD classification of vulvar dermatoses: pathologic subsets and their clinical correlates. J Reprod Med 2007;52:3–9.Search in Google Scholar
5. Lynch, PJ, Moyal-Barracco, M, Scurry, J, Stockdale, C. 2011 ISSVD Terminology and classification of vulvar dermatological disorders: an approach to clinical diagnosis. J Low Genit Tract Dis 2012;16:339, https://doi.org/10.1097/lgt.0b013e3182494e8c.Search in Google Scholar PubMed
6. Bornstein, J, Sideri, M, Tatti, S, Walker, P, Prendiville, W, Haefner, HK. 2011 terminology of the vulva of the International Federation for Cervical Pathology and Colposcopy. J Low Genit Tract Dis 2012;16:290–5, https://doi.org/10.1097/lgt.0b013e31825934c7.Search in Google Scholar PubMed
7. Maldonado, VA. Benign vulvar tumors. Best Pract Res Clin Obstet Gynaecol 2014;28:1088–97, https://doi.org/10.1016/j.bpobgyn.2014.07.014.Search in Google Scholar PubMed
8. Li, Y, Duan, X, Sui, L, Xu, F, Xu, S, Zhang, H, et al.. Closer to a uniform language in colposcopy: study on the potential application of 2011 International Federation for Cervical Pathology and Colposcopy Terminology in Clinical Practice. BioMed Res Int 2017;2017:8984516, https://doi.org/10.1155/2017/8984516.Search in Google Scholar PubMed PubMed Central
9. Stuebs, FA, Mehlhorn, G, Gass, P, Schulmeyer, CE, Adler, W, Strehl, J, et al.. Concordance rate of vulvoscopic findings in detecting early vulvar neoplasia. Gynecol Oncol 2020;157:463–8, https://doi.org/10.1016/j.ygyno.2020.02.026.Search in Google Scholar PubMed
10. Virarkar, M, Vulasala, SS, Daoud, T, Javadi, S, Lall, C, Bhosale, P. Vulvar cancer: 2021 revised FIGO staging system and the role of imaging. Cancers 2022;14:2264. https://doi.org/10.3390/cancers14092264.Search in Google Scholar PubMed PubMed Central
11. Torres, A, Zaborek-Lyczba, M, Lyczba, J, Mertowska, P, Mertowski, S, Grywalska, E. The importance of immunological disorders in the pathogenesis of lichen sclerosus in pediatric patients: a Systematic review. Int J Mol Sci 2022;23:14212. https://doi.org/10.1021/acsorginorgau.1c00011.Search in Google Scholar PubMed PubMed Central
12. Pedrao, PG, Guimaraes, YM, Godoy, LR, Possati-Resende, JC, Bovo, AC, Andrade, C, et al.. Management of early-Stage vulvar cancer. Cancers 2022;14:4184. https://doi.org/10.3390/cancers14174184.Search in Google Scholar PubMed PubMed Central
13. Ditescu, D, Istrate-Ofiteru, AM, Rosu, GC, Iovan, L, Liliac, IM, Zorila, GL, et al.. Clinical and pathological aspects of condyloma acuminatum – review of literature and case presentation. Rom J Morphol Embryol 2021;62:369–83, https://doi.org/10.47162/RJME.62.2.03.Search in Google Scholar PubMed PubMed Central
14. Park, IU, Introcaso, C, Dunne, EF. Human papillomavirus and genital warts: a review of the evidence for the 2015 Centers for Disease Control and Prevention Sexually Transmitted Diseases Treatment Guidelines. Clin Infect Dis 2015;61:S849–55, https://doi.org/10.1093/cid/civ813.Search in Google Scholar PubMed
15. Buchanan, TR, Zamorano, AS, Massad, LS, Liu, J, Thaker, PH, Powell, MA, et al.. Risk of cervical and vaginal dysplasia after surgery for vulvar intraepithelial neoplasia or cancer: a 6 year follow-up study. Gynecol Oncol 2019;155:88–92, https://doi.org/10.1016/j.ygyno.2019.07.017.Search in Google Scholar PubMed PubMed Central
16. Clemente, N, Ciavattini, A, Valenti, G, Zannier, F, Di Giuseppe, J, Delli Carpini, G, et al.. Extramammary Paget’s disease of the vulva and concomitant premalignant/malignant vulvar lesions: a potential challenge in diagnosis and treatment. Curr Oncol 2023;30:959–66, https://doi.org/10.3390/curroncol30010073.Search in Google Scholar PubMed PubMed Central
17. Shah, VI, Bergin, L, Rowlands, GL, McCluggage, WG. Unusual manifestations of vulval Paget disease. Int J Gynecol Pathol 2022;41:476–83, https://doi.org/10.1097/pgp.0000000000000824.Search in Google Scholar PubMed
18. Preti, M, Joura, E, Vieira-Baptista, P, Van Beurden, M, Bevilacqua, F, Bleeker, MCG, et al.. The European Society of Gynaecological Oncology (ESGO), the International Society for the Study of Vulvovaginal Disease (ISSVD), the European College for the Study of Vulval Disease (ECSVD) and the European Federation for Colposcopy (EFC) Consensus Statements on pre-invasive vulvar lesions. J Low Genit Tract Dis 2022;26:229–44, https://doi.org/10.1097/lgt.0000000000000683.Search in Google Scholar PubMed PubMed Central
19. Chen, Z, Liu, Z, Pan, S, Liu, J, Zuo, S, Wang, P. The clinical characteristics and prognostic factors of primary extramammary paget’s disease treated with surgery in anogenital regions: a large population study from the SEER database and our Centre. J Clin Med 2023;12:582.10.3390/jcm12020582Search in Google Scholar PubMed PubMed Central
20. Carton, I, Lebreton, M, Tesson, C, Henno, S, Lavoue, V, Leveque, J, et al.. Paget’s disease of the vulva: a challenge for the gynaecologist. J Gynecol Obstet Hum Reprod 2021;50:101896, https://doi.org/10.1016/j.jogoh.2020.101896.Search in Google Scholar PubMed
21. Obstetricians ACo, Bulletins—Gynecology GCoP. Diagnosis and management of vulvar skin disorders: ACOG Practice Bulletin, Number 224. Obstet Gynecol 2020;136:e1–14.10.1097/AOG.0000000000003944Search in Google Scholar PubMed
22. Monreal, J. Safety and efficacy of stromal vascular fraction enriched fat grafting therapy for vulvar lichen sclerosus. Cureus 2020;e7096. https://doi.org/10.7759/cureus.7096.Search in Google Scholar PubMed PubMed Central
23. Edwards, S, Boffa, MJ, Janier, M, Calzavara-Pinton, P, Rovati, C, Salavastru, CM, et al.. 2020 European guideline on the management of genital molluscum contagiosum. J Eur Acad Dermatol Venereol 2021;35:17–26, https://doi.org/10.1111/jdv.16856.Search in Google Scholar PubMed
24. Shalabi, MMK, Homan, K, Bicknell, L. Vulvar syringomas. SAVE Proc 2022;35:113–4, https://doi.org/10.1080/08998280.2021.1977076.Search in Google Scholar PubMed PubMed Central
25. Botsali, A, Caliskan, E, Coskun, A, Tunca, M. Eruptive syringoma: two cases with dermoscopic features. Skin Appendage Disord 2020;6:319–22, https://doi.org/10.1159/000508656.Search in Google Scholar PubMed PubMed Central
26. Zhang, B, Hong, S, Zhang, G, Rong, F. Clinical application of the 2011 IFCPC colposcope terminology. BMC Wom Health 2021;21:257, https://doi.org/10.1186/s12905-021-01395-1.Search in Google Scholar PubMed PubMed Central
27. Sopracordevole, F, Barbero, M, Clemente, N, Fallani, MG, Cattani, P, Agarossi, A, et al.. Colposcopic patterns of vaginal intraepithelial neoplasia: a study from the Italian Society of Colposcopy and Cervico-Vaginal Pathology. Eur J Cancer Prev 2018;27:152–7, https://doi.org/10.1097/cej.0000000000000287.Search in Google Scholar
28. Zhou, Q, Zhang, F, Sui, L, Zhang, H, Lin, L, Li, Y. Application of 2011 International Federation for Cervical Pathology and Colposcopy Terminology on the detection of vaginal intraepithelial neoplasia. Cancer Manag Res 2020;12:5987–95, https://doi.org/10.2147/cmar.s257875.Search in Google Scholar
29. Garcia-Souto, F, Lorente-Lavirgen, AI, Ildefonso Mendonca, FM, Garcia-de-Lomas, M, Hoffner-Zuchelli, MV, Rodriguez-Ojeda, D, et al.. Vulvar dermatoses: a cross-sectional 5-year study. Experience in a specialized vulvar unit. An Bras Dermatol 2022;97:747–56, https://doi.org/10.1016/j.abd.2021.11.006.Search in Google Scholar PubMed PubMed Central
Supplementary Material
This article contains supplementary material (https://doi.org/10.1515/dx-2023-0146).
© 2023 the author(s), published by De Gruyter, Berlin/Boston
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Frontmatter
- Opinion Paper
- Exploring synthesis as a vital cognitive skill in complex clinical diagnosis
- Original Articles
- Physiologic measurements of cognitive load in clinical reasoning
- Impact of diagnostic management team on patient time to diagnosis and percent of accurate and clinically actionable diagnoses
- Game-based learning to improve diagnostic accuracy: a pilot randomized-controlled trial
- A patient follow-up intervention to improve medical decision making at an internal medicine residency program
- Application of a diagnosis flow draft based on appearance impression for detection of vulvar disease
- The consequences of delayed diagnosis and treatment in persons with multiple sclerosis given autologous hematopoietic stem cell transplantation
- Troponin testing in routine primary care: observations from a dynamic cohort study in the Amsterdam metropolitan area
- Use of saliva-based qPCR diagnostics for the accurate, rapid, and inexpensive detection of strep throat
- Short Communications
- Improving communication of diagnostic uncertainty to families of hospitalized children
- Association of diagnostic error education and recognition frequency among Japanese medical students: a nationwide cross-sectional study
- Updated statistics on Influenza mortality
- Letters to the Editor
- How case reports can be used to improve diagnosis
- Clinical assessment of Ortho VITROS SARS-CoV-2 antigen chemiluminescence immunoassay
- Convicting a wrong molecule?
- Case Reports - Lessons in Clinical Reasoning
- Lessons in clinical reasoning – pitfalls, myths, and pearls: a woman brought to a halt
- Lessons in clinical reasoning – pitfalls, myths, and pearls: shoulder pain as the first and only manifestation of lung cancer
- Congress Abstracts
- The Future of Diagnosis: Achieving Excellence and Equity
- The Future of Diagnosis: Navigating Uncertainty
Articles in the same Issue
- Frontmatter
- Opinion Paper
- Exploring synthesis as a vital cognitive skill in complex clinical diagnosis
- Original Articles
- Physiologic measurements of cognitive load in clinical reasoning
- Impact of diagnostic management team on patient time to diagnosis and percent of accurate and clinically actionable diagnoses
- Game-based learning to improve diagnostic accuracy: a pilot randomized-controlled trial
- A patient follow-up intervention to improve medical decision making at an internal medicine residency program
- Application of a diagnosis flow draft based on appearance impression for detection of vulvar disease
- The consequences of delayed diagnosis and treatment in persons with multiple sclerosis given autologous hematopoietic stem cell transplantation
- Troponin testing in routine primary care: observations from a dynamic cohort study in the Amsterdam metropolitan area
- Use of saliva-based qPCR diagnostics for the accurate, rapid, and inexpensive detection of strep throat
- Short Communications
- Improving communication of diagnostic uncertainty to families of hospitalized children
- Association of diagnostic error education and recognition frequency among Japanese medical students: a nationwide cross-sectional study
- Updated statistics on Influenza mortality
- Letters to the Editor
- How case reports can be used to improve diagnosis
- Clinical assessment of Ortho VITROS SARS-CoV-2 antigen chemiluminescence immunoassay
- Convicting a wrong molecule?
- Case Reports - Lessons in Clinical Reasoning
- Lessons in clinical reasoning – pitfalls, myths, and pearls: a woman brought to a halt
- Lessons in clinical reasoning – pitfalls, myths, and pearls: shoulder pain as the first and only manifestation of lung cancer
- Congress Abstracts
- The Future of Diagnosis: Achieving Excellence and Equity
- The Future of Diagnosis: Navigating Uncertainty

