Abstract
Objectives
Contaminated apparatus and surgical tools pose serious health risks. For such purpose, disinfection chambers are employed. However, these systems rely on mercury-based UV lamps which comes with various drawbacks. These limitations have driven interest in Ultraviolet-C Light Emitting Diode (UV-C LED) technology as a safer and more efficient alternative. However, existing studies have not thoroughly explored the impact of varying intensities of pulse width modulation (PWM) on disinfection efficacy.
Methods
To addess this, the present study designed and tested a LED-based disinfection chamber by employing 4-W 275 nm Surface Mount Device (SMD) LEDs against frequently isolated bacteria. By following prior approach, irradiation time was alternated at 30-s intervals and antibacterial efficacy was assessed through various parameters. Additionally, scanning electron microscopy (SEM) was performed to examine the morphological changes.
Results
Results indicated that the reduction was significantly influenced (p<0.05) with varying PWM levels (60–100 %), achieving 2.05-log10 and 1.54-log10 inactivation against Escherichia coli and Staphylococcus aureus, respectively, upon exposure to 51.24 mJ/cm2 under maximum exposure settings. Moreover, complete cellular damage leading to bleb protrusion and cell-leakage confirmed the disruption of bacterial DNA.
Conclusions
In conclusion, UV-LEDs show great potential for disinfection, with efficiency influenced by PWM and dosage.
Introduction
The contamination of enclosed environments, such as public transportation and hospitals, as well as high-contact surfaces like elevator buttons, door handles, and food preparation areas, poses a significant global public health concern due to its potential to facilitate the spread of pathogens [1]. Moreover, Healthcare-associated infections (HAIs) significantly contribute to patient morbidity and mortality while also driving up healthcare costs [2]. While 3.2 % of Americans experience HAIs, the rate is 6.5 % in the European Union/European Economic Area, with even higher frequencies likely in other parts of the world [3]. Environmental contamination plays a critical role in the transmission of these pathogens [4]. Studies have also highlighted the persistence of various nosocomial pathogens in healthcare settings, including Staphylococcus aureus (S. aureus), vancomycin-resistant enterococci (VRE), methicillin-resistant S. aureus (MRSA), Acinetobacter baumannii (A. baumannii), Clostridioides difficile (C. difficile), and Pseudomonas aeruginosa (P. aeruginosa).
Bacterial contamination can also occur through direct transmission from infected or colonized patients or via the hands of healthcare workers (HCWs) [2], [5]. Objects in close proximity to patients are at a higher risk of contamination, and infections often result in increased levels and rates of bacterial presence [6]. One of the studies have identified bed rails, bed surfaces, supply carts, over-bed tables, and intravenous pumps as some of the most frequently touched surfaces by HCWs [7]. Additionally, medical equipment and devices such as hemodialysis machines, infusion pumps, stethoscopes, electronic thermometers, and blood pressure cuffs can serve as potential reservoirs for the transmission of nosocomial infections. More commonly, S. aureus can spread through the air from contaminated mattresses and clothing, settling on various surfaces. Additionally, person-to-person transmission of VRE has been linked to exposure through HCW hands, contaminated surfaces, and equipment like thermometers and electrocardiogram machines [8]. Further research has identified various surfaces prone to infection that contribute to the spread of pathogens, particularly those near patients such as bedrails, bedside tables, taps, and knobs in hospital wards. Additionally, healthcare workers’ hands, cellphones, computers, doorknobs, medical equipment such as pulse oximeter finger probes and electronic rectal thermometers, prescription carts, beds, mop pads, portable beds, and sinks contribute to the transmission of various pathogens. Another study identified contamination sources in ophthalmic settings, including ophthalmic solutions, medications, slit lamps, condensing lenses, and ultrasound bio microscopy probes. Another study showed pathogenic or environmental microbes including VRE, MRSA, and P. aeruginosa may infect stethoscopes, writing pens, white coats, keyboards, case notes, faucets, mobile phones, medical charts, and wrist watches [9].
This continuous interaction emphasizes the vital necessity for infection prevention methods. For such purposes, chemical disinfectants are on the rise. This increasing demand for automated, chemical-free solutions has propelled UV radiation to the forefront as a practical alternative [2], [10]. Most UV-based disinfection chambers available in the market is based on LP mercury lamps [11], which emit radiation at a peak wavelength of 254 nm. However, the delicate design and the need for extended warm-up time in these technologies pose challenges in specific applications [12], and their environmentally harmful mercury content raises concerns, which has been banned due to hazardous component [13]. Hence, numerous industries, such as those involved in water and air disinfection, are transitioning from mercury lamps to UV-LEDs [2], [5], [14]. Therefore, for the disinfection of small-scale apparatus, surgical tools, and personal gadget, disinfection chambers have been introduced [15], [16]. However, these chambers employ excimer or mercury-based lamps for disinfection [16]. One recent study [17] evaluated the ZAPARAY™ UVC LED chamber as a time- and energy-efficient alternative for disinfecting medical instruments. Non-rinsed dental tools showed reductions of 3.23–6.25-log10, while rinsing alone reduced bacterial loads by 2.7 log10. UVC further reduced loads by 3.65-log10. However, the chamber employed 300-s of contact time to achieve adequate bacterial disinfection. Conversely, newly available 4 W 275 nm SMD devices having quad-lens technology has not been explored in regards of disinfection chamber, providing high throughput compared to low-power LEDs [18]. Moreover, existing studies have not thoroughly explored the effects of pulse width modulation (PWM) on disinfection efficacy.
Previous similar research on UV-based disinfection has contributed valuable insights but also exhibits key limitations. For instance, study by Siwe, H., et al. [17] focused only on Gram-positive bacteria which was S. aureus by utilizing commercial UV chambers to asess the antibacterial efficacy. Certain investigations explored large-scale wastewater treatment with 280 nm UV-LED reactors [19], which, while effective for bulk water disinfection, are impractical for small-scale medical tool sterilization. Another study [20] examined UV decontamination of N95 respirators but relied on broad-spectrum UV sources, and emphasized material properties over precise microbial inactivation control. Additionally, while a detailed review highlighted UV-C LED applications across industries, it primarily discussed large-scale reactors and cost barriers without delving into PWM-controlled intensity optimization for the disinfection for surgical tools, apparatus and hand-held healthcare equipment.
Hence, this study introduced PWM-controlled UV intensity modulation to achieve dose-dependent bacterial inactivation which is a critical feature for precision disinfection that prior studies overlooked. For such case, the study initially involved the designing and development of a 3D printed chamber incorporating motor driver to run 8 LEDs simultaneously in a pursuit to achieve optimal disinfection over shorter duration of treatment time contrast to previously developed systems. Moreover, the three different PWM levels (60 , 80 and 100 %) were employed to observe the influence on inactivation against Gram-positive and Gram-negative bacteria, particularly S. aureus and Escherichia coli (E. coli), respectively, being the most common surface bacterium causing nosocomial infection. Further validation of complete damage is conducted through SEM analysis where disruption of treated samples was observed.
Materials and methods
Design and development of circuitry
To ensure stable and efficient operation of the UV-C LEDs, a carefully designed circuit configuration was implemented (Figure 1). The L298P motor driver was utilized to regulate the current supplied to the LEDs, maintaining consistent power delivery and avoiding energy losses. Eight UV-C LEDs were divided into two groups, with each group connected to a separate output channel of the motor driver. The positive and negative terminals for the first group of LEDs were connected to M1+ and M1-, while those for the second group were connected to M2+ and M2-. The LEDs were operated at three distinct efficiency levels, 60 , 80, and 100 %, as recommended by the manufacturer. These levels were achieved by varying the current supplied through the motor driver. A 5 V cooling system was integrated into the circuit to prevent overheating during operation. The cooling fans were directly powered by a 5 V power supply, ensuring that the LEDs and other components remained within safe temperature limits. Additionally, a 12C-compatible LCD was connected via the 12C interface and powered using the same 5 V supply. This LCD provided a convenient way to monitor and control the operation of the LEDs, displaying real-time information about their efficiency levels and other operational parameters. To facilitate manual control and configuration, four push buttons were included in the circuit. Each button was paired with a 10 KΩ pull-down resistor to ensure stable low-state readings when the buttons were not pressed. This arrangement prevented floating values and eliminated the risk of false triggers, thus enhancing the reliability of the system. A stable 5 V power supply was used to power all components, ensuring consistent performance throughout the circuit. The motor driver played a critical role in varying the current supplied to the LEDs, allowing for uniform operation across all LEDs and achieving the desired efficiency levels of 60 , 80, and 100 %. This uniformity was essential to prevent imbalances in illumination or power consumption. To avoid unnecessary exposure causing damage to the skin, the chamber was befitted with the sensor on the door which stops the treatment as soon as the door was opened. Furthermore, the cooling fans worked effectively to dissipate heat, protecting the components and prolonging the lifespan of the system. Overall, the circuit design provided an optimized and robust solution for operating the UV-C LEDs at varying efficiency levels while maintaining system stability and component longevity.
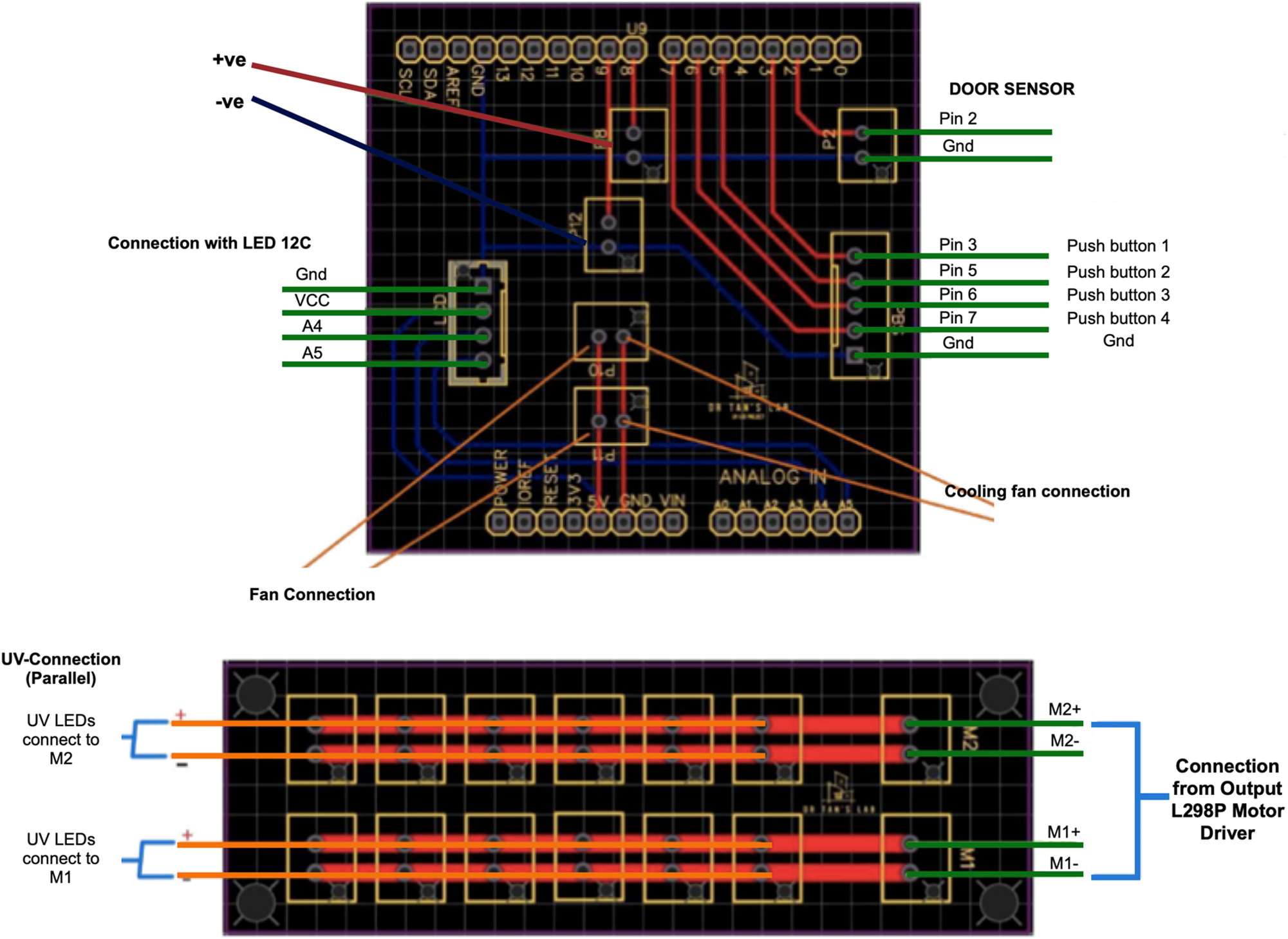
Circuitry of the chamber.
Design and development of chamber
The chamber measuring at (20 × 28.1 × 28.1 cm), was designed using CAD software and fabricated using a 3D printer (Ender3 Pro, Shenzhen, China). The chamber was designed to accommodate four cooling fans strategically positioned (two on the sides and two at the back) (Figure 2F–C). The airflow was directed outward on the opposite side to prevent internal circulation, which could lead to unnecessary external contaminants entering the chamber. To further mitigate the risk of external contamination, the entire chamber was enclosed within an isolated covering. At the center of the chamber, a tray was placed on a dedicated tray holder (Figure 2A) for optimal positioning. The LED panel was mounted on the front side of the chamber (Figure 2B), enabling various operational modes to be implemented.
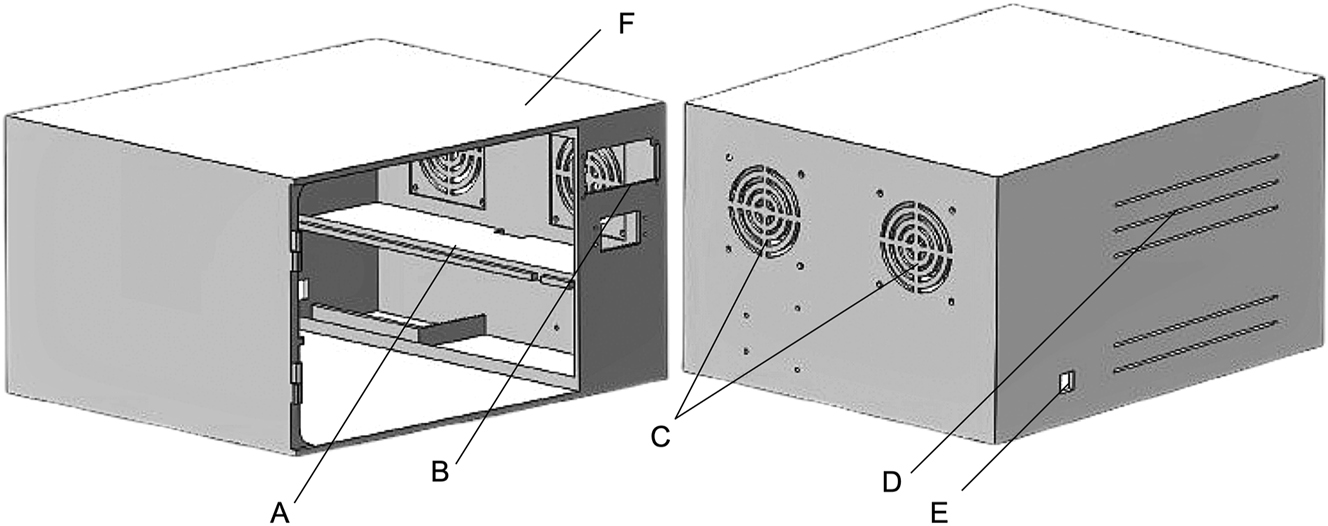
Design consideration of a chamber: (A) Tray holder, (B) front panel section of a chamber, (C, D and F) back and side cooling fans, (E) wiring outlet.
Fluence measurement
For the irradiance measurements, an irradiance meter (LS123, Portable UV Power Meter, China) was employed to examine the radiant output in µW-cm−2. The sensor was placed directly at the center facing upward. At the preliminary stage while assessing different variants of LEDs, irradiance was measured at 10 and 15 cm followed by the increment of PWM from 20 % each time until it reached 100 %. Later, the chosen setup was thoroughly examined three times to validate the performance and radiant output of combined setup at 15 cm. Later, the product of radiant output and time in (s) was computed. This rigorous method was put in place to guarantee accuracy and uniformity when assessing irradiance levels throughout the experimental processes. At each distance, the irradiance (E, µW-cm−2) remained constant.
Where,
H is the dosage in µJ-cm−2,
E is the irradiance in µW-cm−2,
t is the total time of treatment in seconds (s).
Selection and synergistic arrangement of LEDs
The 275 nm wavelength was chosen for this investigation based on extensive evidence demonstrating its superior disinfection capability within the 275–280 nm range [21], which has been widely reported for its potent antimicrobial effects. Therefore, this study employed eight different 275 nm LED variants with varying power ratings, assessing their germicidal efficiencies at distances of 10 and 15 cm. Since LED repositioning with different orientations can significantly affect intensity through strategic and synergistic arrangements [22], this study tested various configurations and identified clustered positioning as optimal for achieving maximum irradiation across 10–15 cm distances. Furthermore, the initial phase of the study involved assessing how irradiance levels varied across different 275 nm UV-C LEDs with varying power ratings. After identifying the highest-performing LED, this unit along with seven comparable variants were selected for further evaluation. The outcome revealed distinct performance trends as the PWM (pulse width modulation) increased from 10 to 100 %. LED 1 (Figure 3a) showed exceptional performance, achieving the highest irradiance at 15 cm with a value of 244 μW/cm2 at 100 % PWM. This underscored its strong efficiency over longer distances. At 10 cm, LED 1 also performed significantly better than other variants, reaching 332 μW/cm2, making it one of the most consistent LEDs across the two measured distances. Its steady increase in irradiance with PWM increments reflects its ability to deliver reliable and robust light output. LED 2 (Figure 3b) exhibited outstanding results, particularly at 10 cm, where it achieved the highest irradiance among all LEDs, with a peak of 381 μW/cm2 at 100 % PWM. At 15 cm, it maintained strong performance with 203 μW/cm2, ranking it as one of the top performers at this distance as well. LED 3 followed a similar trend, with high irradiance values at both distances. It reached 404 μW/cm2 at 10 cm and 211 μW/cm2 at 15 cm, demonstrating its capacity for delivering strong light output under maximum PWM conditions.
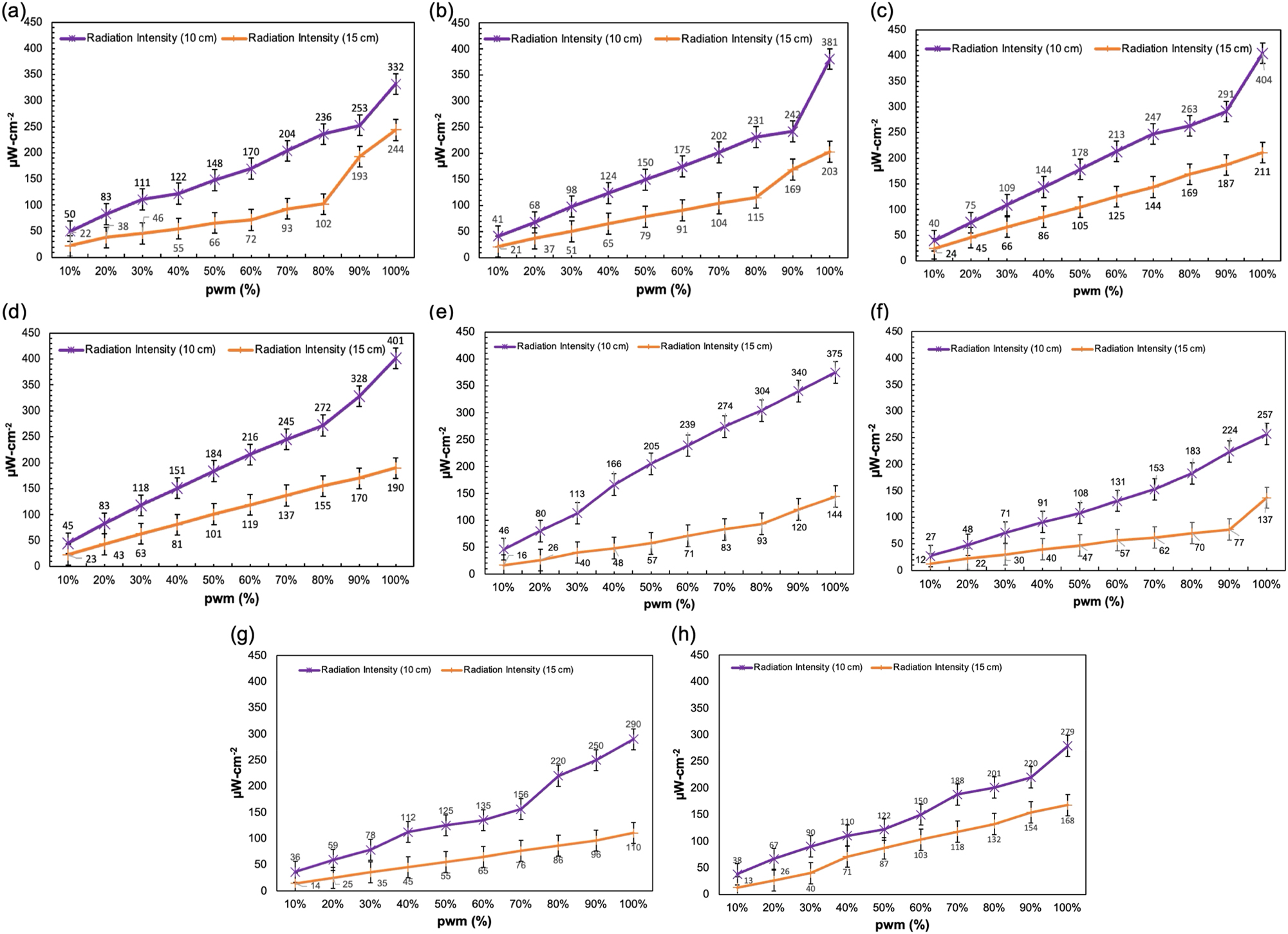
Irradiance assessment (in µW/cm2) for eight LEDs (a–h) evaluated at distances of 10 cm (purple) and 15 cm (orange), with PWM increments from 10 to 100 %.
Other LEDs, such as LED 4 (Figure 3d) and LED 5 (Figure 3e), showed competitive performance. LED 4 delivered 401 μW/cm2 at 10 cm and 190 μW/cm2 at 15 cm, while LED 5 reached 375 μW/cm2 at 10 cm and 144 μW/cm2 at 15 cm. These results highlight their ability to provide substantial irradiance, though their efficiency decreased more significantly at longer distances. LED 6 (Figure 3f), LED 7 (Figure 3g), and LED 8 exhibited lower overall irradiance values, with LED 8 (Figure 3h) showing the lowest peak irradiance at both 10 and 15 cm, with 279 μW/cm2 and 168 μW/cm2, respectively. The results demonstrate that as PWM increases, all LEDs exhibit proportional growth in irradiance. However, the rate of increase and the maximum achievable irradiance varied significantly between LEDs. LEDs such as LED 1, LED 2, and LED 3 emerged as superior options, delivering high irradiance values consistently at both measured distances. These findings underscore the importance of selecting specific LEDs based on application needs, particularly for scenarios requiring consistent light output over varying distances.
Among the eight LEDs, the 4 W 6565 SMD beaded LED, equipped with a quad-lens, produced the highest intensity compared to the others (Figure 3). To determine the optimal output for effective disinfection, different LED arrangements were tested. In finalized setup, four LEDs were positioned at each corner, tilted at a 60-degree angle, while the remaining four were placed in the center, all directed toward the central plate where the specimen was positioned. The electrical properties of such LEDs comprised a forward voltage (VF) of 6.8 V (V), radiant flux of 120 mW, an average forward current (IF) of 300 mA, a peak wavelength of 275 nm, and a viewing angle of thirty-degree. Furthermore, emission spectroscopy study was also performed using a spectrometer (HR4000-Vis-NIR, Ocean Optics, Inc, USA) to precisely identify the actual wavelength of these sources. Furthermore, during the treatment, all LEDs were powered through a motor driver, with varying current sources operating at 60 , 80, and 100 % efficiency levels.
In-vitro tests and validation
Bacterial culture
The criteria involved in choosing bacteria for the assessment were biosafety, prevalence and pathogenic nature. Therefore, for this study, Gram-positive and Gram-negative S. aureus having ATCC 15442 and E. coli ATCC 11229 were targetted to examine the system’s efficiency. Moreover, in the context of disinfection, these bacteria are known to cause Healthcare Associated Infections (HAIs) and are frequently found associated with Nosocomial Infections (NIs). Additionally, to develop a consistent and controlled settings for the study, these bacteria with dual nature offered more precise evaluation of the efficacy of the system.
For culturing of bacteria, level-2 biosafety cabinet (1300s A2, Thermofisher Scientific, USA) was employed to maintain the sterile environment. Next, with the help of wire-loop, streaking was performed on fresh agar followed with incubation using labortatory graded incubator (PSI-50D, Tech Lab Protech, Malaysia) at 37 °C for 18 h to facilitate the growth of bacteria. These standarized suspension was then carefully swabbed over fresh agar for further experiments. The petri dishes containing bacterial suspension was then annoted and marked with different experimental specifications and later sealed to avoid any unnecessary cross-contamination. Following this, the samples were subjected to UV radiation based on the parameters mentioned earlier.
Multi-faceted assessment of bacterial disinfection
Determination of viable bacteria cell count
The technique was employed to evaluate the effect of UV-C LEDs on the remaining viable colony count of the treated bacteria. This approach allowed us to measure the surviving bacterial colonies after the treatment. For the bacterial cell count, frozen bacterial samples were kept under sterilized environment to avoid cross-contamination and later incubated for 1 min. Each sample was collected in a sterilized micro-test tube and transported to biosafety cabinet for further experimentation. For the sampling, 0.1 mL was directly plated onto the fresh medium containing nutrient agar using pour plate technique [41] and later marked with various exposure specifications before getting incubated at 37 °C for 18 h. Following incubation, the seals were removed and grown colonies were manually counted. The CFU/mL was later computed using equation (1). Each step was duplicated to ensure consistency and reliability in the results. Later, mean std was calculated for quantification of log reduction.
Determination of log reduction and inactivation efficiencies
For the quantification of disinfection, the factor by which the bacterial burden was reduced was calculated by computing transformation of logarithm of viable colonies before treatment and logarithm of the number of viable colonies after the treatment as shown in equation (2);
Where,
A is the number of viable microorganisms before treatment,
B is the number of viable microorganisms after treatment.
Furthermore, the inactivation efficiencies achieved from the treatment were investigated throughout the study using equation (3)
Where,
CFUuv-on is the value of CFU/mL of irradiated samples,
CFUuv-off is the value of CFU/mL of non-irradiated samples.
Irradiance distribution
Generally, to evaluate the irradiance distribution on the surface, intricate equipment such as beam profiler is required. However, due to such limitation, the study employed manual approach as suggeested by Le et al. [23] to simulate the pattern of intensity emitting irradiance on the surface where it normally diminishes as the sensor draws away from the source in horizontal direction. This approach provided insight into the coverage of irradiance over the surface and the targeted regions where the influence of inactivation corresponds to the degree of inactivation which could vary with the horizontal movement of the sensor from the center. The data was simulated with each PWM levels of (60 , 80 and 100 %).
The procedure initiated with measuring irradiance at the center point by placing the sensor right beneath the source at 15 cm, followed by a systematic evaluation of readings at 3 cm intervals each time in the horizontal axis. This method was repeated and followed systematically until the recorded readings approached insignificant levels or the irradiance meter reported no value detection.
Scanning electron microscopy (SEM) analysis
Examining bacterial structure was crucial to evaluating the impact of UV radiation on both cellular integrity and DNA disruption. To achieve this, all irradiated and control samples were analyzed to assess the effects of UV exposure on bacterial morphology before and after treatment. The study primarily focused on samples that exhibited maximum inactivation over a specific period of UV irradiation. The preparation process began with the extraction of treated bacteria, which were transferred onto a glass slide using a secondary glass slide followed by the series of processing stages, including fixation, dehydration, and air-drying. For fixation, 4 % glutaraldehyde was applied to the irradiated bacteria and left for 30 min. Following this, the bacteria underwent dehydration using a graded ethanol series (50 , 70, 90, and 100 %), with each step lasting 10 min. Once dehydrated, the cover glass slides were air-dried at room temperature before being sputter-coated. Finally, the prepared samples were examined under a scanning electron microscope (SEM) (Tabletop TM3000, Hitachi, Japan) at 7,000× magnification with an accelerating voltage of 15 kV.
Results & discussion
Dose dependency on bacterial suppression
To verify the anti-bacterial effectiveness of the UV irradiation, S. aureus and E. coli were treated with different PWM intensities (60 , 80, 100 %) for different treatment times (120 s, 150 s, 180 s, and 210 s) (See Figure 4). The minimum distance of 15 cm was chosen which was in accordance with the studies employing UV LED arrays and lamps [24], [25], alternating with the increment of 15 cm. Moreover, based on the recent work [26], where efficient disinfection was reported using time duration alternating with the increment of 30 s. Results show a linear correlation between increasing UV dose and bacterial suppression, with increasing PWM settings and increasing exposure over time producing greater suppression.

Suppression of S. aureus and E. coli against varied UV irradiation upon employment of different irradiation efficiencies.
For S. aureus, at 100 % PWM level, a gradual increase in suppression was noted with UV doses of 29.28 mJ/cm2, 40.38 mJ/cm2, and 51.24 mJ/cm2. This indicated the more intense effect of UV irradiation across the entire surface area as the time increased up to 210-s, however, we did not observe cell death at the edges for the 120-s exposure. Since Gram-negative bacteria are generally more vulnerable to UV irradiation than Gram-positive bacteria [27] due thick peptidoglycan wall of Gram-positive bacteria (∼80 nm) containing amino acids that can absorb UV-C light through their peptide bonds [28] compared to Gram-negative bacteria, having a much thinner peptidoglycan layer (∼8 nm) with a lower amino acid density, making them less resistant to UV-C light. The findings validated this claim as more pronounced inactivation was reported for E. coli with similar dosage in this study. In maximum exposure conditions, both treatments achieved superior disinfection in which E. coli was more sensitive than S. aureus with the increase in exposure time. E. coli, particularly at higher exposure durations, appeared to be more susceptible to maximum UV exposure than S. aureus (p<0.001). Building on recent research, the study by [29] demonstrated that disinfection efficacy is influenced by both distance and exposure duration, where the effect was primarily attributed to focal distance, as moving the UV source farther from the target area proportionally increased the irradiation diameter in both positive and negative directions. Moreover, using a similar distance [30], used a Krypton disinfection setup to examine its antibacterial efficiency. Even after extending the treatment period to 40 min in order to obtain an effective degree of suppression, the effects were less noticeable compared to this study where more effective suppression was achieved in less time and higher distances.
However, in case of both the treatments, 80 % PWM led to less effective disinfection than that achieved with maximum settings, however, still exhibited a pattern with increasing exposure durations resulting in enhanced suppression. This result suggests that S. aureus exhibited weaker suppression compared to E. coli but showed increasing suppression over the same duration. Results showed that closely packed dense colonies persisted after treatment for a minimum duration of 120-s test, indicating that the dosage imparted was not enough to inactivate the bacteria completely and the surviving cells were able to re-colonize. However, with increment in duration formation of clumped colonies degenerated into smaller colonies but viable bacteria thence persisted till 210-s of exposure for both the treatments. The corresponding UV doses varied between 12.24 and 21.42 mJ/cm2, which was significantly lower than that at full intensity, explaining the decrease of the suppression effect.
Nonetheless, lower PWM setting (60 %) resulted in weaker suppression where S. aureus initially showed a weaker response compared to E. coli. However, with prolonged UV exposure up to 210 s, increased bacterial suppression was observed, suggesting that higher irradiation duration led to greater bacterial reduction. However, even the maximum duration of S. aureus treatment resulted in residual dense opaque bacterial growth in regions of UV irradiation that were insufficient to achieve the substantial suppression. This indicated that in addition to duration of treatment, efficiency in which the treatment is carried out was next in line on the scale of achieving enhanced anti-microbial efficacy.
PWM dependency on log reduction
The reduction values resulted for S. aureus and E. coli at different PWM settings (100 , 80 and 60 %) and exposure times (120 s, 150 s, 180 s and 210 s) showed promising disinfection when duration was extended to approximately 4 min (see Figure 5). The effective reduction was increased with higher PWM intensitives and longer exposure durations attributed to the effect of irradiance dose and PWM on bacterial reduction. At 100 % PWM intensity, reduction factor consistently increased from 0.83 log at 120 s to 1.54 log10 at 210-s, corresponding to the doses from 29.28 mJ/cm2 to 51.24 mJ/cm2. The results demonstrated a clear dose-response relationship: as the dose increased, bacterial reduction became more significant [31]. Compared to the study by [32] which achieved a 1-log10 reduction at an exposure dose of 113 mJ/cm2, the inactivation attained with lower doses were more pronounced. The log reduction values at 80 % PWM were lower but followed the identical rising trend, where the reduction factor was evaluated to be 0.28-log10 inactivation at 120 s to 1.11-log10 at 210-s, corresponding to the doses which ranged between 12.24 mJ/cm2 to 21.42 mJ/cm2 in line with the lower irradiance intensity due to the PWM ratio. At 60 % PWM, the reduction was negligible, resulting at 0.19-log10 inactivation at 120 s and 0.28-log10 inactivation at 210-s, with corresponding doses of 8.64 mJ/cm2 to 15.12 mJ/cm2, exhibiting hardly an effect with low irradiance Intensity and extended exposure times.
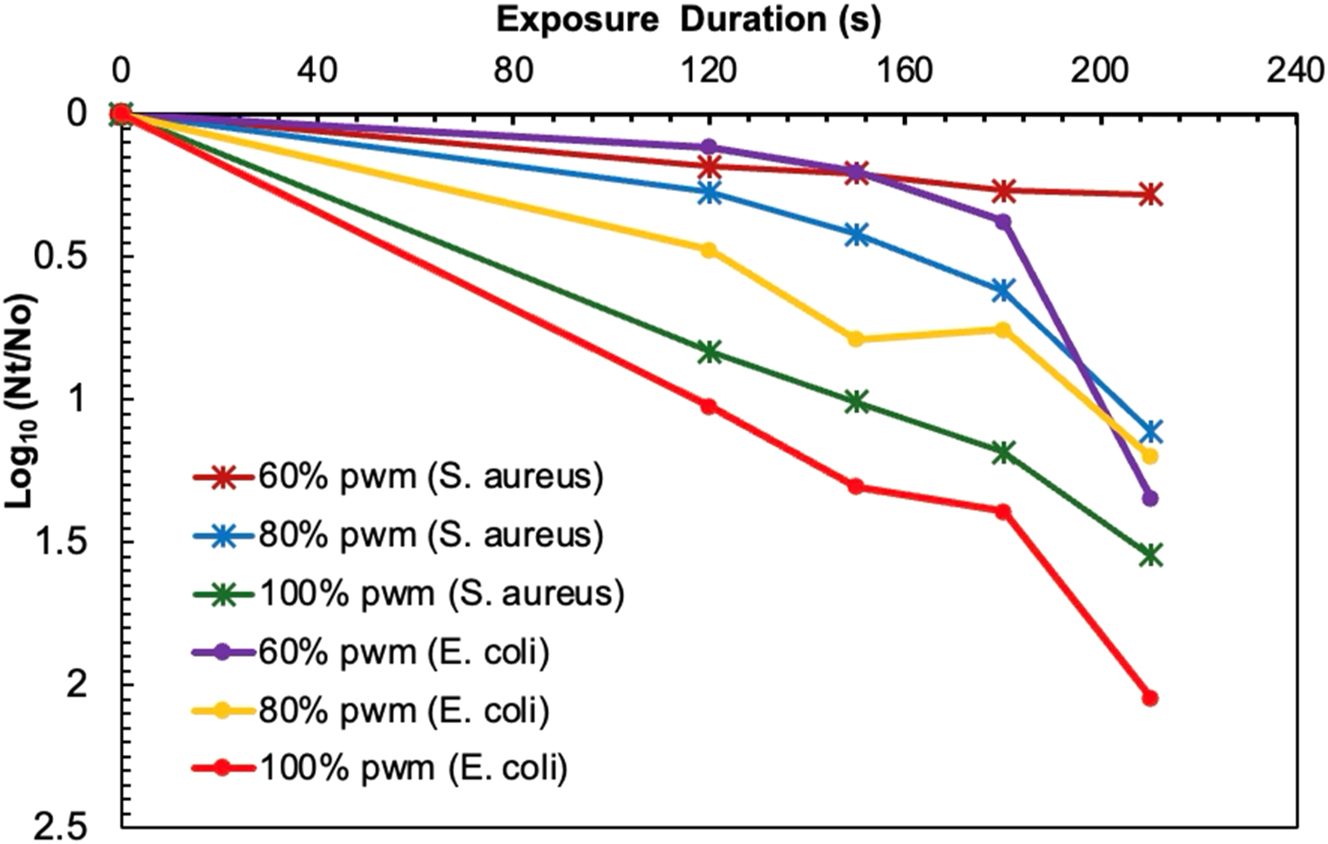
Log reduction of S. aureus and E. coli following UV irradiation at different pulse width modulation (PWM) settings (60 , 80, and 100 %) and exposure times (120 s, 150 s, 180 s, and 210 s).
Unlike S. aureus, the reduction values observed with E. coli was more pronounced. One study explores the effectiveness of UV-C irradiation as a microbial control strategy in indoor public spaces. At comparable PWM intensities and exposure durations, log reduction results indicated similar inactivation trends in E. coli as a function of dose and irradiance intensity. Accoridng to the findings, the reduction values at 100 % PWM varied from 1.02-log10 at 120-s to 2.05-log10 inactivation at 210-s, with corresponding doses ranging between 29.28 mJ/cm2 and 51.24 mJ/cm2, respectively, representing the maximum bacterial inactivation achieved in the various PWM levels. The present study aligns with previous research on E. coli O157:H7 inactivation using UV irradiation [33], where nearly higher dose was required to achieve the highest reduction 1.95-log10 inactivation and 1.83-log10 inactivation with Far UV-C. Hence, the present study showcased a linear dose-reduction relationship, suggesting that higher doses at peak PWM levels produce the enhanced inactivation of E. coli. With the decrease in PWM level, the bacterial reduction was comparatively lower at 80 %, valuing at 0.48-log10 to 1.2-log10 at 120-s and 210-s respectively, with the lower doses ranging from 12.24 mJ/cm2 to 21.42 mJ/cm2. At a PWM of 60 %, the reduction was even lesser for the first couple of exposure times, which later grew with the extension in treatment time. In that case, log reduction was found to be 0.12-log10 at 120-s which rose to 1.35-log10 inactivation at 210-s, while doses in the range of 8.64 mJ/cm2 to 15.12 mJ/cm2 were the indication that for lower irradiance intensity, longer exposure times were needed for an effective bacterial inactivation. These results wer more pronounced compared to the recent study which demonstrated the effectiveness of UVC irradiation against L. monocytogenes which required 21.6 mJ/cm2 achieved a 1.0–1.6-log10 reduction. Overall, the results indicated that E. coli was susceptible to the UV irradiance dose and exposure time, where with maximum exposure exposure and PWM level, highest reduction was achieved which was effective for the inactivation of microbes.
This heightened inactivation of E. coli could be due to its cellular structure and susceptibility to UV light. E. coli is Gram-negative and its cell wall is thinner and more susceptible to the UV damage than S. aureus’s thick peptidoglycan layer in its cell wall that provides for additional protection to its cells. As mentioned previously, the primary lesion of UV irradiation is the pyrimidine dimers in the DNA, while structural weaknesses of E. coli may be the cause of better absorption of the UV energy and more disruption of the cellular functions.
Overall, the results emphasized the importance of optimizing both dose and exposure time to achieve effective microbial inactivation. Higher PWM settings and longer exposure times consistently resulted in greater log reductions for both microorganisms. However, the difference in inactivation rates between E. coli and S. aureus highlighted the need to consider microbial species’ specific characteristics when designing disinfection protocols. The findings suggest that while E. coli is more easily inactivated, achieving comparable reduction levels for S. aureus requires higher doses or extended exposure durations.
Irradiance distribution and treatment coverage
As four UV LEDs were positioned at the center point facing downward, with one additional LED at each edge of the chamber tilted at cetain angle, the arrangement ensured optimal coverage and maximum average attainability of irradiance at the center where the sample is to be placed for effective disinfection. Figure 6 further illustrated the irradiance distribution across the entire disinfection chamber.

Irradiance distribution profile over petri dishes at a 15 cm distance for (a) S. aureus and (b) E. coli.
The resulting irradiance values showed a gradient from 244 μW/cm2 (PWM setting at 100 %) to 72 μW/cm2 (PWM setting at 60 %) for vertical distances from the centre of the beam over 15 cm (Figure 7). The reported range of irradiance achieved was higher than that from studies done by Nunayon et al. [34], achieving 0.31 μW/cm2. In contrary [35], showed similar efficiencies with treatment requiring 60 min of exposure. The horizontal measurements at the distance across comparable vertical distances showed a consistent trend, with the values decreasing from 244 μW/cm2 to 45 μW/cm2, 102 μW/cm2 to 8 μW/cm2 and 72 μW/cm2 to 7 μW/cm2, for 100 , 80 and 60 % PWM levels, respectively. Hence, at maximum PWM intensity, the optimal coverage for effective disinfection was reported to be 21 cm in diameter.

Varying level of PWMs influencing irradiance attainability and distribution across targeted surface.
SEM observations
To validate bacterial cell disruption, SEM analysis was conducted on both treated and untreated samples. In the control samples, S. aureus exhibited a cocci shape, while E. coli displayed a rod-shaped morphology [36], [37]. Upon exposure to the irradiation at a distance of 15 cm for 210 s, major changes in morphology were observed (Figure 8c and d) when compared to untreated samples (Figure 8a and b). Figure 8d depicted the SEM images of S. aureus cells when subjected to 210 s of irradiation. Interestingly, the treated cells revealed the formation of micro-colonies distributed across the surface. The irradiated E. coli cells revealed the incidence of lysis and bleb protrusions on their surfaces (Figure 8c) [37], [38], [39], [40], highlighting the impact of the UV irradiaiton in damaging cellular structure. This observation suggested that a 275 nm UV-C LEDs irradiation could induce diverse morphological changes in E. coli and S. aureus both, potentially influencing its growth and physical integrity. Furthermore, the adverse effect of UV radiation resulted in leakage of extracellular matrix resulting in bacterial lysis and appearance of medium-sized amorphous structures (Figure 8c and d) shown by orange, red and cyan colored arrow, widespread along the surface of the agar which was consistent with [38]. The microgram validated enough evidences in damaging the cellular integrity of S. aureus and E. coli, disrupting the DNA at greater extent, supporting the evidence of cellular damage.

SEM images depicting controlled and irradiated bacteria under the influence of a UV irradiation: (a) Control sample revealing unharmed S. aureus, (b) control sample revealing unharmed E. coli (c) irradiated samples revealing lysis and bleb protrusion of damaged S. aureus, (d) irradiated samples revealing bleb protrusion of damaged E. coli.
Conclusions
The present study concludes that the antibacterial activity of a disinfection chamber with SMD beaded UV-C LEDs was grately influenced by varying PWM levels in disinfecting Gram-positive and Gram-negative bacteria. The detailed results confirms that the chamber is able to effectively inactivate both kinds of bacteria, achieving maximum reduction of 2.1-log10 at a distance of 15 cm achieved against E. coli. Notably, E. coli exhibited greater susceptibility to UV irradiation in contrast to S. aureus. The study also determined that the optimal irradiance coverage extended to a diameter of 21 cm, ensuring uniform disinfection across the targeted surface. SEM analysis further substantiated bacterial cell disruption, revealing structural damage such as lysis, bleb formation, and compromised cell walls, leading to widespread micro-colony formation and amorphous structures on the agar surface. Ultimately, the quincunx arrangement of UV-C SMD beaded LEDs facilitated consistent and sustained irradiation, necessitating 210-s of exposure to achieve effective bacterial inactivation.
Funding source: Ministry of Higher Education, Malaysia
Award Identifier / Grant number: Q.J130000.3823.23H68
Acknowledgments
The research received funding from UTM Fundamental Research (UTMFR) under cost centre number Q.J130000.3823.23H68.
-
Research ethics: None declared.
-
Informed consent: None declared.
-
Use of Large Language Models, AI and Machine Learning Tools: None declared.
-
Conflict of interest: None declared.
-
Research funding: The research received funding from UTM Fundamental Research (UTMFR) under cost centre number Q.J130000.3823.23H68.
-
Data availability: None declared.
References
1. Raeiszadeh, M, Adeli, BA. A critical review on ultraviolet disinfection systems against COVID-19 outbreak: applicability, validation, and safety considerations. ACS Photon 2020;7:2941–51. https://doi.org/10.1021/acsphotonics.0c01245.Search in Google Scholar PubMed
2. Sheikh, J, Swee, TT, Saidin, S, Ahmed, S, Chua, LS, Thye, MF, et al.. Classic and alternative disinfection practices for preventing of hospital-acquired infections: a systemic review. Int J Environ Sci Technol 2024;21:8261–96. https://doi.org/10.1007/s13762-024-05635-3.Search in Google Scholar
3. Suetens, C, Latour, K, Karki, T, Ricchizzi, E, Kinross, P, Moro, ML, et al.. Healthcare-associated infections. Prevalence of healthcare-associated infections, estimated incidence and composite antimicrobial resistance index in acute care hospitals and long-term care facilities: results from two European point prevalence surveys 2016 to 2017. Euro Surveill 2018;23:1800516. https://doi.org/10.2807/1560-7917.es.2018.23.46.1800516.Search in Google Scholar PubMed PubMed Central
4. Sheikh, J, Swee, TT, Saidin, S, Ahmed, S, Serrero-Olmedo, J, Chua, LS, et al.. Comparative multivariate analysis for high-touch surface disinfection using optimized ultraviolet-C LEDs configuration. Hyg Environ Health Adv 2024;11:100101. https://doi.org/10.1016/j.heha.2024.100101.Search in Google Scholar
5. Tan, TS, Sheikh, J, Saidin, S, Ahmed, S, Chua, LS, Tiong, FT, et al.. Surface bacterium disinfection using everlight 6565 UV-C SMD. HumEnTec 2023;2:11–17. https://doi.org/10.11113/humentech.v2n1.33.Search in Google Scholar
6. Tanner, WD, Leecaster, MK, Zhang, Y, Stratford, KM, Mayer, J, Visnovsky, LD, et al.. Environmental contamination of contact precaution and non-contact precaution patient rooms in six acute care facilities. Clin Infect Dis 2021;72:S8–16. https://doi.org/10.1093/cid/ciaa1602.Search in Google Scholar PubMed PubMed Central
7. Shams, AM, Rose, LJ, Edwards, JR, Cali, S, Harris, AD, Jacob, JT, et al.. Assessment of the overall and multidrug-resistant organism bioburden on environmental surfaces in healthcare facilities. Infect Control Hosp Epidemiol 2016;37:1426–32. https://doi.org/10.1017/ice.2016.198.Search in Google Scholar PubMed PubMed Central
8. Douangboupha, V. Applied epidemiology of infectious diseases in Lao people’s democratic republic [PhD thesis]. Australia: The Australian National University; 2022.Search in Google Scholar
9. Cong, Y, Yang, S, Rao, X. Vancomycin resistant Staphylococcus aureus infections: a review of case updating and clinical features. J Adv Res 2020;21:169. https://doi.org/10.1016/j.jare.2019.10.005.Search in Google Scholar PubMed PubMed Central
10. Sheikh, J, Swee, TT, Saidin, S, Yahya, A, Ahmed, S, Chua, LS. Comparative efficacy assessment of solitary SMD beaded ultraviolet-C light emitting diodes for enhanced disinfection of high-touch surfaces. J Kejuruteraan 2024;36:1329–36.Search in Google Scholar
11. Xiao, D, Zhang, T, Wang, F. Inactivation of microorganisms on surfaces of a refrigerator compartment with low-pressure mercury lamps. Int J Refrig 2024;164:132–42. https://doi.org/10.1016/j.ijrefrig.2024.05.008.Search in Google Scholar
12. Sheikh, J, Saidin, S, Malik, SA, Tan, TS, Chua, LS, Ahmed, AA, et al.. Enhanced irradiance levels using synergistically engineered monochromatic wavelength ultraviolet-C arrays configuration. J Hum Cent Technol 2024;3:53–60. https://doi.org/10.11113/humentech.v3n1.73.Search in Google Scholar
13. Wu, X, Liang, S, Li, H, Yang, Z. Changing production and consumption patterns for win-win of minamata convention on mercury and common prosperity in China. J Clean Prod 2024;462:142713. https://doi.org/10.1016/j.jclepro.2024.142713.Search in Google Scholar
14. Alam, GT, María, PH, Paula, SM, Eva, JH, Ana, A, Casas López, JL, et al.. Enhancing disinfection and microcontaminant removal by coupling LED driven UVC and UVA/photo-Fenton processes in continuous flow reactors. Sci Total Environ 2024;918:170655. https://doi.org/10.1016/j.scitotenv.2024.170655.Search in Google Scholar PubMed
15. Azher, MA, Asmaa, Y, Ghassan, F, Hawra, T. Design and sizing of stand-alone photovoltaic (PV) system for powered mobile cleaning and disinfection chamber system. AIP Conf. Proc 2023;2776:050001.10.1063/5.0136519Search in Google Scholar
16. Wang, MH, Zhang, HH, Chan, CK, Lai, ACK. Experimental study of the disinfection performance of a 222-nm Far-UVC upper-room system on airborne microorganisms in a full-scale chamber. Build Environ 2023;236:110260. https://doi.org/10.1016/j.buildenv.2023.110260.Search in Google Scholar
17. Siwe, H, Aerssens, A, Flour, MV, Ternest, S, Van, SL, Verstraeten, D, et al.. Microbiological evaluation of ultraviolet C light-emitting diodes for disinfection of medical instruments. Heliyon 2024;10:e37281. https://doi.org/10.1016/j.heliyon.2024.e37281.Search in Google Scholar PubMed PubMed Central
18. Wilm, A. Advancements in UV-C LED technology: improving external quantum and system efficiency. Light-Emitting Dev, Mater Appl 2024;12906:56–66.10.1117/12.3002443Search in Google Scholar
19. Nicolau, T, Gomes Filho, N, Padrão, J, Zille, A. A comprehensive analysis of the UVC LEDs’ applications and decontamination capability. Materials 2022;15:2854. https://doi.org/10.3390/ma15082854.Search in Google Scholar PubMed PubMed Central
20. Ontiveros, CC, Shoults, DC, MacIsaac, S, Rauch, KD, Sweeney, CL, Stoddart, AK, et al.. Specificity of UV-C LED disinfection efficacy for three N95 respirators. Sci Rep 2021;11:15350. https://doi.org/10.1038/s41598-021-94810-4.Search in Google Scholar PubMed PubMed Central
21. Liang, JJ, Liao, CC, Chang, CS, Lee, CY, Chen, SY, Huang, SB, et al.. The effectiveness of far-ultraviolet (UVC) light prototype devices with different wavelengths on disinfecting SARS-CoV-2. Appl Sci 2021;11:10661. https://doi.org/10.3390/app112210661.Search in Google Scholar
22. Chien-Ping, W, Jun-You, L. Effect of UV-C LED arrangement on the sterilization of Escherichia coli in planar water disinfection reactors. J Water Proc Eng 2023;56:104399. https://doi.org/10.1016/j.jwpe.2023.104399.Search in Google Scholar
23. Le, TT, Liao, CT, Lin, SK, Wu, CS, Nguyen, QK, Yang, TH, et al.. Study of banana preservation extension by UVC radiation in precise monitoring LED irradiation cavity. Sci Rep 2022;12:21352. https://doi.org/10.1038/s41598-022-25716-y.Search in Google Scholar PubMed PubMed Central
24. Trivellin, N, Buffolo, M, Onelia, F, Pzzolato, A, Barbato, M, Orlandi, VT, et al.. Inactivating SARS-CoV-2 using 275 nm UV-C LEDs through a spherical irradiation box: design, characterization and validation. Materials 2021;14:2315. https://doi.org/10.3390/ma14092315.Search in Google Scholar PubMed PubMed Central
25. Jaiaue, P, Piluk, J, Sawattrakool, K, Thammakes, J, Malasuk, C, Sitanan, T, et al.. Mathematical modeling for evaluating inherent parameters affecting UVC decontamination of indicator bacteria. Appl Environ Microbiol 2022;88:e02148–21. https://doi.org/10.1128/aem.02148-21.Search in Google Scholar PubMed PubMed Central
26. Glaab, J, Lobo-Ploch, N, Cho, HK, Filler, T, Gundlach, H, Guttmann, M, et al.. Skin tolerant inactivation of multiresistant pathogens using far-UVC LEDs. Sci Rep 2021;11:14647. https://doi.org/10.1038/s41598-021-94070-2.Search in Google Scholar PubMed PubMed Central
27. Kang, JW, Kim, SS, Kang, DH. Inactivation dynamics of 222 nm krypton-chlorine excilamp irradiation on Gram-positive and Gram-negative foodborne pathogenic bacteria. Food Res Int 2018;109:325–33. https://doi.org/10.1016/j.foodres.2018.04.018.Search in Google Scholar PubMed
28. Narita, K, Asano, K, Naito, K, Ohashi, H, Sasaki, M, Morimoto, Y, et al.. 222-nm UVC inactivates a wide spectrum of microbial pathogens. J Hosp Infect 2020;105:459–67. https://doi.org/10.1016/j.jhin.2020.03.030.Search in Google Scholar PubMed
29. Messina, G, Fattorini, M, Nante, N, Rosadini, D, Serafini, A, Tani, M, et al.. Time effectiveness of ultraviolet C light (UVC) emitted by light emitting diodes (LEDs) in reducing stethoscope contamination. Int J Environ Res Publ Health 2016;13:940. https://doi.org/10.3390/ijerph13100940.Search in Google Scholar PubMed PubMed Central
30. Alnaser, W, Ghanem, A, Alnaser, N, Alaraibi, Q, Alshaikh, F. The Null effect of UVC ceiling light exposure on SARS-CoV2. Health 2020;12:1481–95. https://doi.org/10.4236/health.2020.1211108.Search in Google Scholar
31. Kim, HJ, Yoon, HW, Lee, MA, Kim, YH, Lee, CJ. Impact of UV-C irradiation on bacterial disinfection in a drinking water purification system. J Microbiol Biotechnol 2022;33:106. https://doi.org/10.4014/jmb.2211.11027.Search in Google Scholar PubMed PubMed Central
32. Taylor-Edmonds, L, Lichi, T, Rotstein-Mayer, A, Mamane, H. The impact of dose, irradiance and growth conditions on Aspergillus Niger (renamed A. brasiliensis) spores low-pressure (LP) UV inactivation. J Environ Sci Health – Part A Toxic/Hazard Subst Environ Eng 2015;50:341–7. https://doi.org/10.1080/10934529.2015.987519.Search in Google Scholar PubMed
33. Yin, F, Zhu, Y, Koutchma, T, Gong, J. Inactivation and potential reactivation of pathogenic Escherichia coli O157:H7 in apple juice following ultraviolet light exposure at three monochromatic wavelengths. Food Microbiol 2015;46:329–35. https://doi.org/10.1016/j.fm.2014.08.015.Search in Google Scholar PubMed
34. Nunayon, SS, Zhang, HH, Lai, ACK. Comparison of disinfection performance of UVC-LED and conventional upper-room UVGI systems. Indoor Air 2020;30:180–91. https://doi.org/10.1111/ina.12619.Search in Google Scholar PubMed
35. Bentancor, M, Vidal, S. Programmable and Low-Cost ultraviolet room disinfection device. Hardwarex 2017;4:00046. https://doi.org/10.1016/j.ohx.2018.e00046.Search in Google Scholar
36. Sheikh, J, Swee, TT, Saidin, S, Yahya, AB, Malik, SA, Yin, JSS, et al.. Bacterial disinfection and cell assessment post ultraviolet-C LED exposure for wound treatment. Med Biol Eng Comput 2021;59:1055–63. https://doi.org/10.1007/s11517-021-02360-8.Search in Google Scholar PubMed
37. Sheikh, J, Tan, TS, Saidin, S, Ahmed, S, Chua, LS. Bacterial morphology and microscopic advancements: navigating from basics to breakthroughs. Microbiol Immunol Commun 2024;3:03–41. https://doi.org/10.55627/mic.003.001.0567.Search in Google Scholar
38. Attallah, NGM, Elekhnawy, E, Negm, WA, Hussein, IA, Mokhtar, FA, Al-Fakhrany, OM. In vivo and in vitro antimicrobial activity of biogenic silver nanoparticles against Staphylococcus aureus clinical isolates. Pharmaceuticals 2022;15:194. https://doi.org/10.3390/ph15020194.Search in Google Scholar PubMed PubMed Central
39. Zekanović, MS, Begić, G, Mežnarić, S, Badovinac, J, Kristof, R, Linsak, DT, et al.. Effect of UV light and sodium hypochlorite on formation and destruction of Pseudomonas fluorescens biofilm in vitro. Processes 2022;10:1901. https://doi.org/10.3390/pr10101901.Search in Google Scholar
40. Li, J, Wang, G, Zhu, H, Zhang, M, Zheng, X, Di, Z, et al.. Antibacterial activity of large-area monolayer graphene film manipulated by charge transfer. Sci Rep 2018;4:4359. https://doi.org/10.1038/srep04359.Search in Google Scholar PubMed PubMed Central
41. Terrones-Fernandez, I, Casino, P, López, A, Peiró, S, Ríos, S, Nardi-Ricart, A, et al.. Improvement of the pour plate method by separate sterilization of agar and other medium components and reduction of the agar concentration. Microbiol Spectr 2023;11:03161–22. https://doi.org/10.1128/spectrum.03161-22.Search in Google Scholar PubMed PubMed Central
© 2025 the author(s), published by De Gruyter, Berlin/Boston
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Frontmatter
- Research Articles
- Dynamic loading leakage test of dental abutment/implant connections based upon a novel implant system with an abutment switch feature: an in vitro study
- A computational 3D analysis for assessing bone remodeling following total hip arthroplasty: a longitudinal study spanning six years
- The impact of physiological load and support conditions on axial implant system testing of locking plates for femoral shaft fractures – a biomechanical analysis
- Validation and comparison of three different methods for automated identification of distal femoral landmarks in 3D
- Chamber design and intensity-modulated ultraviolet-C LEDs for advanced pulsed photonic disinfection
- Correlation of electrogastrography and bioelectric impedance techniques for the gastric motility assessment
- Detection and analysis of fatigue flight features using the fusion of pilot motion behavior and EEG information
- How does research output and impact in medical informatics vary among EU member states? – A bibliometric analysis
Articles in the same Issue
- Frontmatter
- Research Articles
- Dynamic loading leakage test of dental abutment/implant connections based upon a novel implant system with an abutment switch feature: an in vitro study
- A computational 3D analysis for assessing bone remodeling following total hip arthroplasty: a longitudinal study spanning six years
- The impact of physiological load and support conditions on axial implant system testing of locking plates for femoral shaft fractures – a biomechanical analysis
- Validation and comparison of three different methods for automated identification of distal femoral landmarks in 3D
- Chamber design and intensity-modulated ultraviolet-C LEDs for advanced pulsed photonic disinfection
- Correlation of electrogastrography and bioelectric impedance techniques for the gastric motility assessment
- Detection and analysis of fatigue flight features using the fusion of pilot motion behavior and EEG information
- How does research output and impact in medical informatics vary among EU member states? – A bibliometric analysis

