Abstract
The 2024 Kidney Disease: Improving Global Outcomes (KDIGO) guidelines for chronic kidney disease (CKD) evaluation and management bring important updates, particularly for European laboratories. These guidelines emphasize the need for harmonization in CKD testing, promoting the use of regional equations. In Europe, the European Kidney Function Consortium (EKFC) equation is particularly suited for European populations, particularly compared to the CKD-EPI 2021 race-free equation. A significant focus is placed on the combined use of creatinine and cystatin C to estimate glomerular filtration rate (eGFRcr-cys), improving diagnostic accuracy. In situations where eGFR may be inaccurate or clinically insufficient, the guidelines encourage the use of measured GFR (mGFR) through exogenous markers like iohexol. These guidelines emphasize the need to standardize creatinine and cystatin C measurements, ensure traceability to international reference materials, and adopt harmonized reporting practices. The recommendations also highlight the importance of incorporating risk prediction models, such as the Kidney Failure Risk Equation (KFRE), into routine clinical practice to better tailor patient care. This article provides a European perspective on how these KDIGO updates should be implemented in clinical laboratories to enhance CKD diagnosis and management, ensuring consistency across the continent.
Introduction
The publication of new Kidney Disease Improving Global Outcomes (KDIGO) guidelines is always a highly anticipated event for the nephrology community. This anticipation was especially strong for the 2024 version of the KDIGO guidelines on the “Evaluation and Management of Chronic Kidney Disease (CKD)” released in April [1]. The previous edition, published over a decade ago in 2013 [2], urgently required updating due to significant advancements and active research in the field.
Specialists in Laboratory medicine have a vast interest in these guidelines given their critical role in the evaluation of CKD. Reflecting this, an expert in clinical chemistry contributed to the KDIGO recommendations.
In this article, proposed by the EFLM Committee on CKD, we will summarize and provide brief comment on these guidelines, with a particular focus on the first two key components (Evaluation of CKD and Risk Assessment), which hold the most relevance for European Specialists in Laboratory Medicine. Our discussion will be limited to recommendations pertinent to adult patients.
Evaluation of CKD
Definition
Chronic kidney disease (CKD) remains defined as abnormalities in kidney structure or function persisting for at least 3 months, with health implications. The 2024 KDIGO guidelines continue to emphasize glomerular filtration rate (GFR) and albuminuria, expressed as urine albumine-creatinine ratio (ACR) as core metrics for CKD diagnosis and classification. CKD is confirmed when GFR falls below 60 mL/min/1.73 m2 or when structural abnormalities, such as persistent albuminuria (ACR>30 mg/g), are present despite higher GFR. Patients are categorized using the CGA system: Cause, GFR (G1–G5), and Albuminuria (A1–A3), which informs prognosis and management, the last two biological parameters being used in the well-known heatmap CKD table (Figure 1). Definition of CKD and CKD classification are justified by epidemiological studies led by the CKD-prognosis consortium (CKD-PC) showing associations between estimated GFR (eGFR) below 60 mL/min/1.73 m2 and/or ACR (A2-A3) and adverse outcomes like mortality or kidney failure [1]. Compared to previous guidelines, the novelty lies in considering associations between outcomes and low GFR levels using both creatinine and cystatin C (and using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) 2021 equation, see below), rather than solely creatinine [3]. Previous guidelines and the epidemiological arguments they relied upon have faced criticism. The new guideline, supported by data using cystatin C, address some of these criticisms but also introduce new challenges, as cystatin C measurement is not universally available. One major criticism of both the previous and new guideline is the lack of consideration of age in defining CKD, which uses a single GFR threshold. Advocates of an age-adapted definition argue that the current definition significantly overestimates CKD prevalence in older populations while underestimating it in younger individuals [4], [5], [6]. Although it is beyond the scope of this analysis to delve deeply into this debate, it is evident that the controversy regarding CKD definition is ongoing and unresolved [7], [8], [9], [10], [11].
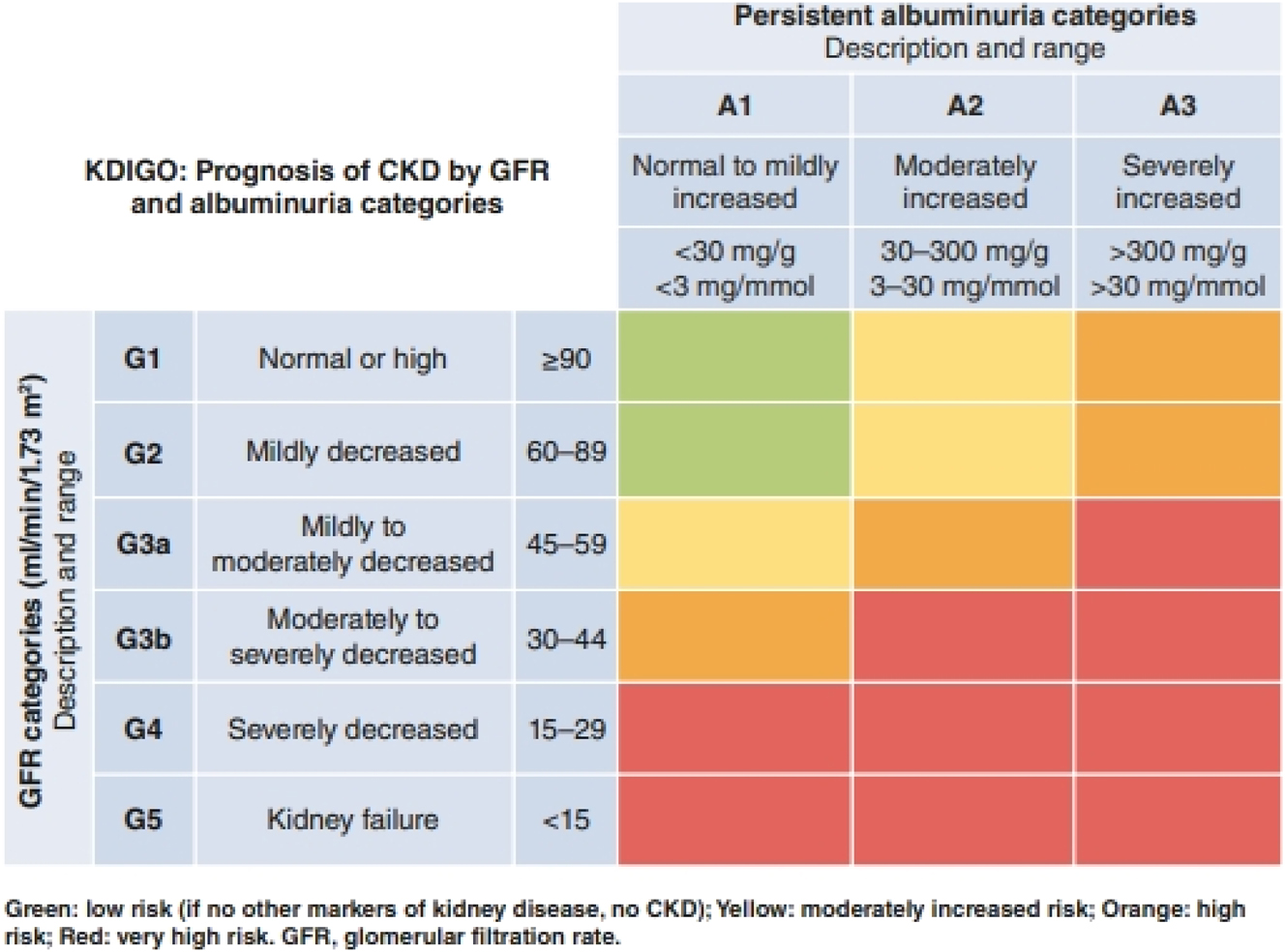
CKD Heatmap for diagnosis and classification. This matrix, derived from the 2024 KDIGO guidelines, illustrates the classification of chronic kidney disease (CKD) using glomerular filtration rate (GFR) and albuminuria (ACR) levels. Patients are categorized using the CGA system (Cause, GFR, Albuminuria) to assess disease stage and prognosis, with CKD confirmed at GFR values below 60 mL/min/1.73 m2 or in cases of persistent albuminuria (ACR>30 mg/g) regardless of GFR.
Similar to the previous version, the 2024 KDIGO guideline emphasizes the importance of the chronicity criterion, to avoid confusion with acute kidney injury, but also because ACR and eGFR have biological and analytical variability. Abnormal GFR and/or ACR results should be thus confirmed [5], 12], 13]. The role of clinical laboratories is crucial in this context. When validating abnormal eGFR and ACR results, specialists in laboratory medicine should review the patient’s medical history to identify any prior abnormalities or known diagnoses of CKD. If no documented history of CKD exists, we recommend repeating the measurements after three months to confirm the persistence of abnormal values, in accordance with CKD diagnostic guidelines.
Glomerular filtration rate
Compared to the 2013 KDIGO guidelines, the 2024 update places greater emphasis on the role of cystatin C as a valuable complement to serum creatinine, particularly in cases where creatinine-based estimates may be less accurate. This is clearly articulated in the recommendation 1.1.2.1: “In adults at risk for CKD, we recommend using creatinine-based estimated glomerular filtration rate (eGFRcr). If cystatin C is available, the GFR category should be estimated from the combination of creatinine and cystatin C (creatinine and cystatin C–based estimated glomerular filtration rate [eGFRcr-cys]).” This recommendation is based on two arguments. The first argument is coming from several studies showing that measured GFR is better estimated by eGFRcr-cys than eGFRcr alone [14], [15], [16], [17], [18], [19], and the second one is epidemiological, with data showing a higher association between outcomes and eGFRcr-cys than eGFRcr [3]. We have recently shown that cystatin C was available in most European countries, but certainly not in every clinical laboratory [20]. Moreover, cystatin C determination remains expensive (at least, compared to creatinine) and the test is not systematically reimbursed in every patient, with the exception of Sweden. This is indirectly acknowledged by the KDIGO who still suggest that eGFRcr should be the initial test and propose to measure cystatin C “in clinical situations when eGFRcr is less accurate and GFR affects clinical decision -making” (recommendation 1.2.21). The level of this recommendation is rather low (1C). Indeed, in clinical practice, it is not always clear when eGFR based on creatinine might be inaccurate. The theoretical indications for using cystatin C (summarized in Table 8 of the 2024 KDIGO guidelines) are numerous and can apply to most patients seen in a hospital setting. Additionally, it is well known that abnormalities in muscle mass can affect the validity of eGFRcr. Consequently, in individuals with significantly altered muscle mass, such as those with muscular dystrophy or cachexia, eGFRcr may not accurately reflect the true glomerular filtration rate. However, estimating a patient’s muscle mass in clinical practice remains challenging [21], 22]. Finally, yet importantly, the recommendation to measure cystatin C, especially in specific situations where a relevant clinical decision depends on GFR and the risk of inaccuracy of eGFRcr is high, can be questioned. Indeed, cystatin C can be affected by “non-GFR determinants”, which are different factors, other than glomerular filtration rate, that affect its serum levels (this is particularly true for thyroid function, but other factors like adiposity, smoking, inflammation, diabetes, proteinuria, high cell turnover diseases and corticosteroids have already been evoked in the literature [23]. In such cases, KDIGO recommends considering equations that combine creatinine and cystatin C (eGFRcr-cys). The overall goal of the Guideline is to equip the clinician with more knowledge about the inherent limitations of endogenous biomarkers to enable him/her to interpret the results against the background of the clinical characteristics of the patient. However, it may be also more appropriate to measure GFR using a reference method, at least for certain patients [24], [25], [26] like stated in Practice Point 1.2.2.2 “Where more accurate ascertainment of GFR will impact treatment decisions, measure GFR using plasma or urinary clearance of an exogenous filtration marker.” Although iohexol clearance is currently limited to specialized centers, it remains one of the few methods available in Europe for accurately measuring GFR. Clinical laboratories play a crucial role in facilitating this method, particularly by ensuring proper testing protocols, interpretation of results, and collaboration with specialized centers to support accurate GFR measurement when needed [27], [28], [29].
In this context, the European Kidney Function Consortium (EKFC) and the EFLM have recently proposed standardized procedures for iohexol plasma clearance determination [30].
In conclusion, the 2024 KDIGO guideline clearly promotes the measurement of cystatin C, even though the indications for its prescription are not always straightforward. When cystatin C results are available, estimating GFR equations including both biomarkers (eGFRcr-cys) might be preferred. The 2024 KDIGO guideline also provides implementation standards for clinical laboratories to ensure the accuracy and reliability of eGFR assessments using creatinine and cystatin C. These recommendations are detailed in Table 1. From a laboratory perspective, it is of great importance to assure the accuracy of results obtained with metrological traceable and standardized methods to ensure comparability of results across different laboratories, enhancing the reliability of CKD assessments. Methods used for serum creatinine determination (either Jaffe or enzymatic ones) should all be traceable to the NIST Standard Reference Material (SRM) 967a whose creatinine concentration has been certified using a reference isotope-dilution LC/MS (IDMS) method. Moreover, all cystatin C assays should be traceable to the CERM-DA471/IFCC reference material to enhance the standardization and reproducibility of measurements, which is paramount for consistent clinical decision-making across different settings.
Implementation standards to ensure accuracy and reliability of GFR assessments using creatinine and cystatin C.
|
|
|
|
|
|
|
|
Estimating equations
Compared to 2013 KDIGO, there are new recommendations for the choice of the estimating equations. In 2013, only the equations proposed by the US-based CKD epidemiology collaboration (CKD-EPI) were recommended [18], 31]. The creatinine-based CKD-EPI equation from 2009 [31] and the cystatin C-based equation from 2012 [18] have however recently been challenged. First, in the US, the 2009 CKD-EPI eGFRcr equation has been criticized for including race as a variable, which was considered as discriminatory in Black Americans [32]. This led the authors of the CKD-EPI equation to propose a new race-free equation based on creatinine [17]. The impact of these new race-free equation on GFR estimation performance has been largely discussed [33], 34]. Briefly, several studies showed that the racial coefficient correction used in the 2009 CKD-EPI equation was inaccurate for African and European Black individuals [35], [36], [37], [38]. The new 2021 CKD-EPI equation is less accurate for non-Black individuals than the 2009 version, both in the US and Europe [17], 36]. The use of the 2021 race-free CKD-EPI equation has been questioned in Europe and Africa and the European Renal Association (ERA) and the EFLM argued against adopting this less accurate equation for reasons related to a racial coefficient that should not apply to these continents [39], 40]. Concurrently, the EKFC highlighted other limitations of the creatinine-based CKD-EPI equations and proposed its own creatinine- and cystatin C-based equations. These EKFC equations are recognized and validated by KDIGO, which implicitly supports their use in Europe or any other region, provided that there is harmonization of equation use within distinct geographical areas. Indeed, KDIGO specifically recommends to “Use the same equation within geographical regions (as defined locally [e.g., continent, country, region] and as large as possible).” The EKFC equations are developed primarily from European cohorts and are extensively validated in Europe [15], 16], 36], 41]. They are based on rescaled creatinine or cystatin C using the Q value, which represents the mean serum creatinine and cystatin C concentration observed in a given population (e.g., creatinine Q-values for adult women and men in Europe are 0.70 mg/dL and 0.90 mg/dL, respectively). This formula is flexible, allowing for the use of Q-values in specific or mixed populations or totally race-free Q values if required [36], 42], 43]. The EKFC equations have additional strengths:
They can be used across children, adolescents and adults, ensuring continuity in eGFR results,
They avoid the strong overestimation of GFR in young adults observed with the CKD-EPI equations,
Albuminuria
ACR is the other cornerstone variable in nephrology. This parameter is of outstanding importance to stage the patient and to evaluate the risk of CKD progression, but also the risk of mortality. Using ACR on a urine sample is the best balance between feasibility and relevance. In absence of albumin, protein-to-creatinine ratio can also be considered. While measurement in the first morning void is not necessary for initial testing, a pathological result (ACR>30 mg/g) should ideally be confirmed with a midstream urine sample obtained from the first morning void. The KDIGO guideline also reminds us about the factors that can influence both urine albumin (exercise, infection) and urine creatinine (sex, weight, protein intake) concentrations. An ACR of less than 30 mg/g is typically considered within the normal or mildly increased range. However, specific attention should be given to very low ACR levels, particularly those below 10 mg/g, as emerging evidence suggests that even slight elevations in ACR may be associated with an increased risk of cardiovascular and renal complications. While values below 10 mg/g generally do not require immediate intervention, regular monitoring is advised to detect early changes and guide preventive care [3], 46]. Accordingly, this underscores the importance for laboratories to select measurement procedures that can accurately detect and report these lower levels, considering the lower limit of their analytical measurement range (AMR). It is crucial to emphasize that colorimetric methods like Bromocresol Green (BCG) and Bromocresol Purple (BCP) for albuminuria measurement are not suitable because they lack the necessary sensitivity and specificity for detecting low levels of albumin in urine. These methods are prone to interference from other proteins, leading to inaccurate results, especially in the lower ranges of albuminuria (e.g., ACR<30 mg/g). Instead, immunoassay-based methods should be recommended for albuminuria measurement, as they provide greater accuracy and reliability, particularly for early detection of kidney damage.
For higher ACR values that exceed the upper measurement limit, specimens should be diluted, and the results should be adjusted by the dilution factor. If the ACR still surpasses the validated reportable range after dilution, the final value should be reported as greater than a specified threshold, calculated based on the highest valid urine albumin reporting limit divided by the creatinine concentration. This ensures that clinicians receive accurate and clinically relevant information across the full spectrum of ACR values [47], 48].
Extensive work is ongoing regarding standardization of urine albumin measurement. A reference system (reference measurement procedures (RMP), pure substance primary reference materials and commutable secondary reference materials) should be available in the coming years to improve the standardization of manufacturers’ routine assays. A recent report has shown that three candidate RMP provided suitable agreement of results and uncertainties for use as higher order reference methods [49]. This is of greatest importance since the cut-offs that are provided by KDIGO are the same, whatever the method used by the laboratories. A recent review indeed showed that, among 10 commercially available assays, either using immunoturbidimetry or immunonephelometry (which are the only methods recommended for routine urine albuminuria), eight were calibrated against the IRMM-ERM DA470 (CRM 470) standard whereas two were calibrated against an in-house serum albumin preparation [48]. Using the same calibrators with the same calibrator value assignments has been shown to significantly reduce the bias between immunoassays and LC-MS/MS methods [50]. Table 2 presents the KDIGO recommendations for implementing standards to ensure the accuracy and reliability of urine samples. Additionally, the analytical goals of imprecision for albuminuria based on biological variation have been defined as 13 % for albumin and 6 % for ACR [51].The 2023 EFLM European Urinalysis Guideline recently updated quality requirements, sample collections and performance specifications of these assays [52].
Standards to ensure the accuracy and reliability of albumin measurement in urine samples.
| Standard | Description |
|---|---|
| Sample handling | Samples for albumin measurement should be analyzed fresh or stored at 4 °C for up to 7 days. Samples should not be stored frozen at −20 °C. |
| Reporting ACR | Report albumin-to-creatinine ratio (ACR) in untimed urine samples in addition to urine albumin concentration rather than the concentrations alone. |
| Reporting precision | Report ACR to 1 decimal place, for mg/mmol and 0 decimal for mg/g. |
| Analytical precision | The analytical coefficient of variation (CV) of methods to measure urine albumin should be less than 15 %. |
| External Quality Assessment (EQA) | Implement an EQA scheme for urine albumin and creatinine, including ACR calculation. |
Point-of-care testing
Point-of-care testing (POCT) is now clearly recognized as a valuable tool for both blood creatinine and ACR measurement, particularly in countries with limited access to laboratory facilities and in remote areas within countries with sufficient resources. The KDIGO emphasizes the importance of quality check across all phases of measurement: preanalytical, analytical and postanalytical. Specialists in laboratory medicine play a key role in ensuring the accuracy and reliability of POCT by managing the entire testing process. In the preanalytical phase, they provide training to staff on proper sample collection, develop standard operating procedures (SOPs), validate POCT devices before use, and ensure adequate patient preparation. During the analytical phase, they establish and monitor quality control and calibration protocols, maintain POCT devices, and verify results for accuracy by comparing them with laboratory-based methods. In the postanalytical phase, they ensure the integration of POCT results into patient medical records for full traceability and provide ongoing training and documentation to maintain consistent quality. For ACR, POCT should ideally include the analysis of creatinine to produce an ACR result. While reagent strip urinalysis ACR PCR, whether read automatically or manually, is not preferred, it is not entirely excluded, especially in settings where validation studies have demonstrated a significant agreement with laboratory-based assays. However, if strip urinalysis is used, any positive result must be confirmed by a quantitative method to ensure accuracy. Test strip performance specifications, diagnostic significance and qualified procedures for strip readings are extensively detailed in the EFLM 2023 Urinalysis Guideline [52].
Risk assessment in people with CKD
Assessing the risk of progression of CKD is of paramount importance in clinical practice for the effective management of CKD patients. KDIGO provides practical recommendation in this context:
measure ACR and estimate GFR at least annually in CKD patients
repeat these measurements more frequently for individuals at higher risk of progression especially if it can impact therapeutic decisions
a change in eGFR of more than 20 % on a subsequent test exceeds the expected variability and warrants evaluation
doubling of ACR on a subsequent test exceeds laboratory variability and warrants evaluation.
Once again, the KDIGO recommends repeating the test, whether eGFR or ACR, to confirm significant progression. The choice of the threshold for significant changes in eGFR and ACR could be discussed, as evidence suggests that significant change may depend on the initial levels of eGFR and ACR. This is especially true for ACR, where a significant change in albuminuria can be as high as 200 % in the range of 30 mg/g or as low as 50 % in the nephrotic range (>3,000 mg/g) [53]. KDIGO’s choice to set the threshold for ACR at 100 % variation (doubling) is supported by data from studies showing an association between an ACR increase of 50–100 % and an increase risk of kidney failure [54], 55], even if sensu stricto the evidence is more based on the association between a decrease in ACR by therapeutic intervention and improved renal outcomes [56]. As specialists in laboratory medicine, we might also regret the lack of discernment behind the term “laboratory variability,” which encompasses not only analytical variability but, more importantly, individual variability.
A major new and high graded (1A) recommendation by KDIGO is the use of an externally validated risk equation to estimate the absolute risk of kidney failure. From this risk calculation, several practical decisions for the patients can be drawn:
A 2-year kidney failure risk of >10 % can guide the timing of multidisciplinary care [60].
However, more data are needed to support these practical recommendations as they are currently based on a relatively few [57], [58], [59], [60], [61], [62], and sometimes controversial [63], 64] studies. Additionally, KDIGO emphasizes that the risk equations developed in patients with eGFR<60 mL/min/1.73 m2 might not be valid for those with higher GFR levels for whom other models have been recently developed [65], [66], [67] and are currently validated [68], [69], [70]. Different disease-specific equations can also be used like in immunoglobulin A nephropathy [71], 72] or polycystic kidney diseases [73]. In the same vein, several equations specifically dedicated to CKD patients (and including eGFR and ACR) are available to estimate the risk of cardiovascular or all-cause mortality [1].
Coming back to estimation of risk of kidney failure, different equations are proposed for CKD patients (i.e. those with eGFR below 60 mL/min/1.73 m2). For the externally validated equations [69], [74], [75], [76], [77], [78], it is beyond the scope of this article to discuss the strengths and limitations of each of them [79], 80]. As a matter of fact, the most commonly used equation is the Kidney Failure Risk Equation (KFRE) (www.kidneyfailurerisk.com) [74]. The variables included in the KFRE are age, sex, eGFR, ACR and region (North America vs. non-North America). If available, additional (but non-essential) variables like albumin, phosphorus, bicarbonate and (not “or” as all variables must be considered) corrected calcium can be used in the calculator. The equation provides a percentage risk of kidney failure at 2 and 5 years. Given that these variables are available to clinical laboratories, it could be feasible to automatically provide a risk prediction for patients with eGFR<60 mL/min/1.73 m2 when ACR is prescribed.
The proliferation of risk equations (one for risk of kidney failure in CKD patients, another for risk failure prediction in non-CKD, disease-specific equations, and other equation for prediction of the risk of cardiovascular or all-cause mortality) can obviously be a source of confusion. Clinicians may also find it challenging to use both the well-known “heatmap” based on ACR and eGFR, and risk prediction equations. The heatmap mirrors pure associations between GFR/ACR and adverse outcomes. It reflects the relative risk for populations as classified in each box whereas the risk prediction equations are intended to evaluate the absolute risk at the individual level. Risk prediction is thus presented as a step towards personalized medicine, even though using both heatmaps and prediction equations in clinical practice can be challenging and seemingly duplicative. All risk prediction equations include also the “age” variable which could be the first step in reconciling KDIGO with proponents of an age-adapted CKD definition [81].
Conclusions
The 2024 KDIGO guideline offers a comprehensive framework for the evaluation and management of CKD, with significant implications for laboratory practice. Key updates include the promotion of cystatin C as a complementary marker to creatinine for eGFR, the use of mGFR in specific clinical situations, the acceptance of alternative equations beyond CKD-EPI, the central role of ACR in CKD management and prognosis, and the recommendation of individual risk assessment equations for CKD patients. These updates have significant implications for European laboratories and align well with the findings of the 2022 EFLM Task Group on CKD survey, emphasizing the need for harmonized and improved CKD testing practices across Europe [20]. We can summarize these implications in a few points:
The EFLM TG-CKD survey highlighted significant disparities in CKD testing practices across European laboratories, leading to inconsistent patient care. The KDIGO 2024 guidelines recommend harmonization at the geographical level. European laboratories should prioritize adopting these recommendations, ensuring the use of standardized equations validated within European populations, such as the EKFC equations. We strongly advocate against the widespread adoption of the 2021 CKD-EPI equation in Europe due to concerns over its applicability and accuracy.
The KDIGO guideline promotes cystatin C not only as an alternative but as a complementary marker to creatinine for GFR estimation. This is particularly relevant given the limited use of cystatin C in many European countries due to cost and lack of reimbursement. Efforts should be made to increase the availability and use of cystatin C testing, advocating for its reimbursement and emphasizing its clinical value. Laboratories should ensure that when both creatinine and cystatin C are measured, the combined eGFR equation is reported to enhance diagnostic accuracy.
The survey identified significant variations in the methods and units used for measuring creatinine and cystatin C. To improve the accuracy and reliability of these measurements, European laboratories should use enzymatic methods for creatinine measurement, especially in children, standardize the units for reporting creatinine and cystatin C concentrations at the regional level, and ensure uniformity within regions to minimize discrepancies and improve patient care continuity.
While the KDIGO guideline highlights the importance of iohexol clearance as a reference method for measuring GFR, the survey revealed limited availability of this technique in European laboratories. It is crucial to expand the capacity for reference GFR measurements, ensuring that at least one laboratory in each country can perform iohexol clearance. This will provide a reliable benchmark for validating estimated GFR results and improving clinical decision-making.
The survey highlighted confusion regarding albuminuria measurements, with variations in units and cut-offs. KDIGO guidelines recommend using the albumin-to-creatinine ratio (ACR) as the preferred measure, expressed in mg/g or mg/mmol. Laboratories should abandon the term “microalbuminuria” and adopt these standardized practices to ensure consistency and clarity in reporting results.
The combination of our knowledge of the European situation from the survey helps us draw future directions to better comply with the 2024 KDIGO recommendations. The first one is related to participation in EQA programs. KDIGO recommends implementing external quality assessment programs for urine albumin and creatinine. European laboratories should participate in these programs to benchmark their performance and continuously improve their testing standards. The new KDIGO guideline emphasizes the use of externally validated risk equations to estimate the absolute risk of kidney failure. Laboratories should integrate these risk prediction tools into their reporting systems, providing clinicians with valuable insights to guide individual patient management decisions. This aligns with the move towards personalized medicine, where individualized risk assessments can significantly impact treatment outcomes. Finally, it is essential to provide education and training for laboratory professionals on the updated KDIGO guideline and its implementation. This will ensure that all staff are proficient in the new testing protocols and understand the importance of harmonization in improving patient care.
By adopting these recommendations and embracing the updates from the KDIGO 2024 guidelines, European laboratories can enhance their role in CKD management, ensuring accurate diagnostics, better patient outcomes, and a cohesive approach across the continent. As the field of nephrology continues to evolve, ongoing collaboration between clinicians and specialists in laboratory medicine will be essential to address emerging challenges and improve the standard of care for CKD patients.
-
Research ethics: Not applicable.
-
Informed consent: Not applicable.
-
Author contributions: All authors have accepted responsibility for the entire content of this manuscript and approved its submission.
-
Use of Large Language Models, AI and Machine Learning Tools: None declared.
-
Conflict of interest: EC and PD are consultant for Nephrolyx. All other authors state no conflict of interest.
-
Research funding: None declared.
-
Data availability: Not applicable.
References
1. KDIGO 2024. Clinical Practice Guideline for the evaluation and management of chronic kidney disease. Kidney Int Suppl 2024;105:S1–314.10.1016/j.kint.2023.09.002Search in Google Scholar PubMed
2. KDIGO 2012. Clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int Suppl 2013;3:1–150.Search in Google Scholar
3. Grams, ME, Coresh, J, Matsushita, K, Ballew, SH, Sang, Y, Surapaneni, A, et al.. Estimated glomerular filtration rate, albuminuria, and adverse outcomes: an individual-participant data meta-analysis. JAMA; 2023, 330:1266–77 pp. https://doi.org/10.1001/jama.2023.17002.Search in Google Scholar PubMed PubMed Central
4. Delanaye, P, Jager, KJ, Bökenkamp, A, Christensson, A, Dubourg, L, Eriksen, BO, et al.. CKD: a call for an age-adapted definition. J Am Soc Nephrol 2019;30:1785–805. https://doi.org/10.1681/asn.2019030238.Search in Google Scholar
5. Jonsson, AJ, Lund, SH, Eriksen, BO, Palsson, R, Indridason, OS. The prevalence of chronic kidney disease in Iceland according to KDIGO criteria and age-adapted estimated glomerular filtration rate thresholds. Kidney Int. Elsevier Inc 2020;98:1286–95. https://doi.org/10.1016/j.kint.2020.06.017.Search in Google Scholar PubMed
6. Liu, P, Quinn, RR, Lam, NN, Elliott, MJ, Xu, Y, James, MT, et al.. Accounting for age in the definition of chronic kidney disease. JAMA Intern Med 2021;181:1359–66. https://doi.org/10.1001/jamainternmed.2021.4813.Search in Google Scholar PubMed PubMed Central
7. Delanaye, P, Cavalier, E. Staging chronic kidney disease and estimating glomerular filtration rate: an opinion paper about the new international recommendations. Clin Chem Lab Med 2013;51:1911–7. https://doi.org/10.1515/cclm-2013-0223.Search in Google Scholar PubMed
8. Delanaye, P. Too much nephrology? The CKD epidemic is real and concerning. A CON view. Nephrol Dial Transpl 2019;34:581–4. https://doi.org/10.1093/ndt/gfy331.Search in Google Scholar PubMed
9. Gansevoort, RT. Too much nephrology? The CKD epidemic is real and concerning. A PRO view. Nephrol Dial Transpl 2019;34:577–80. https://doi.org/10.1093/ndt/gfy330.Search in Google Scholar PubMed
10. Zoccali, C, Santoro, A, Plebani, M. Age, stage and biomarkers for the definition of CKD: a construction in progress. Clin Chem Lab Med 2013;51:1–5. https://doi.org/10.1515/cclm-2013-0431.Search in Google Scholar PubMed
11. Eriksen, BO, Palsson, R, Ebert, N, Melsom, T, van der Giet, M, Gudnason, V, et al.. GFR in healthy aging: an individual participant data meta-analysis of iohexol clearance in european population-based cohorts. J Am Soc Nephrol 2020;31:1602–15. https://doi.org/10.1681/asn.2020020151.Search in Google Scholar PubMed PubMed Central
12. Delanaye, P, Glassock, RJ, De Broe, ME. Epidemiology of chronic kidney disease: think (at least) twice. Clin Kidney J 2017;10:370–4. https://doi.org/10.1093/ckj/sfw154.Search in Google Scholar PubMed PubMed Central
13. Benghanem Gharbi, M, Elseviers, M, Zamd, M, Belghiti Alaoui, A, Benahadi, N, Trabelssi, EH, et al.. Chronic kidney disease, hypertension, diabetes, and obesity in the adult population of Morocco: how to avoid “over”- and “under”-diagnosis of CKD. Kidney Int 2016;89:1363–71. https://doi.org/10.1016/j.kint.2016.02.019.Search in Google Scholar PubMed
14. Fu, EL, Levey, AS, Coresh, J, Elinder, C, Rotmans, JI, Dekker, FW, et al.. Accuracy of GFR estimating equations in patients with discordances between creatinine and cystatin C-based estimations. J Am Soc Nephrol 2023;34:1241–51. https://doi.org/10.1681/asn.0000000000000128.Search in Google Scholar
15. Pottel, H, Björk, J, Rule, AD, Ebert, N, Eriksen, BO, Dubourg, L, et al.. Cystatin C–based equation to estimate GFR without the inclusion of race and sex. N Engl J Med 2023;388:333–43. https://doi.org/10.1056/nejmoa2203769.Search in Google Scholar
16. Pottel, H, Björk, J, Courbebaisse, M, Couzi, L, Ebert, N, Eriksen, BO, et al.. Development and validation of a modified full age spectrum creatinine-based equation to estimate glomerular filtration rate. A cross-sectional analysis of pooled data. Ann Intern Med 2021;174:183–91. https://doi.org/10.7326/m20-4366.Search in Google Scholar
17. Inker, LA, Eneanya, ND, Coresh, J, Tighiouart, H, Wang, D, Sang, Y, et al.. New creatinine- and cystatin C–based equations to estimate GFR without race. N Engl J Med 2021;385:1737–49. https://doi.org/10.1056/nejmoa2102953.Search in Google Scholar PubMed PubMed Central
18. Inker, LA, Schmid, CH, Tighiouart, H, Eckfeldt, JH, Feldman, HI, Greene, T, et al.. Estimating glomerular filtration rate from serum creatinine and cystatin C. N Engl J Med 2012;367:20–9. https://doi.org/10.1056/nejmoa1114248.Search in Google Scholar
19. Björk, J, Bäck, SE, Ebert, N, Evans, M, Grubb, A, Hansson, M, et al.. GFR estimation based on standardized creatinine and cystatin C: a European multicenter analysis in older adults. Clin Chem Lab Med 2018;56:422–35. https://doi.org/10.1515/cclm-2017-0563.Search in Google Scholar PubMed
20. Cavalier, E, Makris, K, Portakal, O, Nikler, A, Datta, P, Zima, T, et al.. Assessing the status of European laboratories in evaluating biomarkers for chronic kidney diseases (CKD) and recommendations for improvement: insights from the 2022 EFLM Task Group on CKD survey. Clin Chem Lab Med 2024;62:253–61. https://doi.org/10.1515/cclm-2023-0987.Search in Google Scholar PubMed
21. Stehlé, T, Ouamri, Y, Morel, A, Vidal-Petiot, E, Fellahi, S, Segaux, L, et al.. Development and validation of a new equation based on plasma creatinine and muscle mass assessed by CT-scan to estimate glomerular filtration rate: a cross-sectional study. Clin Kidney J 2023;16:1265–77. https://doi.org/10.1093/ckj/sfad012.Search in Google Scholar PubMed PubMed Central
22. Adingwupu, OM, Barbosa, ER, Palevsky, PM, Vassalotti, JA, Levey, AS, Inker, LA. Cystatin C as a GFR estimation marker in acute and chronic illness: a systematic review. Kidney Med. 2023;5. https://doi.org/10.1016/j.xkme.2023.100727.Search in Google Scholar PubMed PubMed Central
23. Inker, LA, Schmid, CH, Greene, T, Li, L, Beck, GJ, Joffe, MM, et al.. Factors other than glomerular filtration rate affect serum cystatin C levels. Kidney Int 2009;75:652–60. https://doi.org/10.1038/ki.2008.638.Search in Google Scholar PubMed PubMed Central
24. Delanaye, P, Pottel, H, Cavalier, E, Flamant, M, Stehlé, T, Mariat, C. Diagnostic standard: assessing glomerular filtration rate. Nephrol Dial Transpl 2024;39:1088–96. https://doi.org/10.1093/ndt/gfad241.Search in Google Scholar PubMed
25. Ebert, N, Bevc, S, Bökenkamp, A, Gaillard, F, Hornum, M, Jager, KJ, et al.. Assessment of kidney function : clinical indications for measured GFR. Clin Kidney J 2021;14:1861–70. https://doi.org/10.1093/ckj/sfab042.Search in Google Scholar PubMed PubMed Central
26. Agarwal, R, Delanaye, P. Glomerular filtration rate: when to measure and in which patients? Nephrol Dial Transpl 2019;34:2001–7. https://doi.org/10.1093/ndt/gfy363.Search in Google Scholar PubMed
27. Nyssen, L, Delanaye, P, Le Goff, C, Peeters, S, Cavalier, É. A simple LC-MS method for the determination of iohexol and iothalamate in serum, using ioversol as an internal standard. Clin Chim Acta 2016;463:96–102. https://doi.org/10.1016/j.cca.2016.10.021.Search in Google Scholar PubMed
28. Gaspari, F, Thakar, S, Carrara, F, Perna, A, Trillini, M, Aparicio, MC, et al.. Safety of iohexol administration to measure glomerular filtration rate in different patient populations: a 25-year experience. Nephron 2018;140:1–8. https://doi.org/10.1159/000489898.Search in Google Scholar PubMed
29. Nordin, G, Ekvall, S, Kristoffersson, C, Jonsson, A-S, Bäck, S-E, Rollborn, N, et al.. Accuracy of determination of the glomerular filtration marker iohexol by European laboratories as monitored by external quality assessment. Clin Chem Lab Med 2019;57:1006–11. https://doi.org/10.1515/cclm-2018-1175.Search in Google Scholar PubMed
30. Ebert, N, Schaeffner, E, Seegmiller, JC, Londen, M, Bökenkamp, A, Cavalier, E, et al.. Iohexol plasma clearance measurement protocol standardization for adults – a consensus paper of the European kidney function consortium. Kidney Int 2024;106:583–96. https://doi.org/10.1016/j.kint.2024.06.029.Search in Google Scholar PubMed
31. Levey, AS, Stevens, LA, Schmid, CH, Zhang, YL, Castro, AFIII, Feldman, HI, et al.. A new equation to estimate glomerular filtration rate. Ann Intern Med 2009;150:604–12. https://doi.org/10.7326/0003-4819-150-9-200905050-00006.Search in Google Scholar PubMed PubMed Central
32. Eneanya, ND, Boulware, LE, Tsai, J, Bruce, MA, Ford, CL, Harris, C, et al.. Health inequities and the inappropriate use of race in nephrology. Nat Rev Nephrol 2022;18:84–94. https://doi.org/10.1038/s41581-021-00501-8.Search in Google Scholar PubMed PubMed Central
33. Delanaye, P, Pottel, H, Glassock, RJ. Americentrism in estimation of GFR equations. Kidney Int 2022;101:856–8. https://doi.org/10.1016/j.kint.2022.02.022.Search in Google Scholar PubMed
34. Delanaye, P, Mariat, C, Cavalier, E, Glassock, RJ, Gemenne, F, Pottel, H. The « race » correction in estimating glomerular filtration rate. Curr Opin Nephrol Hypertens 2021;30:525–30. https://doi.org/10.1097/mnh.0000000000000739.Search in Google Scholar
35. Flamant, M, Vidal-Petiot, E, Metzger, M, Haymann, JP, Letavernier, E, Delatour, V, et al.. Performance of GFR estimating equations in African Europeans: basis for a lower race-ethnicity factor than in African Americans. Am J Kidney Dis 2013;62:182–4. https://doi.org/10.1053/j.ajkd.2013.03.015.Search in Google Scholar PubMed
36. Delanaye, P, Vidal-Petiot, E, Björk, J, Ebert, N, Eriksen, B, Dubourg, L, et al.. Performance of creatinine-based equations to estimate glomerular filtration rate in White and Black populations in Europe, Brazil, and Africa. Nephrol Dial Transpl 2023;38:106–18. https://doi.org/10.1093/ndt/gfac241.Search in Google Scholar PubMed
37. Bukabau, JB, Yayo, E, Gnionsahé, A, Monnet, D, Pottel, H, Cavalier, E, et al.. Performance of creatinine- or cystatin C–based equations to estimate glomerular filtration rate in sub-Saharan African populations. Kidney Int 2019;95:1181–9. https://doi.org/10.1016/j.kint.2018.11.045.Search in Google Scholar PubMed
38. Fabian, J, Kalyesubula, R, Mkandawire, J, Hansen, CH, Nitsch, D, Musenge, E, et al.. Measurement of kidney function in Malawi, South Africa, and Uganda: a multicentre cohort study. Lancet Glob Health 2022;10:e1159–69. https://doi.org/10.1016/s2214-109x(22)00239-x.Search in Google Scholar
39. Gansevoort, RT, Anders, H-J, Cozzolino, M, Fliser, D, Ortiz, A, Soler, MJ, et al.. What should European nephrology do with the new CKD-EPI equation? Nephrol Dial Transpl 2022;38:1–6. https://doi.org/10.1093/ndt/gfac254.Search in Google Scholar PubMed PubMed Central
40. Delanaye, P, Schaeffner, E, Cozzolino, M, Langlois, M, Plebani, M, Ozben, T, et al.. The new, race-free, chronic kidney disease epidemiology consortium (CKD-EPI) equation to estimate glomerular filtration rate: is it applicable in Europe? A position statement by the European Federation of clinical chemistry and laboratory medicine (EFLM). Clin Chem Lab Med 2023;61:44–7. https://doi.org/10.1515/cclm-2022-0928.Search in Google Scholar PubMed
41. Fu, EL, Levey, AS, Coresh, J, Grams, ME, Faucon, A-L, Elinder, C-G, et al.. Accuracy of GFR estimating equations based on creatinine, cystatin C or both in routine care. Nephrol Dial Transpl 2024;39:694–706. https://doi.org/10.1093/ndt/gfad219.Search in Google Scholar PubMed
42. Delanaye, P, Rule, AD, Schaeffner, ES, Cavalier, E, Shi, J, Hoofnagle, AN, et al.. Performance of the European kidney function consortium (EKFC) creatinine-based equation in American cohorts. Kidney Int 2024;105:629–37. https://doi.org/10.1016/j.kint.2023.11.024.Search in Google Scholar PubMed
43. Ma, Y, Wei, L, Yong, Z, Yu, Y, Chen, Y, Zhu, B, et al.. Validation of the European Kidney Function Consortium (EKFC) equation in Chinese adult population: an equation standing on the shoulders of predecessors. Nephron 2023. In process. https://doi.org/10.1159/000531030.Search in Google Scholar PubMed
44. Delanaye, P, Cavalier, E, Stehlé, T, Pottel, H. Glomerular filtration rate estimation in adults: myths and promises. Nephron 2024;148:408–14. https://doi.org/10.1159/000536243.Search in Google Scholar PubMed
45. Delanaye, P, Cavalier, E, Pottel, H, Stehlé, T. New and old GFR equations: a European perspective. Clin Kidney J. 2023;16:1375–83. https://doi.org/10.1093/ckj/sfad039.Search in Google Scholar PubMed PubMed Central
46. Matsushita, K, van der Velde, M, Astor, BC, Woodward, M, Levey, AS, de Jong, PE, et al.. Association of estimated glomerular filtration rate and albuminuria with all-cause and cardiovascular mortality in general population cohorts: a collaborative meta-analysis. Lancet 2010;375:2073–81. https://doi.org/10.1016/S0140-6736(10)60674-5.Search in Google Scholar PubMed PubMed Central
47. Miller, WG, Bachmann, LM, Fleming, JK, Delanghe, JR, Parsa, A, Narva, AS, et al.. Recommendations for reporting low and high values for urine albumin and total protein. Clin Chem 2019;65:349–50. https://doi.org/10.1373/clinchem.2018.297861.Search in Google Scholar PubMed
48. Seegmiller, JC, Bachmann, LM. Urine albumin measurements in clinical diagnostics. Clin Chem 2024;70:382–91. https://doi.org/10.1093/clinchem/hvad174.Search in Google Scholar PubMed
49. Miller, WG, Bachmann, LM, Budd, J, Beasley-Green, A, Phinney, KW, Tan, HT, et al.. Extent of equivalence of results for urine albumin among 3 candidate mass spectrometry reference measurement procedures. Clin Chem 2024:hvae122.10.1093/clinchem/hvae122Search in Google Scholar PubMed
50. Shaikh, A, Seegmiller, JC, Borland, TM, Burns, BE, Ladwig, PM, Singh, RJ, et al.. Comparison between immunoturbidimetry, size-exclusion chromatography, and LC-MS to quantify urinary albumin. Clin Chem 2008;54:1504–10. https://doi.org/10.1373/clinchem.2008.107508.Search in Google Scholar PubMed PubMed Central
51. Mosca, A, Paleari, R, Ceriotti, F, Lapolla, A, Fedele, D, Group, LS. Biological variability of albumin excretion rate and albumin-to-creatinine ratio in hypertensive type 2 diabetic patients. Clin Chem Lab Med 2003;41:1229–33. https://doi.org/10.1515/cclm.2003.188.Search in Google Scholar
52. Kouri, TT, Hofmann, W, Falbo, R, Oyaert, M, Schubert, S, Gertsen, JB, et al.. The EFLM European urinalysis guideline 2023. Clin Chem Lab Med 2024;62:1653–786. https://doi.org/10.1515/cclm-2024-0070.Search in Google Scholar PubMed
53. Naresh, CN, Hayen, A, Weening, A, Craig, JC, Chadban, SJ. Day-to-day variability in spot urine albumin-creatinine ratio. Am J Kidney Dis 2013;62:1095–101. https://doi.org/10.1053/j.ajkd.2013.06.016.Search in Google Scholar PubMed
54. Norris, KC, Smoyer, KE, Rolland, C, Van der Vaart, J, Grubb, EB. Albuminuria, serum creatinine, and estimated glomerular filtration rate as predictors of cardio-renal outcomes in patients with type 2 diabetes mellitus and kidney disease: a systematic literature review. BMC Nephrol 2018;19:36. https://doi.org/10.1186/s12882-018-0821-9.Search in Google Scholar PubMed PubMed Central
55. Berhane, AM, Weil, EJ, Knowler, WC, Nelson, RG, Hanson, RL. Albuminuria and estimated glomerular filtration rate as predictors of diabetic end-stage renal disease and death. Clin J Am Soc Nephrol 2011;6:2444–51. https://doi.org/10.2215/cjn.00580111.Search in Google Scholar PubMed PubMed Central
56. Lambers Heerspink, HJ, Gansevoort, RT. Albuminuria is an appropriate therapeutic target in patients with CKD: the pro view. Clin J Am Soc Nephrol 2015;10:1079–88. https://doi.org/10.2215/cjn.11511114.Search in Google Scholar PubMed PubMed Central
57. Hingwala, J, Wojciechowski, P, Hiebert, B, Bueti, J, Rigatto, C, Komenda, P, et al.. Risk-based triage for nephrology referrals using the kidney failure risk equation. Can J Kidney Health Dis 2017;4. https://doi.org/10.1177/2054358117722782.Search in Google Scholar PubMed PubMed Central
58. Che, M, Iliescu, E, Thanabalasingam, S, Day, AG, White, CA. Death and dialysis following discharge from chronic kidney disease clinic: a retrospective cohort study. Can J Kidney Health Dis. 2022;9. https://doi.org/10.1177/20543581221118434.Search in Google Scholar PubMed PubMed Central
59. Bhachu, HK, Cockwell, P, Subramanian, A, Adderley, NJ, Gokhale, K, Fenton, A, et al.. Impact of using risk-based stratification on referral of patients with chronic kidney disease from primary care to specialist care in the United Kingdom. Kidney Int Rep 2021;6:2189–99. https://doi.org/10.1016/j.ekir.2021.05.031.Search in Google Scholar PubMed PubMed Central
60. Smekal, MD, Tam-Tham, H, Finlay, J, Donald, M, Thomas, C, Weaver, RG, et al.. Patient and provider experience and perspectives of a risk-based approach to multidisciplinary chronic kidney disease care: a mixed methods study. BMC Nephrol 2019;20:110. https://doi.org/10.1186/s12882-019-1269-2.Search in Google Scholar PubMed PubMed Central
61. Lundström, UH, Ramspek, CL, Dekker, FW, van Diepen, M, Carrero, JJ, Hedin, U, et al.. Clinical impact of the kidney failure risk equation for vascular access planning. Nephrol Dial Transpl 2024. Online ahead of print. https://doi.org/10.1093/ndt/gfae064.Search in Google Scholar PubMed PubMed Central
62. Atiquzzaman, M, Zhu, B, Romann, A, Er, L, Djurdjev, O, Bevilacqua, M, et al.. Kidney Failure Risk Equation in vascular access planning: a population-based study supporting value in decision making. Clin Kidney J 2024;17:sfae008. https://doi.org/10.1093/ckj/sfae008.Search in Google Scholar PubMed PubMed Central
63. Schulz, C, Messikh, Z, Reboul, P, Cariou, S, Ahmadpoor, P, Pambrun, E, et al.. Characteristics of outpatients referred for a first consultation with a nephrologist: impact of different guidelines. J Nephrol 2022;35:1375–85. https://doi.org/10.1007/s40620-021-01204-w.Search in Google Scholar PubMed
64. Kuningas, K, Stringer, S, Cockwell, P, Khawaja, A, Inston, N. Is there a role of the kidney failure risk equation in optimizing timing of vascular access creation in pre-dialysis patients? J Vasc Access 2023;24:1305–13. https://doi.org/10.1177/11297298221084799.Search in Google Scholar PubMed
65. Grams, ME, Brunskill, NJ, Ballew, SH, Sang, Y, Coresh, J, Matsushita, K, et al.. Development and validation of prediction models of adverse kidney outcomes in the population with and without diabetes. Diabetes Care 2022;45:2055–63. https://doi.org/10.2337/dc22-0698.Search in Google Scholar PubMed PubMed Central
66. Ferguson, T, Ravani, P, Sood, MM, Clarke, A, Komenda, P, Rigatto, C, et al.. Development and external validation of a machine learning model for progression of CKD. Kidney Int Rep 2022;7:1772–81. https://doi.org/10.1016/j.ekir.2022.05.004.Search in Google Scholar PubMed PubMed Central
67. Chan, L, Nadkarni, GN, Fleming, F, McCullough, JR, Connolly, P, Mosoyan, G, et al.. Derivation and validation of a machine learning risk score using biomarker and electronic patient data to predict progression of diabetic kidney disease. Diabetologia 2021;64:1504–15. https://doi.org/10.1007/s00125-021-05444-0.Search in Google Scholar PubMed PubMed Central
68. Tangri, N, Ferguson, TW, Bamforth, RJ, Leon, SJ, Arnott, C, Mahaffey, KW, et al.. Machine learning for prediction of chronic kidney disease progression: validation of the Klinrisk model in the CANVAS Program and CREDENCE trial. Diabetes Obes Metab 2024. Online ahead of print. https://doi.org/10.1111/dom.15678.Search in Google Scholar PubMed
69. Grams, ME, Brunskill, NJ, Ballew, SH, Sang, Y, Coresh, J, Matsushita, K, et al.. The kidney failure risk equation: evaluation of novel input variables including eGFR estimated using the CKD-EPI 2021 equation in 59 cohorts. J Am Soc Nephrol 2023;34:482–94. https://doi.org/10.1681/ASN.0000000000000050.Search in Google Scholar PubMed PubMed Central
70. Tio, MC, Butler, J, Zhu, X, Obi, Y, Yen, TE, Kalantar-Zadeh, K, et al.. Individualized risk of CKD progression among US adults. J Am Soc Nephrol 2024. Online ahead of print. https://doi.org/10.1681/asn.0000000000000377.Search in Google Scholar
71. Berthoux, F, Mohey, H, Laurent, B, Mariat, C, Afiani, A, Thibaudin, L. Predicting the risk for dialysis or death in IgA nephropathy. J Am Soc Nephrol 2011;22:752–61. https://doi.org/10.1681/asn.2010040355.Search in Google Scholar
72. Barbour, SJ, Coppo, R, Zhang, H, Liu, Z-H, Suzuki, Y, Matsuzaki, K, et al.. Evaluating a new international risk-prediction tool in IgA nephropathy. JAMA Intern Med 2019;179:942–52. https://doi.org/10.1001/jamainternmed.2019.0600.Search in Google Scholar PubMed PubMed Central
73. Cornec-Le Gall, E, Audrézet, M-P, Rousseau, A, Hourmant, M, Renaudineau, E, Charasse, C, et al.. The PROPKD score: a new algorithm to predict renal survival in autosomal dominant polycystic kidney disease. J Am Soc Nephrol 2016;27:942–51. https://doi.org/10.1681/ASN.2015010016.Search in Google Scholar PubMed PubMed Central
74. Tangri, N, Stevens, LA, Griffith, J, Tighiouart, H, Djurdjev, O, Naimark, D, et al.. A predictive model for progression of chronic kidney disease to kidney failure. JAMA 2011;305:1553–9. https://doi.org/10.1001/jama.2011.451.Search in Google Scholar PubMed
75. Zacharias, HU, Altenbuchinger, M, Schultheiss, UT, Raffler, J, Kotsis, F, Ghasemi, S, et al.. A predictive model for progression of CKD to kidney failure based on routine laboratory tests. Am J Kidney Dis 2022;79:217–30.e1. https://doi.org/10.1053/j.ajkd.2021.05.018.Search in Google Scholar PubMed
76. Schroeder, EB, Yang, X, Thorp, ML, Arnold, BM, Tabano, DC, Petrik, AF, et al.. Predicting 5-year risk of RRT in stage 3 or 4 CKD: development and external validation. Clin J Am Soc Nephrol 2017;12:87–94. https://doi.org/10.2215/cjn.01290216.Search in Google Scholar
77. Landray, MJ, Thambyrajah, J, McGlynn, FJ, Jones, HJ, Baigent, C, Kendall, MJ, et al.. Epidemiological evaluation of known and suspected cardiovascular risk factors in chronic renal impairment. Am J Kidney Dis 2001;38:537–46. https://doi.org/10.1053/ajkd.2001.26850.Search in Google Scholar PubMed
78. Major, RW, Shepherd, D, Medcalf, JF, Xu, G, Gray, LJ, Brunskill, NJ. The Kidney Failure Risk Equation for prediction of end stage renal disease in UK primary care: an external validation and clinical impact projection cohort study. PLoS Med 2019;16:e1002955. https://doi.org/10.1371/journal.pmed.1002955.Search in Google Scholar PubMed PubMed Central
79. Ramspek, CL, de Jong, Y, Dekker, FW, van Diepen, M. Towards the best kidney failure prediction tool: a systematic review and selection aid. Nephrol Dial Transpl 2020;35:1527–38. https://doi.org/10.1093/ndt/gfz018.Search in Google Scholar PubMed PubMed Central
80. Tangri, N, Kitsios, GD, Inker, LA, Griffith, J, Naimark, DM, Walker, S, et al.. Risk prediction models for patients with chronic kidney disease: a systematic review. Ann Intern Med 2013;158:596–603. https://doi.org/10.7326/0003-4819-158-8-201304160-00004.Search in Google Scholar PubMed
81. Oliva-Damaso, N, Delanaye, P, Oliva-Damaso, E, Payan, J, Glassock, RJ. Risk-based versus GFR threshold criteria for nephrology referral in chronic kidney disease. Clin Kidney J 2022;15:1996–2005. https://doi.org/10.1093/ckj/sfac104.Search in Google Scholar PubMed PubMed Central
© 2024 Walter de Gruyter GmbH, Berlin/Boston
Articles in the same Issue
- Frontmatter
- Editorials
- Multi-cancer early detection: searching for evidence
- High sensitivity cardiac troponin assays, rapid myocardial infarction rule-out algorithms, and assay performance
- Reviews
- Consensus statement on extracellular vesicles in liquid biopsy for advancing laboratory medicine
- Copeptin as a diagnostic and prognostic biomarker in pediatric diseases
- Opinion Papers
- The Unholy Grail of cancer screening: or is it just about the Benjamins?
- Critical appraisal of the CLSI guideline EP09c “measurement procedure comparison and bias estimation using patient samples”
- Tumor markers determination in malignant pleural effusion: pearls and pitfalls
- Contribution of laboratory medicine and emerging technologies to cardiovascular risk reduction via exposome analysis: an opinion of the IFCC Division on Emerging Technologies
- Guidelines and Recommendations
- Recommendations for European laboratories based on the KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease
- Genetics and Molecular Diagnostics
- Expanded carrier screening for 224 monogenic disease genes in 1,499 Chinese couples: a single-center study
- General Clinical Chemistry and Laboratory Medicine
- How do experts determine where to intervene on test ordering? An interview study
- New concept for control material in glucose point-of-care-testing for external quality assessment schemes
- Vitamin B12 deficiency in newborns: impact on individual’s health status and healthcare costs
- Analytical evaluation of eight qualitative FIT for haemoglobin products, for professional use in the UK
- Colorimetric correcting for sample concentration in stool samples
- Reference Values and Biological Variations
- Assessment of canonical diurnal variations in plasma glucose using quantile regression modelling and Chronomaps
- Inconsistency in ferritin reference intervals across laboratories: a major concern for clinical decision making
- Establishing the TSH reference intervals for healthy adults aged over 70 years: the Australian ASPREE cohort study
- Hematology and Coagulation
- The EuroFlow PIDOT external quality assurance scheme: enhancing laboratory performance evaluation in immunophenotyping of rare lymphoid immunodeficiencies
- Clinical value of smear review of flagged samples analyzed with the Sysmex XN hematology analyzer
- Cardiovascular Diseases
- Evidence for stability of cardiac troponin T concentrations measured with a high sensitivity TnT test in serum and lithium heparin plasma after six-year storage at −80 °C and multiple freeze-thaw cycles
- Letters to the Editor
- Impact of high-sensitivity cardiac troponin I assay imprecision on the safety of a single-sample rule-out approach for myocardial infarction
- Why is single sample rule out of non-ST elevation myocardial infarction using high-sensitivity cardiac troponin T safe when analytical imprecision is so high? A joint statistical and clinical demonstration
- Iron deficiency and iron deficiency anemia in transgender populations: what’s different?
- The information about the metrological traceability pedigree of the in vitro diagnostic calibrators should be improved: the case of plasma ethanol
- Time to refresh and integrate the JCTLM database entries for total bilirubin: the way forward
- Navigation between EQA and sustainability
- C-terminal alpha-1-antitrypsin peptides as novel predictor of hospital mortality in critically ill COVID-19 patients
- Neutralizing antibodies against KP.2 and KP.3: why the current vaccine needs an update
- A simple gatekeeping intervention improves the appropriateness of blood urea nitrogen testing
- Congress Abstracts
- 16ª Reunião Científica da Sociedade Portuguesa de Medicina Laboratorial - SPML
Articles in the same Issue
- Frontmatter
- Editorials
- Multi-cancer early detection: searching for evidence
- High sensitivity cardiac troponin assays, rapid myocardial infarction rule-out algorithms, and assay performance
- Reviews
- Consensus statement on extracellular vesicles in liquid biopsy for advancing laboratory medicine
- Copeptin as a diagnostic and prognostic biomarker in pediatric diseases
- Opinion Papers
- The Unholy Grail of cancer screening: or is it just about the Benjamins?
- Critical appraisal of the CLSI guideline EP09c “measurement procedure comparison and bias estimation using patient samples”
- Tumor markers determination in malignant pleural effusion: pearls and pitfalls
- Contribution of laboratory medicine and emerging technologies to cardiovascular risk reduction via exposome analysis: an opinion of the IFCC Division on Emerging Technologies
- Guidelines and Recommendations
- Recommendations for European laboratories based on the KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease
- Genetics and Molecular Diagnostics
- Expanded carrier screening for 224 monogenic disease genes in 1,499 Chinese couples: a single-center study
- General Clinical Chemistry and Laboratory Medicine
- How do experts determine where to intervene on test ordering? An interview study
- New concept for control material in glucose point-of-care-testing for external quality assessment schemes
- Vitamin B12 deficiency in newborns: impact on individual’s health status and healthcare costs
- Analytical evaluation of eight qualitative FIT for haemoglobin products, for professional use in the UK
- Colorimetric correcting for sample concentration in stool samples
- Reference Values and Biological Variations
- Assessment of canonical diurnal variations in plasma glucose using quantile regression modelling and Chronomaps
- Inconsistency in ferritin reference intervals across laboratories: a major concern for clinical decision making
- Establishing the TSH reference intervals for healthy adults aged over 70 years: the Australian ASPREE cohort study
- Hematology and Coagulation
- The EuroFlow PIDOT external quality assurance scheme: enhancing laboratory performance evaluation in immunophenotyping of rare lymphoid immunodeficiencies
- Clinical value of smear review of flagged samples analyzed with the Sysmex XN hematology analyzer
- Cardiovascular Diseases
- Evidence for stability of cardiac troponin T concentrations measured with a high sensitivity TnT test in serum and lithium heparin plasma after six-year storage at −80 °C and multiple freeze-thaw cycles
- Letters to the Editor
- Impact of high-sensitivity cardiac troponin I assay imprecision on the safety of a single-sample rule-out approach for myocardial infarction
- Why is single sample rule out of non-ST elevation myocardial infarction using high-sensitivity cardiac troponin T safe when analytical imprecision is so high? A joint statistical and clinical demonstration
- Iron deficiency and iron deficiency anemia in transgender populations: what’s different?
- The information about the metrological traceability pedigree of the in vitro diagnostic calibrators should be improved: the case of plasma ethanol
- Time to refresh and integrate the JCTLM database entries for total bilirubin: the way forward
- Navigation between EQA and sustainability
- C-terminal alpha-1-antitrypsin peptides as novel predictor of hospital mortality in critically ill COVID-19 patients
- Neutralizing antibodies against KP.2 and KP.3: why the current vaccine needs an update
- A simple gatekeeping intervention improves the appropriateness of blood urea nitrogen testing
- Congress Abstracts
- 16ª Reunião Científica da Sociedade Portuguesa de Medicina Laboratorial - SPML

