Abstract
The effects of short-term treatment with phenylurea (DCMU, isoproturon) and phenol-type (ioxynil) herbicides on the green alga Chlorella kessleri and the cyanobacterium Synechocystis salina with different organizations of photosystem II (PSII) were investigated using pulse amplitude modulated (PAM) chlorophyll fluorescence and photosynthetic oxygen evolution measured by polarographic oxygen electrodes (Clark-type and Joliot-type). The photosynthetic oxygen evolution showed stronger inhibition than the PSII photochemistry. The effects of the studied herbicides on both algal and cyanobacterial cells decreased in the following order: DCMU>isoproturon>ioxynil. Furthermore, we observed that the number of blocked PSII centers increased significantly after DCMU treatment (204–250 times) and slightly after ioxynil treatment (19–35 times) in comparison with the control cells. This study suggests that the herbicides affect not only the acceptor side but also the donor side of PSII by modifications of the Mn cluster of the oxygen-evolving complex. We propose that one of the reasons for the different PSII inhibitions caused by herbicides is their influence, in different extents, on the kinetic parameters of the oxygen-evolving reactions (the initial S0−S1 state distribution, the number of blocked centers SB, the turnover time of Si states, misses and double hits). The relationship between the herbicide-induced inhibition and the changes in the kinetic parameters is discussed.
1 Introduction
The application of herbicides in agricultural practice leads to an increase of pollution levels in soil and water, causing environmental problems. Their high toxicity, even at low concentrations, is dangerous for all living organisms. A number of herbicides with an agricultural importance are reported to act on photosystem II (PSII). They can be differentiated into two groups according to their chemical specification and inhibitory patterns: urea/triazine and phenol-type herbicides [1]. PSII is a major multisubunit chlorophyll-protein complex embedded in the thylakoid membrane, which drives electron transfer from water to plastoquinone (PQ) to produce molecular oxygen and protons using energy derived from light [2], [3], [4]. The herbicides, affecting the functions of PSII, inhibit electron transfer from QA to QB due to the competition of herbicides with PQ binding to the exchangeable QB site in the PSII complex [5], [6]. An accumulation of reduced QA, as a result from blocking of the electron transfer from QA to QB, leads to an increase in the proportion of QB nonreducing centers and an influence of the PSII function. It is known that the phenylurea-type and phenol-type herbicides interact with different amino acid residues on the D1 protein of PSII [7].
Moreover, Ajlani et al. [8] have suggested that the phenol-type herbicide ioxynil inhibits both the acceptor and the donor side of PSII. In addition, studies with mutants presupposed that asparagine 266, valine 249, phenylalanine 255 and alanine 251 participate in the binding niche of ioxynil [8]. It has also been shown that the mutant of Synechocystis 6714, in which asparagine at position 266 of the D1 protein is replaced with tyrosine, is resistant to ioxynil but not to the phenylurea herbicide DCMU [8]. Later studies on the double mutant of Synechocystis 6714 with additional substitution of serine by alanine at position 264 revealed that this mutant is insensitive to DCMU [9]. Therefore, all these results have proposed different binding sites for different types of herbicides such as DCMU and ioxynil. It has also been demonstrated that the binding properties of the herbicides DCMU and atrazine to the Synechocystis membranes are comparable with those of higher plant thylakoid membranes, i.e. they act by binding to the D1 protein of PSII [10]. Another commonly used herbicide is isoproturon, which, similar to DCMU, inhibits PSII functions by binding to the QB site of D1 protein and leads to a decrease of the biomass in freshwater ecosystems [11]. Modeling and simulation of D1 protein resistant to isoproturon indicates that the resistance is due to an alteration in the secondary structure near the binding site, resulting in loss of hydrogen bonds, hydrophobic interaction and complete loss of hydrophobic sites [12]. In addition, treatment with herbicides under illumination may induce oxidative damage, caused by the formation of singlet oxygen in the reaction center, which can lead to functional and/or structural modification of the photosynthetic membranes [13], [14] as their effectiveness varies according to the environmental conditions [15].
It is well known that cyanobacteria and microalgae are very sensitive to PSII inhibitors [10], [16], [17], [18], [19], [20], [21]. In addition, the effect of abiotic factors strongly depends on the organization of the PSII complex [22]. Naber and van Rensen [11] have suggested that the differences in the inhibition of PSII functions by herbicides are related to a variation of the residence time at D1 protein. It has also been proposed that phenol-type herbicide ioxynil binding into the QB niche produces a change of the D1 protein conformation and perturbs the donor side of PSII [8]. However, the mechanisms of the PSII herbicide action on the photosynthetic apparatus are still not wholly understood [14]. The aim of the present study is to examine the effects of phenylurea (DCMU, isoproturon) and phenol-type (ioxynil) herbicides on the functional activity of the photosynthetic apparatus with different organization. For this purpose, we studied in more detail the kinetic parameters of the oxygen-evolving reactions and photochemistry of PSII in green alga Chlorella kessleri and cyanobacterium Synechocystis salina. The effect of herbicides on the PSII photochemistry and the kinetic parameters of the oxygen-evolving reaction in both studied organisms as well as the herbicide-induced changes in these parameters are shown.
2 Materials and methods
2.1 Organisms and growth conditions
The mesophilic S. salina and C. kessleri were obtained from the Culture Collection of Autotrophic Organisms at the Institute of Botany, Academy of Sciences of the Czech Republic. Cells were cultivated as described previously by Apostolova et al. [22]. The nutrient medium of Setlik modified by Georgiev et al. [23] was used for cultivation of C. kessleri. The twice-diluted medium of Pekárková et al. [24], with the addition of NaNO3 (1.26 g L−1), NaHCO3 (3.0 g L−1) and C10H14N2O8.2H2O (200 mg L−1), was used for cultivation of S. salina. The strains were cultivated aseptically at 30°C in 200 mL glass vessels, under continuous illumination of white light at an intensity of 80 μmol m−2 s−1 and aeration of 100 L m−3 h−1 of air enriched with 2% CO2. The growth of the culture was monitored by the measurement of the cell density by spectrophotometry. Cultures with an optical density of 0.7 at 760 nm in the exponential phase of growth were used in all measurements.
2.2. Treatment with herbicides
The dark-adapted cells of S. salina and C. kessleri were treated in dim light for 20 min with DCMU (3-(3, 4-dichlorophenyl)-1,1-dimethylurea), isoproturon (3-(4- isopropylphenyl)-1,1-dimethylurea) and ioxynil (4-hydroxy- 3,5-diiodobenzonitrile) using various concentrations (0–500 nM). The reagents are from Sigma-Aldrich. The viability of the cells was checked before each treatment. For characterization of viability, we used the PAM parameter: Fv/Fm, which characterizes the primary photochemistry of PSII. Cells with Fv/Fm values of approximately 0.60 for the cyanobacterium and approximately 0.70 for the green alga were used. Herbicide stock solutions were prepared in ethanol at a concentration of 1 mM. Thus, the final concentration of ethanol in the suspension was less than 1%, which would not have an effect on the herbicide binding affinity and would not influence the photosynthetic efficiency of the cells [25]. To compare the sensitivity of different parameters, we introduced the value RS50: relative sensitivity (RS), which represents the herbicide concentration causing a 50% decrease of the initial parameter value, i.e. control parameter value without herbicides. For each parameter, RS50 value was determined from the curve showing the relationship between the concentration of herbicide and the changes of the studied parameter (as presented in Figures 1, 2, and 4). A smaller RS50 value means higher sensitivity to the herbicide of the respective parameter.
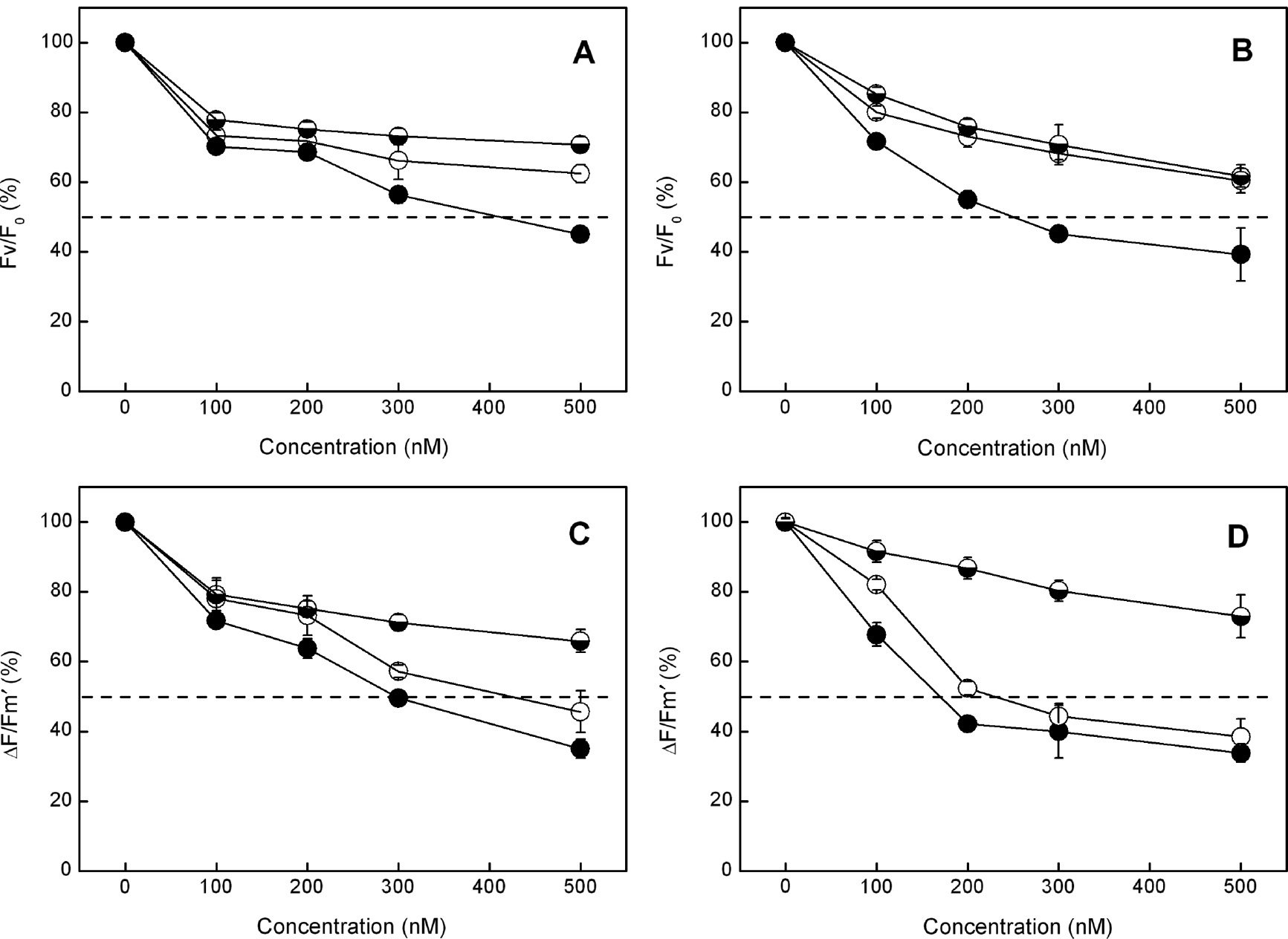
Effect of different concentrations of DCMU (●), isoproturon (○) and ioxynil ( ) on PAM chlorophyll fluorescence parameters of C. kessleri (A and C) and S. salina (B and D). Mean values±SE were calculated from five to seven independent experiments.
) on PAM chlorophyll fluorescence parameters of C. kessleri (A and C) and S. salina (B and D). Mean values±SE were calculated from five to seven independent experiments.
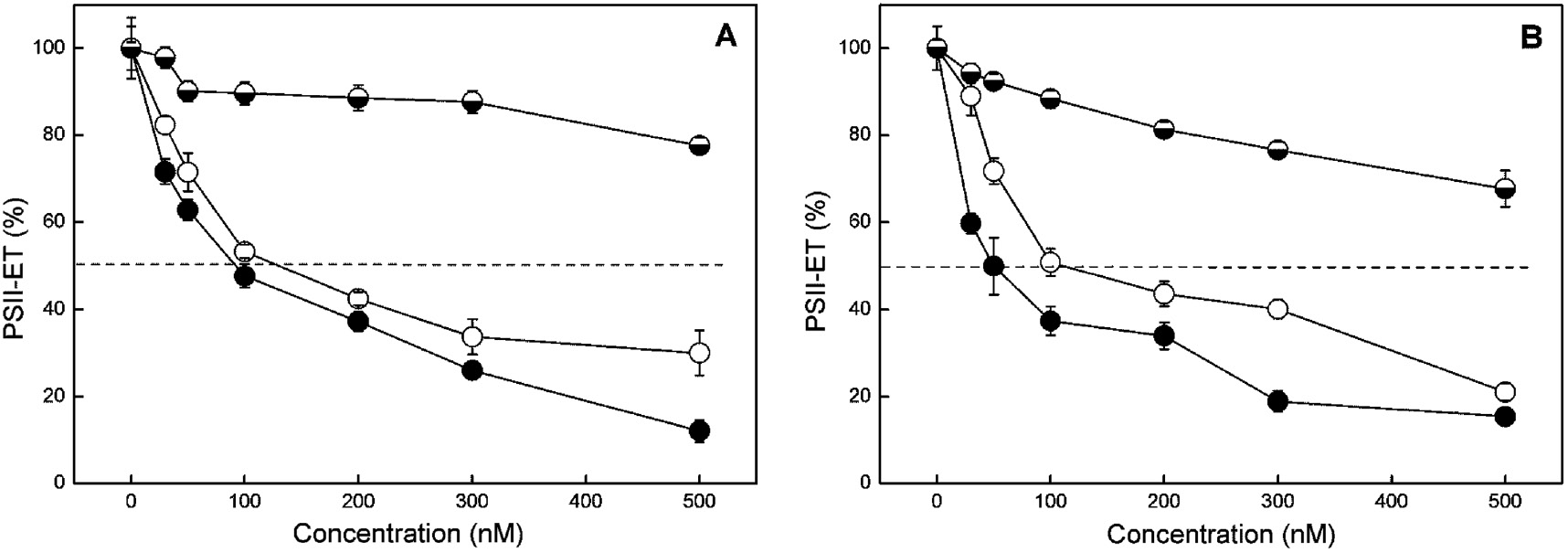
Effect of different concentrations of DCMU (●), isoproturon (○) and ioxynil ( ) on PSII-mediated electron transport (PSII-ET) of cells of C. kessleri (A) and S. salina (B). Mean values±SE were calculated from five to seven independent experiments. The values for untreated cells were 220±21 μmol O2 mg−1 Chl a h−1 and 420±26 μmol O2 mg−1 Chl a h−1 for C. kessleri and S. salina, respectively.
) on PSII-mediated electron transport (PSII-ET) of cells of C. kessleri (A) and S. salina (B). Mean values±SE were calculated from five to seven independent experiments. The values for untreated cells were 220±21 μmol O2 mg−1 Chl a h−1 and 420±26 μmol O2 mg−1 Chl a h−1 for C. kessleri and S. salina, respectively.
2.3 PAM chlorophyll fluorescence
PAM chlorophyll fluorescence was measured with a fluorometer (model PAM 101–103, Heinz Walz, Effeltrich, Germany) as described previously by Apostolova et al. [22]. The cells were dark-adapted for 20 min on the electrode. The minimum fluorescence yield (F0) was measured with very weak modulated light (0.02 μmol m−2 s−1). The maximum fluorescence yield in dark-adapted (Fm) and in light-adapted (Fm′) states were measured by illuminating the sample with a saturating flash of 2500 μmol m−2 s−1 (0.8 s) provided by a Schott lamp KL 1500 (Schott Glaswerke, Mainz, Germany). Actinic light illumination (150 μmol m−2 s−1) was provided by a second Schott lamp KL 1500 for the induction of photosynthesis. The steady state level Fs was estimated after 4 min of illumination with actinic light. The time interval between two consecutive flashes was 20 s. To assess the effect of the herbicides on the photosynthetic apparatus, the following parameters were calculated: the maximum ratio of quantum yields of photochemical to concurrent nonphotochemical processes in PSII, Fv/F0=(Fm−F0)/F0 [26] and the effective quantum yield of the photochemical energy conversion in PSII, ΔF/Fm′=(Fm′−Fs)/Fm′ [27].
2.4 Oxygen evolution measurements
The flash-induced oxygen yields and oxygen evolution under continuous illumination were measured using a custom-built polarographic oxygen rate electrode (Joliot-type) as described by Zeinalov [28]. The cell suspension of 100 μL volume forming a thin layer (2 mm) on the platinum electrode was preilluminated with 25 flashes and then dark-adapted for 10 min before measurements. Oxygen flash yields were induced by saturating (4 J) and short (t1/2=10 μs) periodic flash sequences. The spacing between the flashes was 0.70 s. The initial oxygen burst was recorded during illumination with continuous white light (400 μmol photons m−2 s−1).
According to Kok’s model [29], the oxygen-evolving complex (OEC) goes through five intermediate states of the Mn cluster (S0−S4) in the same PSII center for the production of one oxygen molecule and protons. The oxygen evolution occurs during S4→S0 transitions. The dark-adapted photosynthetic apparatus contains more stable S0 and S1 states. It is supposed that the damping of oscillations in flash-induced oxygen yields is due to the presence of misses (zero-step advances, α) and double-hits (double-step advances, β) [30]. The initial S0−S1 state distribution in the dark, misses (α) and double hits (β) were determined by the least square deviations fitting to the theoretically calculated yields with the experimentally obtained oxygen flash yields.
For the assessment of the effect of herbicides on the oxygen evolution kinetics (under continuous illumination and flashes), we used the following parameters: A – the amplitude of the oxygen burst under continuous illumination; Y – the maximum amplitude of the flash-induced oxygen yields; S0 – the populations of PSII centers in the initial S0 state (S0=100−S1); SB – the concentration of the blocked PSII centers; KD – the rate constant of excited Si states (i.e. the turnover time constant of the oxygen-evolving centers); α – the misses and β – the double hits (for details, see Rashkov et al. [18]). The parameters SB and KD are obtained using an extended kinetic version of Kok’s model [31], [32] on the base of the measurements by variation of the spacing between the flashes: 1.0, 0.70 and 0.55 s. The studied parameters are appropriate indicators for a modification of Mn cluster of OEC due to alterations in the PSII complex [18], [33], [34].
2.5 PSII-mediated electron transport
PSII-mediated electron transport was measured polarographically with a Clark-type electrode (Model DW1, Hansatech, Instruments Ltd., King’s Lynn, Norfolk, England) in a temperature-controlled cuvette at 22°C, using saturated white (achromatic) light. The PSII activity was measured by the rate of oxygen evolution in the presence of exogenous electron acceptor 1,4-benzoquinone (BQ, Sigma-Aldrich). The cells were measured in cultivation medium with 0.4 mM BQ.
2.6 Statistical analysis
The results are mean values from five to seven independent experiments. The statistical differences among the means were determined using a two-tailed paired Student’s t-test. Values of p<0.05 were considered as significant differences between the control and herbicide-treated samples.
3 Results
3.1 PAM chlorophyll fluorescence measurements
To evaluate the effect of the different PSII herbicides on the green alga C. kessleri and the cyanobacterium S. salina, we calculated the following PAM chlorophyll fluorescence ratios: Fv/F0 and ΔF/Fm′. It was observed that after treatment with herbicides, these PAM parameters decreased with an increase of the herbicide concentration for both Synechocystis and Chlorella (Figure 1). In addition, it can be seen that the decrease of both ratios ΔF/Fm′ and Fv/F0 was less pronounced in the green alga than in the cyanobacterium (especially at low concentrations). The observed changes in the studied PAM chlorophyll fluorescence parameters were stronger for DCMU in comparison with isoproturon and ioxynil (Figure 1).
The calculated values for the parameter RS50, showing the concentration that causes 50% decrease of the initial value before the herbicide treatment, are given in Table 1. This parameter is a useful tool for comparing the effect of herbicides on different systems. As can be seen, the RS50 values of DCMU for the Fv/F0 fluorescence ratio were significantly higher for Chlorella than for Synechocystis cells (Table 1). For the other two tested herbicides (isoproturon and ioxynil), a 50% reduction in this ratio was not detected after treatment with herbicide concentrations of up to 500 nM. The effective quantum yield of photochemical energy conversion in PSII (ΔF/Fm′) was inhibited in the following order: DCMU>isoproturon>ioxynil, as the cyanobacterial cells are more sensitive than algal cells (Table 1). RS50 values after treatment with up to 500 nM of ioxynil were not established, indicating the stronger effect of the phenylurea herbicides than the phenol-type herbicides.
The RS50 values for parameters of the photosynthetic oxygen evolution and the PAM chlorophyll fluorescence.
| Parameter | RS50 (nM) | |||||
|---|---|---|---|---|---|---|
| DCMU | Isoproturon | Ioxynil | ||||
| C. kessleri | S. salina | C. kessleri | S. salina | C. kessleri | S. salina | |
| A | 50.0±4.1 | 58.8±3.5 | 89.0±4.3 | 94.3±2.7 | 500.0±2.0 | 385.7±2.0 |
| Y | 27.5±2.3 | 28.1±2.5 | 41.8±4.6 | 37.6±3.0 | 498.6±2.8 | 367.0±3.4 |
| PSII-ET | 92.2±2.6 | 50.5±6.6 | 129.6±1.5 | 109.5±3.0 | >500 | >500 |
| Fv/F0 | 409.0±2.2 | 248.3±2.4 | >500 | >500 | >500 | >500 |
| ∆F/Fm′ | 295.8±2.2 | 168.8±2.7 | 422.6±3.9 | 228.2±2.8 | >500 | >500 |
Mean values±SE were calculated from five to seven independent experiments.
3.2 PSII-mediated electron transport
The influence of the different herbicides on oxygen evolution in the presence of the exogenous electron acceptor BQ (PSII-mediated electron transport, H2O→BQ) is shown in Figure 2. Experimental results showed that DCMU most strongly inhibits oxygen evolution followed by isoproturon in both cyanobacterium and green algae, whereas ioxynil has the smallest effect. After treatment with the phenol-type herbicide ioxynil, 50% inhibition of PSII-mediated electron transport was not registered at the studied concentration interval (0–500 nM) (Table 1). Data clearly showed that Synechocystis cells are more strongly inhibited compared with Chlorella cells concerning the phenylurea and phenol-type herbicides studied (Figure 2; Table 1).
3.3 Photosynthetic oxygen evolution
The oxygen evolution of Synechocystis and Chlorella cells measured with an oxygen rate electrode (Joliot type) after giving flash sequences or continuous illumination is shown in Figure 3. It is seen that the flash-induced oxygen yields and the oxygen burst amplitude under continuous illumination registered for algal cells are higher than those obtained from cyanobacterial cells (Figure 3). The maximum amplitude of the oxygen evolution observed after the third flash (Y) and the oxygen burst under continuous illumination (A) were used to assess the effect of herbicides on oxygen production. The parameter A correlates with the number of the functionally active PSII reaction centers, i.e. the fast and slow operating centers, whereas the parameter Y mainly characterizes the fast operating centers [33], [34].
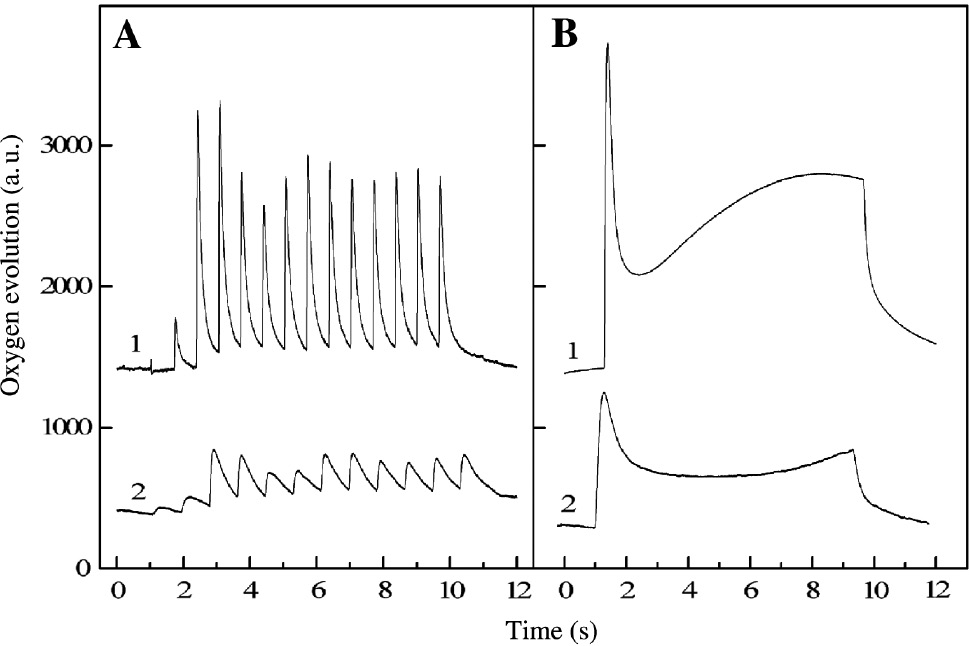
Flash-induced oxygen yields (A) and oxygen evolution under continuous illumination (B) of untreated cells of C. kessleri (1) and S. salina (2).
Treatment with the studied herbicides of algal and cyanobacterial cells led to an inhibition of the oxygen evolution and consequently influences the parameters A and Y, which gradually decrease with increasing herbicide concentration (Figure 4). Comparison of the RS50 values for the studied parameters A and Y of the cyanobacterium and the green alga showed that they have similar sensitivities to phenylurea herbicides, whereas toward ioxynil, the cyanobacterium is more sensitive than the green alga (Table 1). The data obtained also revealed that the sensitivity of S. salina and C. kessleri is much higher to phenylurea-type herbicides (DCMU and isoproturon) than to phenol-type herbicides (ioxynil). It can also be seen that the RS50 values decrease in the following order: ioxynil>isoproturon>DCMU (Table 1).
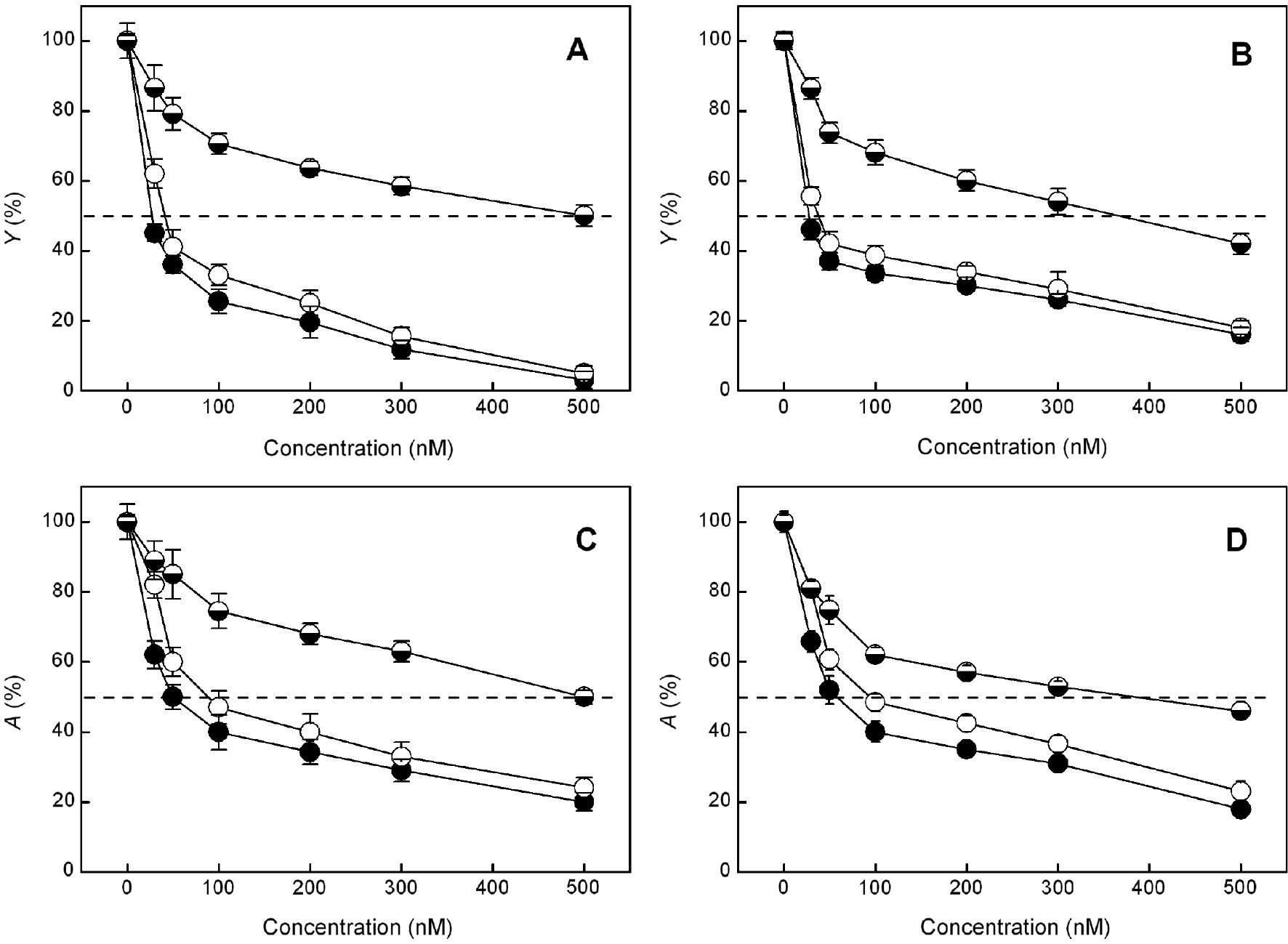
Effect of different concentrations of DCMU (●), isoproturon (○) and ioxynil ( ) on the maximum oxygen flash yields (Y) and the amplitude of oxygen burst under continuous illumination (A) of C. kessleri (A and C) and S. salina (B and D). Mean values±SE were calculated from five to seven independent experiments.
) on the maximum oxygen flash yields (Y) and the amplitude of oxygen burst under continuous illumination (A) of C. kessleri (A and C) and S. salina (B and D). Mean values±SE were calculated from five to seven independent experiments.
Calculated values (according to Kok’s model [29]) for the kinetic parameters of the initial dark distribution of the PSII centers in S0 and S1 states, misses and double hits before and after treatment with 100 nM herbicides of Chlorella and Synechocystis cells are given in Table 2. Data showed an increase of the PSII centers in the most reduced S0 state and of the misses (α) after the treatment with studied herbicides, which suggests an alteration in the OEC on the donor side. This observed increase was most pronounced for DCMU followed by isoproturon and ioxynil (Table 2). The herbicide treatment also led to an increase of double hits (β) in cyanobacterial cells, whereas in algal cells, this parameter was changed only after treatment with DCMU.
The parameters of flash oxygen yields for Chlorella and Synechocystis cells before and after treatment with 100 nM herbicides.
| Parameter | S0 (%) | SB (a.u.) | KD (s−1) | α (%) | β (%) |
| Chlorella kessleri | |||||
| Control | 50.5 | 0.0025 | 2.874 | 31.6 | 8.8 |
| DCMU | 78.4b | 0.625b | 0.895b | 41.5a | 11.0a |
| Isoproturon | 71.2a | 0.214b | 0.977b | 39.6a | 9.3 |
| Ioxynile | 64.6a | 0.047a | 1.55a | 39.2a | 8.5 |
| Synechocystis salina | |||||
| Control | 30.3 | 0.0044 | 1.52 | 25.5 | 8.6 |
| DCMU | 45.7b | 0.898b | 0.435b | 38.9a | 14.2a |
| Isoproturon | 42.1a | 0.545b | 0.659b | 36.4a | 13.7a |
| Ioxynile | 40.5a | 0.153a | 0.994a | 33.7a | 10.3a |
S0, the initial S0 state (S1=100−S0) in the darkness; SB, additional state corresponding to the concentration of the blocked PSII centers; KD, the rate constant of excited Si states; α, the misses; β, the double hits. Mean values were calculated from five to seven independent experiments. The statistical differences among the means determined using Student’s t-test between the control and herbicide treated cells. (ap<0.05, bp<0.01).
Recently, an additional state SB was included in the new scheme, according to the extended kinetic version of Kok’s model, corresponding to the concentration of the blocked PSII centers [18], [31], [32]. The calculated values for SB (blocked or inactive PSII centers) and the rate constant of excited Si states KD (KD is defined as the turnover time constant of the oxygen-evolving centers for the release of one oxygen molecule [32]) are also shown for the algal and cyanobacterial cells in Table 2. Data demonstrated that the rate constant of the turnover time KD decreases in the herbicide-treated cells in comparison with the control cells (Table 2). Therefore, the PSII herbicides increase the turnover time of the oxygen-evolving centers, i.e. delayed the rate of oxygen evolution. As can be seen, this turnover time was higher for control Synechocystis cells than that for control Chlorella cells.
The number of blocked PSII centers (SB) calculated for control Chlorella cells (Table 2) was significantly smaller (SB=0.0025 a.u.) than that for Synechocystis cells (SB=0.0044 a.u.). It can also be seen that SB is increased approximately 250 and 86 times after treatment of Chlorella cells with 100 nM DCMU and isoproturon, respectively, whereas this increase was approximately 19 times after treatment with ioxynil (Table 2). Similarly, SB for Synechocystis cells increased approximately 204 and 124 times after treatment with 100 nM DCMU and isoproturon, respectively, whereas after treatment with ioxynil this increase was approximately 35 times (Table 2).
4 Discussion
It is known that phenylurea and phenol-type herbicides damage photosynthesis by blocking electron transfer on the PSII acceptor side due to herbicide binding with the D1 protein of the PSII complex [11], [21]. Our data showed that the PSII functions (oxygen evolution and photochemistry) were influenced to different degrees by herbicides, depending on their concentration and chemical structure. PAM fluorescence measurements demonstrated that the studied herbicides have a strong effect on Fv/F0 and ΔF/Fm′ ratios (Figure 1; Table 1). Stronger influence on the photochemical energy conversion in PSII (ΔF/Fm′) in comparison with Fv/F0 after the herbicide treatment could be a result from the interruption of the electron transport between the quinone electron acceptors QA and QB [5]. Stronger influence of DCMU on the ΔF/Fm′ ratio under long-term treatment has also been reported for tropical sea grasses [35].
The decline in effective quantum yield of the photochemical energy conversion in PSII (ΔF/Fm′) after herbicide treatment was accompanied by an inhibition of the photosynthetic oxygen evolution as well as the parameters of oxygen evolution (Y, flash-induced oxygen yields and A, oxygen burst amplitudes) displayed higher sensitivity in comparison with the widely used parameters of the chlorophyll fluorescence (Table 1). This is in accordance with previous suggestions by other authors [36]. Furthermore, experimental results clearly showed that the flash-induced oxygen yields (Y) are more sensitive to phenylurea-type herbicides, as their RS50 values are between 1.8 and 2.5 times smaller than those for the oxygen burst amplitudes under continuous illumination (A) for both the cyanobacterium and the green alga (Table 1). This may suggest the effect of phenylurea herbicides predominantly on the fast-operated PSII centers, evolving oxygen by Kok’s mechanisms, as the effect is similar for Synechocystis and Chlorella cells. These data are in agreement with our earlier studies concerning the influence of DCMU and atrazine on Chlorella vulgaris and isolated thylakoid membranes from higher plants (see Rashkov et al. [18]).
Additionally, the present data shows an increase of the PSII centers in the most reduced S0 state after treatment with all the studied herbicides, which could be a result of a modification occurring in the OEC, i.e. alteration in the donor side of PSII as a consequence of the herbicide-binding on the acceptor side. These results supported the assumption of Ajlani et al. [8] that the herbicide ioxynil causes changes in the donor side of PSII induced by conformation of the D1 protein. In addition, this study revealed that the phenylurea herbicides also influence the donor side of PSII, increasing the number of PSII centers in the most reduced S0 state and the number of blocked centers SB (Table 2). Having this observation in mind, it could be proposed that the studied herbicides act as ADRY (acceleration of the deactivation reactions of the water-splitting enzyme system) reagents for the fast deactivation of the higher oxidized S-states in the dark [37], [38]. In addition to the well-known inhibitory effects of these herbicides on the electron acceptor side of PSII, experimental data also revealed ADRY effects on the PSII donor side. The influence on the number of PSII centers in the S0 state in dark-adapted samples (i.e. the S0–S1 states’ distribution) has also been shown after changes in the number and organization of the PSII-antenna complex in higher plants [33] and in mutant strains of Synechocystis sp. PCC 6803 [34], [39] as well as in the Cyt b559 mutants of Synechocystis [40].
Data also revealed that photosynthetic parameters are influenced by the studied QB binding herbicides in varying degrees (Table 1), which could be result of the different binding sites and orientations in the QB binding niche, depending on the chemical structure of the herbicides [1], [11]. It has also been suggested that these differences are mainly due to variations in the release kinetics, i.e. the kinetics related to their residence times at D1 proteins, which are determined principally by the accessibility of the niche to the compound and depend on the chemical structure of herbicides [11]. Compared with the residence time of QB (which is ~20 ms), those of the herbicides were much higher and vary from approximately 10-fold for ioxynil to approximately 25,000-fold for DCMU [11]. Therefore, the herbicide inhibition is stronger when the time of its stays at the D1 protein is longer. A stationary binding, resulting in a significant electron transport inhibition, requires a strict molecular shape and most probably changes the D1 conformation followed by donor side modifications. In addition, our data showed the influence of the studied herbicides on the kinetic parameters of OEC: the initial S0 state, the blocked centers (SB) and the rate constant of the turnover time of oxygen-evolving centers (KD), in different extents for phenylurea and phenol-type herbicides (Table 2). It could be assumed that this variation in the kinetic parameter of the oxygen evolution could result from the different residence times of the herbicides at the QB niche. The inhibitory effects of herbicides on the PSII function decreased in the following order: DCMU>isoproturon>ioxynil. The higher herbicide sensitivity of algal and cyanobacterial cells to phenylurea herbicides in comparison with phenol-type herbicides is accompanied by the strongly increased number of blocked oxygen-evolving centers in them and/or the increased turnover time of the oxygen-evolving centers (a decrease of the rate constant KD) after treatment with phenylurea herbicides. The obtained results also demonstrated that the parameters of the photosynthetic oxygen evolution are more sensitive and appropriate for bioassays of the effect of phenylurea herbicides on aquatic ecosystems.
Data in the present study revealed that the PSII activity (measured with exogenous electron acceptor BQ) was less reduced by herbicides than the oxygen-evolving parameters measured with Joliot-type electrode (without exogenous electron acceptor) (Figures 2 and 4). These findings could be result of different oxidation-reduction properties of the native PQ and artificial BQ and/or of difficult interaction between the QB and PQ, which suggests impairment on the acceptor side of PSII by herbicides. In addition, the decrease of the parameter Fv/F0 also suggests an alteration in the acceptor side of the PSII complex. Our study also proposes alterations in the OEC on the donor side, which is considered by the changes in the kinetic parameters of the oxygen evolution: redox state of the Mn cluster on the PSII donor side in the darkness, misses, double hits and rate constant KD. It has been proposed that structural changes or conformations in the Mn cluster could be responsible for different S0−S1 initial distributions in darkness as well as the kinetics of oxygen-evolving reactions [41].
Moreover, it could be proposed that one of the reasons for the higher sensitivity of the PSII function to herbicides in cyanobacteria in comparison with algae is due to the higher number of blocked centers (SB) in cyanobacterial cells (Table 2). Another reason might be the different organizations of their photosynthetic membranes [3], [4]: the cyanobacterial cells exhibit an internal thylakoid system organized as a series of roughly parallel double-membrane layers distributed within the cytoplasm, whereas green algae have chloroplasts, which contain an inner membrane system formed by bands of stacked thylakoids (grana) and thylakoids running singly in the stroma. It has also been established that there are differences in the polypeptides composition of OEC and peripheral light-harvesting antenna of PSII between cyanobacteria and green algae [3], [4]. In addition, the rate constant of excited Si states (KD) is almost twice smaller in the Synechocystis cells than in the Chlorella cells; indicating bigger turnover times of the oxygen-evolving reactions in cyanobacteria.
5 Conclusion
The present data shows that the phenylurea herbicides (DCMU and isoproturon) more strongly inhibit the PSII activity of cyanobacteria and green algae in comparison with the phenol-type herbicide (ioxynil). Additionally, chlorophyll fluorescence ratios ΔF/Fm′ and Fv/F0, and the PSII-mediated electron transport were more strongly influenced in Synechocystis than in Chlorella cells. Moreover, the parameters of the oxygen evolution were more sensitive to herbicides than the PSII photochemistry, which could be due to changes in the kinetic parameters of the oxygen-evolving reactions on the PSII donor side (the number of PSII centers in the initial S0 state, the blocked centers (SB), the rate constants of the turnover time (KD), misses and double hits). The herbicides studied can be attributed to a group of so-called ADRY reagents considering their strong influence on the OEC. Therefore, it could be suggested that the reasons for the different herbicide inhibitions of PSII are not only their interactions with QB on the acceptor side but also their induced alterations in the OEC on the donor side that influences the kinetic parameters of oxygen evolution to different extents.
Acknowledgements
This work was supported by the Bulgarian Academy of Sciences. The authors would like to thank Assoc. Prof. Irina Puneva from the Institute of Plant Physiology and Genetics, Bulgarian Academy of Sciences, Bulgaria for cultivation of green algae and cyanobacteria.
References
1. Trebst A. The three-dimensional structure of the herbicide binding niche on the reaction center polipeptides of photosystem II. Z Naturforsch 1987;42c:742–50.10.1515/znc-1987-0616Search in Google Scholar
2. Rast A, Heinz S, Nickelsen J. Biogenesis of thylakoid membranes. Biochim Biophys Acta 2015;1847:821–30.10.1016/j.bbabio.2015.01.007Search in Google Scholar
3. Moro I, La Rocca N, Rascio N. Photosynthetic apparatus in cyanobacteria and microalgae. In: Handbook of Photosynthesis (ed. M. Pessarakli), 3rd ed. CRC press, Boca Raton, FL, 2016, pp. 629–49.10.1201/b19498-27Search in Google Scholar
4. Barber J. Crystal structure of the oxygen-evolving complex of photosystem II. Inorg Chem 2008;47:1700–10.10.1021/ic701835rSearch in Google Scholar
5. Zimmermann K, Heck M, Frank J, Kern J, Vass I, Zouni A. Herbicide binding and thermal stability of photosystem II isolated from Thermosynechococcus elongates. Biochim Biophys Acta 2006;1757:106–14.10.1016/j.bbabio.2005.12.002Search in Google Scholar
6. Fai B, Grant A, Reid B. Chlorophyll-a fluorescence as a biomarker for rapid toxicity assessment. Env Toxicol Chem 2007;26:1520–31.10.1897/06-394R1.1Search in Google Scholar
7. Draber W, Tietjen K, Kluth JF, Trebst A. Herbicides in photosynthesis research. Angew Chem Int Ed Engl 1991;30:1621–33.10.1002/anie.199116211Search in Google Scholar
8. Ajlani G, Meyer I, Vernotte C, Astier C. Mutation in phenol type herbicide resistance maps within the psbA gene in Synechocystis 6714. FEBS Lett 1989;246:207–10.10.1016/0014-5793(89)80284-4Search in Google Scholar
9. Perewoska I, Vernotte V, Picaud M, Astier C. Mutation in the D1 subunit of photosystem II and resistance to the phenol type herbicide ioxynil in Synechocystis PCC 6714 and 6803. Z Naturforsch 1992;47c:580–4.10.1515/znc-1992-7-815Search in Google Scholar
10. Allen MM, Turnburke AC, Lagace E, Steinback KE. Effects of photosystem II herbicides on the photosynthetic membranes of the cyanobacterium Aphanocapsa 6308. Plant Physiol 1983;71:388–92.10.1104/pp.71.2.388Search in Google Scholar PubMed PubMed Central
11. Naber DJ, van Rensen JJ. Activity of photosystem II herbicides in related with their residence times at D1 proteins. Z Naturforsch 1991;46c:575–8.10.1515/znc-1991-7-812Search in Google Scholar
12. Singh DV, Adeppa K, Misra K. Mechanism of isoproturon resistance in Phalaris minor: in silico design, synthesis and testing of some novel herbicides for regaining sensitivity. J Mol Model 2012;18:1431–45.10.1007/s00894-011-1169-2Search in Google Scholar
13. Rutherford AW, Krieger-Liszkay A. Herbicide-induced oxidative stress in photosystem II. Trends Biochem Sci 2001;26:648–53.10.1016/S0968-0004(01)01953-3Search in Google Scholar
14. Bashir F, Jan S. Oxidative stress and antioxidant defence system in response to pesticide stress. In: Legumes under environmental stress (eds. M.M. Azooz, P. Ahmad), John Wiley & Sons, Ltd. Chichester, UK, 2015, pp. 103–24.10.1002/9781118917091.ch7Search in Google Scholar
15. Matzenbacher FO, Vidal RA, Merottojr A, Trezzi MM. Environmental and physiological factors that affect the efficacy of herbicides that inhibit the enzyme protoporphyrinogen oxidase: literature review. Planta Daninha Viçosa-MG 2014;32:457–63.10.1590/S0100-83582014000200024Search in Google Scholar
16. Ma J, Xu L, Wang S, Zheng R, Jin S, Huang S, et al. Toxicity of 40 herbicides to the green alga Chlorella vulgaris. Ecotoxicol Environ Saf 2002;51:128–32.10.1006/eesa.2001.2113Search in Google Scholar
17. Rioboo C, González O, Herrero C, Cid A. Physiological response of freshwater microalga (Chlorella vulgaris) to triazine and phenylurea herbicides. Aquat Toxicol 2002;59:225–35.10.1016/S0166-445X(01)00255-7Search in Google Scholar
18. Rashkov G, Dobrikova AG, Pouneva ID, Misra AN, Apostolova EL. Sensitivity of Chlorella vulgaris to herbicides. Possibility of using it as a biological receptor in biosensors. Sens Actuators B Chem 2012;161:151–5.10.1016/j.snb.2011.09.088Search in Google Scholar
19. Ferro Y, Perulloni Y, Jobbagy M, Bilmes SA, Durrieu C. Development of a biosensor for environmental monitoring based on microalgae immobilized in silica hydrogel. Sensors 2012;12:16879–91.10.3390/s121216879Search in Google Scholar PubMed PubMed Central
20. Sjollema SB, García GM, Van der Geest HG, Kraak MH, Booij P, Vethaak AD, et al. Hazart and risk of herbicides for marine microalgae. Environ Pollut 2014;187:106–11.10.1016/j.envpol.2013.12.019Search in Google Scholar PubMed
21. Kumar KS, Dahms HU, Lee JS, Kim HC, Lee WC. Algal photosynthetic responses to toxic metals and herbicides assessed by chlorophyll a fluorescence. Ecotoxicol Environ Saf 2014;104:51–71.10.1016/j.ecoenv.2014.01.042Search in Google Scholar
22. Apostolova EL, Pouneva I, Rashkov G, Dankov K, Grigorova I, Misra AN. Effect of UV-B radiation on photosystem II functions in Antarctic and mesophilic strains of a green alga Chlorella vulgaris and a cyanobacterium Synechocystis salina. Indian J Plant Physiol 2014;19:111–8.10.1007/s40502-014-0084-9Search in Google Scholar
23. Georgiev D, Dilov H, Avramova S. Millieu nutritif tamponne et méthode de culture intensive des microalgues vertes. Hydrobiology (Bulgaria) 1978;7:14–23.Search in Google Scholar
24. Pekárková B, Hindák F, Šmarda J. Morphological characteristics and physiological properties of a coccoid rhodophycean alga Rhodella grisea from thermal springs at Pieštany Czechoslovakia. Arch Protistenkunde 1988;135:69–83.10.1016/S0003-9365(88)80055-1Search in Google Scholar
25. Chow WS, Hope B, Anderson JM. A reassessment of the use of herbicide binding to measure photosystem II reaction centres in plant thylakoids. Photosynth Res 1990;24:109–13.10.1007/BF00032650Search in Google Scholar PubMed
26. Rohacek K. Chlorophyll fluorescence parameters: the definitions, photosynthetic meaning and mutual relationships. Photosynthetica 2002;40:13–29.10.1023/A:1020125719386Search in Google Scholar
27. Lichtenthaler K, Buschmann C, Knapp M. How to correctly determine the different chlorophyll fluorescence parameters and the chlorophyll fluorescence decrease ratio RFd of leaves with the PAM fluorometer. Photosynthetica 2005;43:379–93.10.1007/s11099-005-0062-6Search in Google Scholar
28. Zeinalov Y. An equipment for investigations of photosynthetic oxygen production reactions. Bulg J Plant Physiol 2002;28:57–67.Search in Google Scholar
29. Kok B, Forbush B, McGloin M. Co-operation of charges in photosynthetic O2 evolution. I. A linear four step mechanism. Photochem Photobiol 1970;11:457–75.10.1111/j.1751-1097.1970.tb06017.xSearch in Google Scholar PubMed
30. Shinkarev VP. Flash-induced oxygen evolution in photosynthesis: simple solution for the extended S-state model that includes misses, double-hits, inactivation, and backward-transition. Biophys J 2005;88:412–21.10.1529/biophysj.104.050898Search in Google Scholar PubMed PubMed Central
31. Zeinalov Y. Photosynthesis – behind the fundamental concepts. Saarbrücken: LAP Lambert Academic Publishing, AG & Co. KG, 2010.Search in Google Scholar
32. Zeinalov Y. On the minimum quantum requirement of photosynthesis. Z. Naturforsch. 2009;64c:673–9.10.1515/znc-2009-9-1011Search in Google Scholar PubMed
33. Apostolova EL, Dobrikova AG, Ivanova PI, Petkanchin IB, Taneva SG. Relationship between the organization of the PSII supercomplex and the functions of the photosynthetic apparatus. J Photochem Photobiol B Biol 2006;83:114–22.10.1016/j.jphotobiol.2005.12.012Search in Google Scholar PubMed
34. Dobrikova AG, Domonkos I, Sözer Ö, Laczkó-Dobos H, Kis M, Párduc Á, et al. Effect of partial or complete elimination of light-harvesting complexes on the surface electric properties and the functions of cyanobacterial photosynthetic membranes. Physiol Plantarum 2013;147:248–60.10.1111/j.1399-3054.2012.01648.xSearch in Google Scholar
35. Flores EJ, Collier CJ, Mercurio P, Negri AP. Phytotoxicity of four photosystem II herbicides to tropical seagrasses. PLoS One 2013;8:e75798.10.1371/journal.pone.0075798Search in Google Scholar
36. Singh S, Datta P, Tirkey A. Response of multiple herbicide resistant strain of diazotrophic cyanobacterium, Anabaena variabilis, exposed to atrazine and DCMU. Indian J Exp Biol 2011;49:298–303.Search in Google Scholar
37. Rutherford AW, Zimmermann JL, Mathis P. The effect of herbicides on components of the PS II reaction centre measured by EPR. FEBS Lett 1984;165:156–62.10.1016/0014-5793(84)80161-1Search in Google Scholar
38. Abdel-Basset R, Bader KP. Physiological analyses of the hydrogen gas exchange in cyanobacteria. J Photochem Photobiol B Biol 1998;43:146–51.10.1016/S1011-1344(98)00097-9Search in Google Scholar
39. Nagarajan A, Page LE, Liberton M, Pakrasi HB. Consequences of decreased light harvesting capability on photosystem II function in Synechocystis sp. PCC 6803. Life 2014;4:903–14.10.3390/life4040903Search in Google Scholar
40. Chiu Y-F, Chen Y-H, Roncel M, Dilbeck PL, Huang J-Y, Ke S-C, et al. Spectroscopic and functional characterization of cyanobacterium Synechocystis PCC 6803 mutants on the cytoplasmic-side of cytochrome b559 in photosystem II. Biochim Biophys Acta 2013;827:507–19.10.1016/j.bbabio.2013.01.016Search in Google Scholar
41. Nugent JH, Rich AM, Evans MC. Photosynthetic water oxidation: towards a mechanism. Biochim Biophys Acta 2001;1503:138–46.10.1016/S0005-2728(00)00223-1Search in Google Scholar
©2017 Walter de Gruyter GmbH, Berlin/Boston
Articles in the same Issue
- Frontmatter
- Special Section – Tribute
- Lothar Jaenicke and C1-metabolism: his first 25 years of research
- Lothar Jaenicke (1923–2015) zum Gedächtnis
- Special Section – Research Articles
- The AtMYB12 activation domain maps to a short C-terminal region of the transcription factor
- 3,7-Isoquinoline quinones from the ascidian tunicate Ascidia virginea
- Scent gland constituents of the Middle American burrowing python, Loxocemus bicolor (Serpentes: Loxocemidae)
- Effects of extracts and compounds from Tricholoma populinum Lange on degranulation and IL-2/IL-8 secretion of immune cells
- A succinct access to ω-hydroxylated jasmonates via olefin metathesis
- Research Articles
- PEGylation potentiates hepatoma cell targeted liposome-mediated in vitro gene delivery via the asialoglycoprotein receptor
- A detailed experimental study of a DNA computer with two endonucleases
- Different sensitivities of photosystem II in green algae and cyanobacteria to phenylurea and phenol-type herbicides: effect on electron donor side
- Response of alternative splice isoforms of OsRad9 gene from Oryza sativa to environmental stress
Articles in the same Issue
- Frontmatter
- Special Section – Tribute
- Lothar Jaenicke and C1-metabolism: his first 25 years of research
- Lothar Jaenicke (1923–2015) zum Gedächtnis
- Special Section – Research Articles
- The AtMYB12 activation domain maps to a short C-terminal region of the transcription factor
- 3,7-Isoquinoline quinones from the ascidian tunicate Ascidia virginea
- Scent gland constituents of the Middle American burrowing python, Loxocemus bicolor (Serpentes: Loxocemidae)
- Effects of extracts and compounds from Tricholoma populinum Lange on degranulation and IL-2/IL-8 secretion of immune cells
- A succinct access to ω-hydroxylated jasmonates via olefin metathesis
- Research Articles
- PEGylation potentiates hepatoma cell targeted liposome-mediated in vitro gene delivery via the asialoglycoprotein receptor
- A detailed experimental study of a DNA computer with two endonucleases
- Different sensitivities of photosystem II in green algae and cyanobacteria to phenylurea and phenol-type herbicides: effect on electron donor side
- Response of alternative splice isoforms of OsRad9 gene from Oryza sativa to environmental stress


