Abstract
Objectives
The present study identifies the level changes of markers (Apelin, Elabela, Meteorin, ALT, AST, GGT) in the liver after acute intake of high-dose paracetamol and their role in determining liver damage.
Methods
The rats were categorized into two groups (Control and Toxication), each consisting of seven rats. Apelin, Elabela, Meteorin, ALT, AST and GGT levels were determined in liver tissue. The levels of Apelin, Elabela, and Meteorin in liver tissue homogenate were assessed following the kit procedure, utilizing commercial enzyme-linked immunosorbent assay. ALT, AST and GGT analyzes were performed from liver tissue homogenate with Erba colorimetric kit according to the kit procedure. It was also evaluated histopathologically from liver tissue.
Results
Looking at the findings, a significant decrease was determined in the Elabela and Meteorin data in the Apap group compared to the control group. In the Apelin data, a statistical increase was observed in the Apap group compared to the control group. A significant increase in ALT analysis was detected in the APAP group compared to the control group. Considering GGT and AST data, a statistical increase was detected in the APAP group compared to the control group.While Bax, Bcl-2 and iNOS immunoreactivity was not observed in the control group, a yellowish-brown positive reaction was detected in the cytoplasm of degenerated and necrotic hepatocytes, especially around the vena centralis, in the paracetamol group.
Conclusions
As a result, it is thought that Apelin, Elabela and Meteorin markers may be decisive markers in determining toxicity in liver damage.
Introduction
Paracetamol, which is one of the most commonly used drugs for analgesic and antipyretic purposes nowadays, is the active metabolite of acetanilide and phenacetin used as strong analgesics/antipyretics [1]. The fact that the use of phenacetin was discontinued due to its nephrotoxic effect ensured the use of paracetamol as an analgesic and antipyretic. Acetaminophen has many mechanisms of action. The analgesic effect of acetaminophen is similar to NSAIDs, and its first described mechanism is cyclooxygenase (COX) inhibition. It has been demonstrated that acetaminophen exerts its antipyretic effect in the brain by the inhibition of prostaglandin synthesis. The physiological and pharmacological properties of two cyclooxygenase enzymes (COX-1 and COX-2), which are the targets of nonsteroidal anti-inflammatory drugs and catalyze the conversion of arachidonic acid to prostaglandin, thromboxane, and prostacyclin, were discovered in the 1990s. Prostaglandins are mediators of fever, pain, and inflammation [2].
The nitric oxide (NO) side pathway is another analgesic mechanism of action of acetaminophen. The l-arginine-NO side pathway is activated by substance P and N-methyl-d-aspartate (NMDA). At the end of this activation, nociceptive transmission is achieved. Acetaminophen inhibits substance P, a mediator of hyperalgesia. Then, along with the activation of nitric oxide synthase (NOS) inhibitors, antinociception is achieved, and the analgesic effect of acetaminophen increases [3].
Many studies indicate that Acetaminophen (APAP) causes apoptotic cell death [4]. Apoptosis is characterized by regulated signaling mechanisms, and its induction occurs through two main pathways: the extrinsic pathway, mediated by cell death receptors, and the intrinsic pathway, involving the mitochondria. In these two main pathways, intracellular proteases and endonucleases are triggered and cell lysis occurs [5].
Apelin, the ligand of the endogenous receptor of angiotensin-like receptor 1 (APJ), is a peptide that is both synthesized and secreted in adipose tissue associated with angiogenesis and inflammation. Considering its distribution in the body, apelin plays a role in many important pathways, including immune response, hunger and satiety response, gastric acid secretion, insulin production and cholecystokinin secretion [6].
Although apelin was initially identified as the only known ligand of APJ, studies have shown that APJ exhibits functional differences. Based on this, the hypothesis that APJ may have another ligand emerged. It has been determined that both Apelin and Elabela function through the APJ receptor. Moreover, it has been found that Elabela is expressed in the central nervous system, brain and muscle tissue, heart, adipocytes of adipose tissue, kidneys, liver, lung and many other organs and tissues [7].
A new adipokine called meteorin, which is regulated by adipogenesis and obesity, has been discovered. It is well known that the adipokine meteorin improves insulin sensitivity. It is well recognized that adipokines have a role in both lipid metabolism and inflammation.
Macrophages and dendritic cells are also produced by some granulocytes. However, they are not produced by lymphoid cells. They are known to have a role in inflammation and lipid peroxidation.
In this study, we aimed to determine the level changes of biochemical markers (Apelin, Elabela, meteorin) in the liver after acute intake of high dose paracetamol and their role in determining liver damage and to determine the damage mechanism by which it is effective.
Materials and methods
This study was initiated after obtaining approval from the Animal Experiments Local Ethics Committee of Kafkas University (KAÜ-HADYEK 2020/094). This study was conducted between 23/06/2020 and 10/07/2021. The study utilized a total of 14 female Wistar albino rats, aged 4–6 months, and with an average weight ranging between 190 and 250 g. All rats were subjected to vaginal cytology and those in diestrus were included in the study. The rats were categorized into two groups, each consisting of seven rats, based on their weight. These rats were obtained from the Experimental Animals Application and Research Center of Kafkas University, which is officially authorized to produce and sell experimental animals. During the study, the animals were kept in rooms with a temperature of approximately 25 °C in a ventilated environment that could be automatically adjusted to 12 h of light and 12 h of darkness. All animals were fed ad libitum. N-acetyl-p-aminophenol (paracetamol) used in the study was obtained from Atabay Chemical Industry.
Groups;
Group I (Control): Group orally administered only 0.9 % NaCl.
Group II (Toxication): Group in which intoxication was induced by a single oral dose of N-acetyl-p-aminophenol (1 g/kg).
At the last part of the study, the animals were subjected to an overnight fasting period, and the necessary tissue (liver) samples for the study were obtained by euthanizing the animals through cervical spine dislocation under anesthesia, following ethical guidelines.
The obtained tissue samples were homogenized (Wiggen Hauser, D-500) using phosphate buffer (pH 7.4), and the resulting homogenates were separated by centrifugation at 1,500 g for 5 min [8]. Subsequently, these homogenates were stored at −20 °C until the day of analysis. The levels of Apelin, Elabela, and Meteorin (Art No: YLA0312RA, YLA1445RA, YLA1399RA) in liver tissue homogenate were assessed following the kit procedure, utilizing commercial enzyme-linked immunosorbent assay (ELISA) kits procured from YL Biotech Company, Shanghai. For Apelin, the assay range of the kit was 10 ng/L→3000 ng/L, its sensitivity was 5.17 ng/L, its inter assay CV was 10 %, and its intra assay CV was 8 %. For Elabela, the assay range of the kit was 5 pg/mL→700 pg/mL, its sensitivity was 2.23 pg/mL, its inter assay CV was 10 %, and its intra assay CV was 8 %. For Meteorin, the assay range of the kit was 0.05 ng/mL→15 ng/mL, its sensitivity was 0.01 ng/mL, its inter assay CV was 10 %, and its intra assay CV was 8 %. ALT, AST and GGT (Cat. No: BLT00052, BLT00050, BLT00023) analyzes were performed from liver tissue homogenate with Erba colorimetric kit (Erba Lachema, Czech Republic and Slovakia) according to the kit procedure. For ALT, the assay range of the kit was 4.4 U/L→360 U/L, its inter assay CV was 3.12 %, and its intra assay CV was 4.31 %. For AST, the assay range of the kit was 3.84U/l→390U/l, its inter assay CV was 1.37 %, and its intra assay CV was 0.54 %. For GGT, the assay range of the kit was 1.68 U/L→500 U/L, its inter assay CV was 1.61 %, and its intra assay CV was 0.89 %. Lowry protein assay was used as the protein measurement method in tissue [9]. Measurements were made on a molecular devices spectramax brand spectrophotometry device.
Liver tissue samples obtained from the rats were preserved in a 10 % buffered formaldehyde solution. In our study, we utilized the avidin-biotin-peroxidase method as an immunohistochemical technique. For immunohistochemical staining, 4-μm thick sections extracted from paraffin blocks underwent rehydration. To inhibit endogenous peroxidase activity, the sections underwent treatment with a 3 % hydrogen peroxide solution for 15 min. Following this, the microwave method was applied to the sections to expose the antigenic receptors, utilizing Citrate Buffer Solution at pH six for a duration of 25 min. The sections underwent a 30-min incubation with non-immune serum (Genemed Biotechnologies REF 54-0003) to prevent nonspecific staining. Following this, they were incubated every other day with primary antibodies, which were diluted 1/200 in phosphate-buffered salt solution (PBS). The primary antibodies used included Bax (Bioss, bs-0127 R) and Bcl-2 (Bioss, bs-0032 R). The sections were subjected to three 5-min washes in a PBS solution at room temperature, totaling 30 min, before the application of the biotinylated secondary antibody (Genemed Biotechnologies REF 54-0003). Following these PBS washes (3*5 min), all sections underwent a 30-min incubation with peroxidase-conjugated Strep Avidin (Genemed Biotechnologies REF 54-0003). A color-revealing substrate, 3.3-diaminobenzidine tetrahydrochloride (DAB) (Genemed Biotechnologies REF 10-0048), was utilized. Staining with Mayer’s hematoxylin was performed, and the sections were sealed with immune-mount.
Statistical analyses
Analysis of Unpaired t-test was conducted for all the biochemical parameters to test if there is a difference between the two groups. A p-value of <0.05 was accepted as significant. GraphPad 8.1 (San Diego, CA, USA) was used for statistical analyses.
Results
Apelin, Elabela, and Meteorin data were measured to determine inflammatory parameters in liver tissue (Figure 1–A, B, C). The Apelin analysis showed a significant increase in the APAP group compared to the control group (p<0.05). The analysis revealed a statistically significant decrease in the APAP group compared to the control group, considering Elabela (p<0.001) and meteorin (p<0.01) data. Parameters that aspartate aminotransferase (AST), Alanine Aminotransferase (ALT), and Gamma Glutamyl Transferase (GGT) were measured to assess the impact of APAP toxicity on liver tissue (Figure 2–A, B, C). According to the analysis, a significant increase in ALT analysis was detected in the APAP group compared to the control group (p<0.05). Considering GGT (p<0.05) and AST (p<0.01) data, a statistical increase was detected in the APAP group compared to the control group (Figure 2–A, B, C).

Means and std. errors of the two groups (A, B, C) for ECG parameters. *p<0.05, **p<0.01, ***p<0.001.
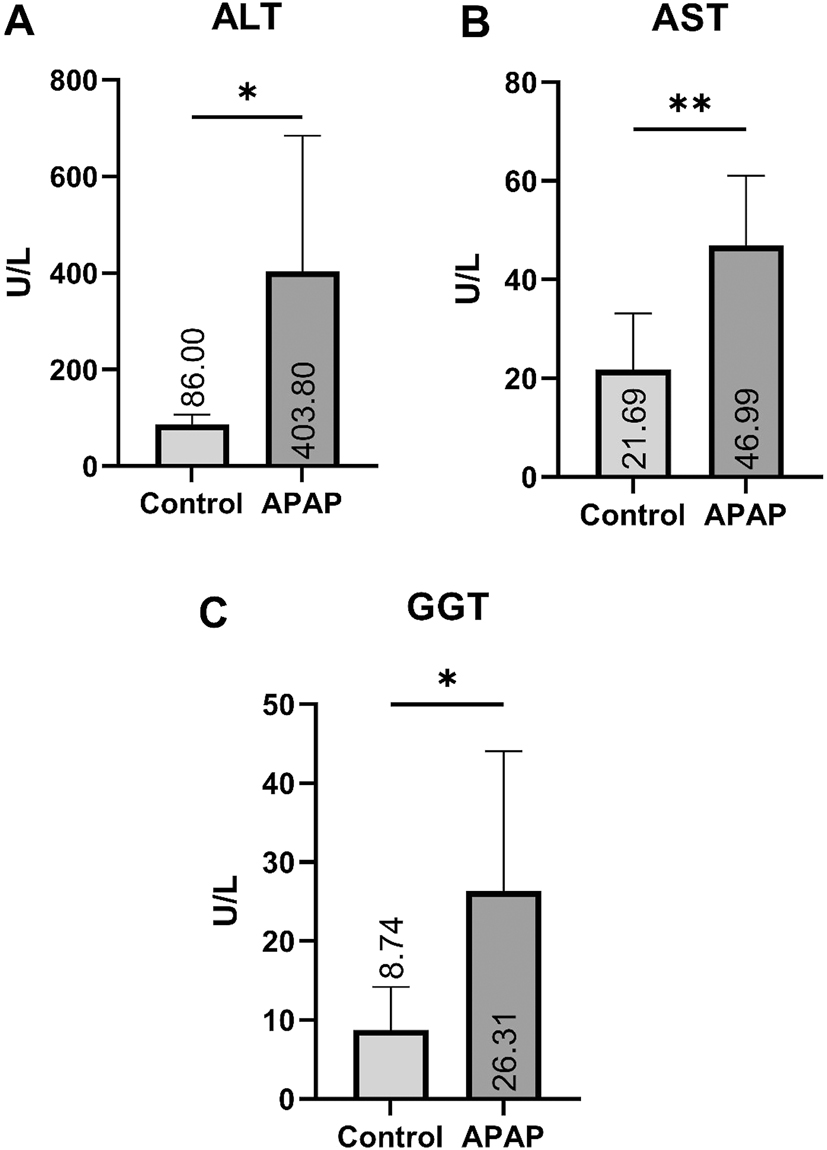
Means and std. errors of the two groups (A, B, C) for AST, ALT, GGT parameters. *p<0.05, **p<0.01.
According to histopathological examinations, there was no lesions in the liver tissue samples taken from the rats in the control group. In the paracetamol group rats, sinusoidal hyperemia in the liver tissue, as well as local degeneration and necrosis in the hepatocytes, were observed. In addition to these changes, the activation of Kupffer’s stellate cells and mononuclear cell infiltrates around the portal triad and vena centralis were observed (Figure 3).

Hematoxylin and eosin staining of the liver. A: Control group, liver tissue, H&E, Bar=50 μm B: Paracetamol group, liver tissue, necrosis in hepatocytes (arrows), H&E, Bar=50 μm.
While no Bax and Bcl-2 immunoreactivity was observed in the control group (Figure 4–a–c), a yellowish-brown Bax and Bcl-2 positive reaction was found in the cytoplasm of degenerated and necrotic hepatocytes, mainly around the vena centralis, in the paracetamol group. The intensity of Bcl-2 expression was higher compared to Bax expression (Figure 4–b–d).
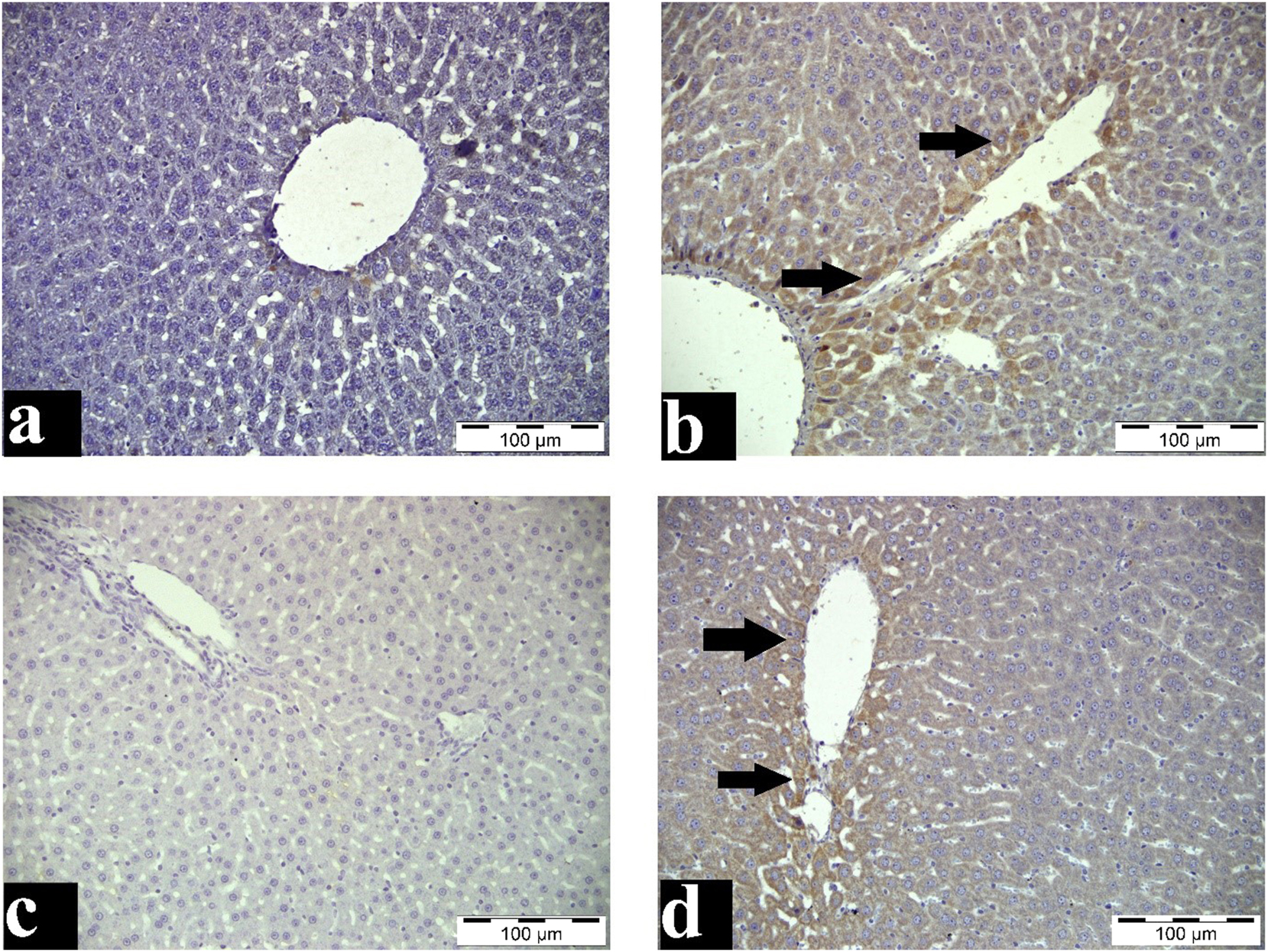
Bax and Bcl-2 immunoreactivity staining of the liver. a: Control group, liver tissue, IHC, Bar=100 μm b: Paracetamol group, liver tissue, Bcl-2-positive immunoreaction in hepatocytes (arrows), IHC, Bar=100 μm c: Control group, liver tissue, IHC, Bar=100 μm d: Paracetamol group, liver tissue, Bax-positive immunoreaction in hepatocytes (arrows), IHC, Bar=100 μm.
While no iNOS immunoreactivity was observed in the control group, a yellowish-brown iNOS positive reaction was found in the cytoplasm of hepatocytes, especially around the vena centralis, in the paracetamol group (Figure 5).
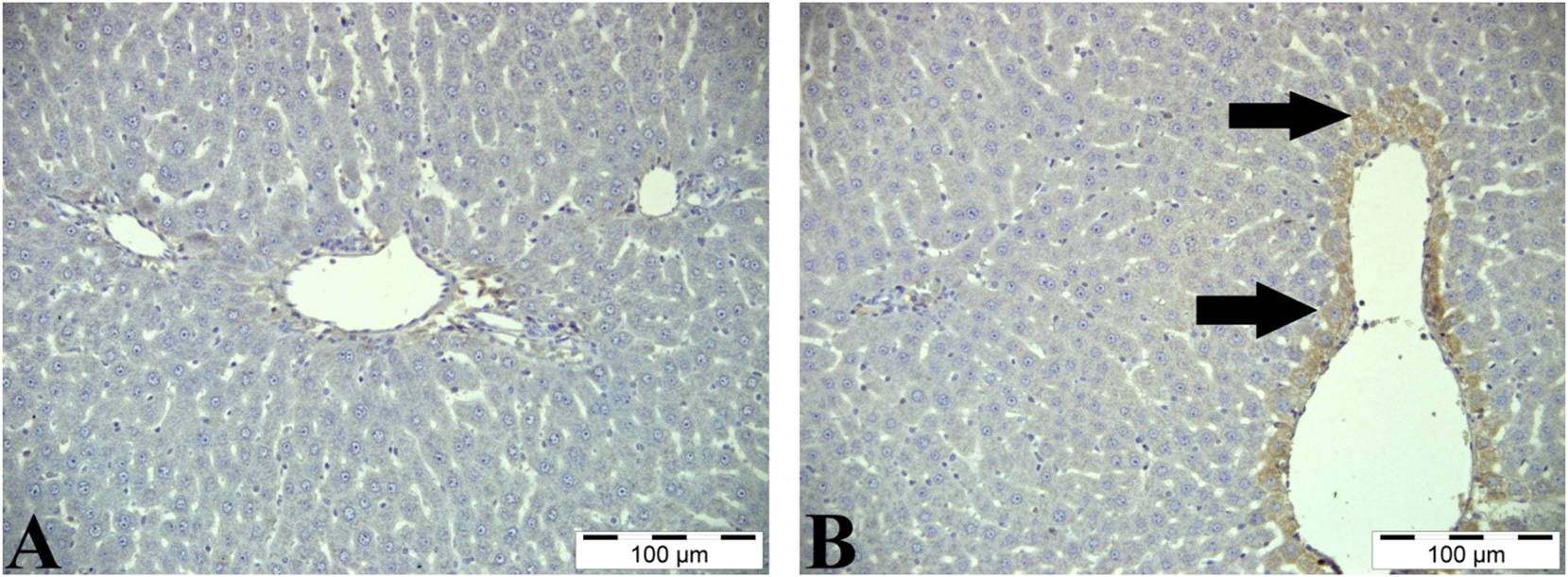
iNOS immunoreactivity staining of the liver. A: Control group, liver tissue, IHC, Bar=100 μm B: Paracetamol group, liver tissue, positive expression of iNOS in hepatocytes (arrows), IHC, Bar=100 μm.
Discussion
Paracetamol (PCT), used as an over-the-counter analgesic and antipyretic, is one of the leading pharmacological causes of acute liver failure. Degeneration in the liver resulting from APAP intoxication is the centrizonal necrosis with relative inflammation and without steatosis. In severe cases, submassive or massive necrosis may be observed. An increase in AST, ALT, and GGT is indicative of liver damage [8]. In the current study, histopathological degenerations in the liver were identified in the group exposed to toxicity.
The toxic dose of APAP plays an active role in nitrotyrosine-protein [9] and NO [10] synthesis. Nitrotyrosine is localized with APAP-protein adducts in the necrotic centrilobular region of the liver. Peroxynitrite is known to be detoxified by GSH/GSH peroxidase. However, intracellular GSH significantly decreases in APAP toxicity. It has been demonstrated that APAP hepatotoxicity is associated with increased iNOS expression and the formation of nitrotyrosine-protein adducts [11]. In the paracetamol group, we found a yellowish-brown iNOS positive reaction in the cytoplasm of hepatocytes, especially around the vena centralis, and we thought that the reason for this was the decreased GSH/GSH peroxidase activity.
High doses of acetaminophen were found to be associated with inflammation in the liver through cytokines [12].
The high-dose intake of APAP leads to protein damage within the mitochondria. This damage subsequently inhibits the electron transport chain (ETC), causing electron loss and resulting in oxidative stress.
Elevated oxidative stress, coupled with the relocation of Bax from the cytoplasm to the mitochondria, triggers mitochondrial permeability transition. This mechanism culminates in the liberation of apoptosis-inducing factor (AIF) and endonuclease G into the cytoplasm. Consequently, Endonuclease G migrates to the nucleus, instigating DNA fragmentation in a subsequent phase. Excessive APAP intake also induces an upregulation of RIP3, resulting in the translocation of DRP1 to the mitochondria, instigating mitochondrial fission and mitophagy. Collectively, these occurrences contribute to necrotic cell death [13].
Apelin has been identified as a participant in various processes within the liver, including hepatocyte apoptosis, glycogen synthesis, and the formation of fibrosis. Specifically, in the liver, apelin has been found to paticipate in hepatocyte apoptosis, glycogen synthesis, and the development of fibrosis. Under hypoxic or proinflammatory conditions, apelin is notably expressed at high levels in hepatic stellate cells (HSCs), contributing to the progression of liver fibrosis or cirrhosis [14]. A study reported a significant increase in apelin expression in the hepatic tissue of cirrhotic rats compared to control subjects [15].
In our study, apelin levels were found to be higher in liver tissue in the APAP group compared to the control group after the development of liver cellular damage, and we can think of this as the increased release by adipocytes secondary to inflammation or insulin resistance after liver tissue damage and hepatic stellate cell damage.
It is emphasized that the C-terminal residue of elabela has a crucial role in its binding to the APJ receptor and signaling interaction, mirroring the mechanism observed with apelin. The C-terminal residues of elabela are vital for both stimulation and binding processes. Additionally, the plasma half-life of elabela was determined to be a few minutes [16]. Elabela/Apelin has the capability to activate the APJ receptor, exerting an inhibitory effect on adenylyl cyclase (AC) activity and cAMP production by triggering diverse intracellular signaling pathways.
It has been reported that elabela is synthesized in human embryonic stem cells, even though these cells do not produce the APJ receptor. This noteworthy discovery indicates that APJ is not the exclusive receptor for elabela. Studies have demonstrated that ELA (Elabela) inhibits apoptosis by suppressing apoptotic proteins and activating anti-apoptotic proteins [17]. Another investigation revealed that the non-coding region of elabela plays a regulatory role in p53-mediated DNA damage-induced apoptosis in mouse embryonic stem cells [17]. In a separate study, it was found that the absence of elabela in cells with high levels of p53 activity resulted in a decrease in cell number due to cell death, attributed to p53-induced apoptosis [18]. Furthermore, another study indicated that inflammatory and fibrotic genes significantly downregulated the mRNA levels of the ischemia-reperfusion (H/R)-induced kidney injury marker (Kim1) and elabela in H/R-damaged NRK-52 E cells. This study also highlighted that the H/R-induced elevation of Kim1 in damaged cells, along with the inflammatory and fibrotic genes, contributed to apoptosis-associated DNA damage during renal injury [19].
It was demonstrated that after the injection of elabela peptide into rats undergoing MI, the infarct size, LDH serum levels, CK-MB, Troponin I and MDA levels formed in tissues decreased significantly, and that elabela peptide played an important role in heart development, vascular and modulation of heart functions, increased antioxidants, reduced apoptosis, reduced oxidative damage and might have healing effects on muscle damage [20]. In the study, it is thought that elabela application has a healing effect on muscle damage. In our study, it is seen that elabela is high in the control group and low in the study group. In this context, the decrease in elabela levels in liver damage is compatible with the study. Thus, we think that elabela may be a definitive marker of lung damage.
In our study, Elabela levels were determined to be significantly lower in liver tissue, and this can be interpreted as the mechanism by which it reduces apoptosis, but since it is predominantly present in our tissues, or the oxidant-antioxidant mechanism balance shifts in the oxidant direction after serious liver damage, or the half-life of Elabela ends within minutes, or it suggests that apelin is more sensitive to the APJ receptor, or we can think that it inhibits the synthesis of inflammatory and fibrotic genes after the damage.
Elabela (ELA) is a signaling molecule or peptide hormone that holds significance in endoderm differentiation, morphogenesis, angiogenesis, and the normal development of heart tissue [22]. In the research conducted by Rakhshan K and colleagues, it was demonstrated that hazel peptide could procure cardioprotection against myocardial ischemia/reperfusion (I/R) damage. This protective effect was attributed to the targeted reduction of oxidative stress, leading to a decrease in heart dysfunction. In this case, it shows that the crime is used in prevention of oxidative stress [23]. In our study, elabela was used in the organism against oxidif damage caused by APAP. At the same time, as the elabela is dependent on the receptor of the elabela, the existing APELIN receptor level increased as the level of Elabela increased. Metrnl establishes connections between host-adaptive responses and the regulation of energy homeostasis, playing a pivotal role in addressing tissue inflammation. This adipomyokine, recently identified as meteorin-like (metrnl), exerts positive effects on glucose metabolism. Despite its recognized impact on glucose homeostasis, insulin sensitivity, glucose tolerance, and fat oxidation, the comprehensive mechanism of its action remains not fully understood. Metrnl holds therapeutic potential for addressing metabolic and inflammatory diseases [24].
Metrnl/mtrn activates AMP-activated kinase (AMPK) in various tissues, including skeletal muscles, adipose tissue, and the liver, through processes that elevate the AMP/ATP ratio. The activated AMPK, in turn, promotes glucose uptake in skeletal muscles, triggers fatty acid oxidation in adipose tissue, and decreases hepatic glucose production. Moreover, it has been reported that the suppression or prevention of phosphorylation of AMPK leads to increased apoptosis in ADR-induced heart failure [25]. Other studies have demonstrated that AMPK is associated with pro-apoptotic (p53 protein, BAX, and p38 MAPK) or anti-apoptotic pathways. It has been found that AMPK activates apoptosis via p53 when suppressed [26].
In our study, meteorin levels were found to be significantly lower compared to the control group. We think that the reason for the low levels of meteorin in liver tissue may be that the structure of meteorin has been disrupted as a result of NAPQI formation and serious liver damage due to acute intoxication with paracetamol, causing a decrease in the number of cells due to cell death as a result of loss of AMP-activated kinase (AMPK) activity, and subsequently p53-induced cell apoptosis.
Conclusions
We believe that serious damage occurs in the liver after high-dose intake of paracetamol, detoxified by the liver, and that apelin, Elabela, and meteorin may be new biomarkers that can be used to show the presence of damage. A total of seven animals had to be used in the study within the framework of experimental animal ethics rules. Likewise, it is among our scientific recommendations to include more studies and different biomarkers for the reliability of markers.
-
Research ethics: This study was initiated after obtaining approval from the Animal Experiments Local Ethics Committee of University (HADYEK).
-
Informed consent: Not applicable.
-
Author contributions: TD, MM, HFG, and FB analyzed and interpreted the data and were major contributors to writing the manuscript. EK analyzed and interpreted the pathology data. All authors read and approved the final manuscript.
-
Use of Large Language Models, AI and Machine Learning Tools: None declared.
-
Conflict of interest: The authors state no conflict of interest.
-
Research funding: No funding sources sponsored the study.
-
Data availability: The datasets during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
1. Makav, M, Dolanbay, T, Gül, HF, Karakurt, E. Sıçanlarda APAP kaynaklı toksisitede EKG, oksidatif stres ve anjiyogenezin belirlenmesi. Kafkas Univ Vet Fak Derg 2021;27:483–8.10.9775/kvfd.2021.25733Search in Google Scholar
2. Mattia, C, Coluzzi, F. What anesthesiologists should know about paracetamol (acetaminophen). Minerva Anestesiol 2009;75:644–53.Search in Google Scholar
3. Bujalska, M. Effect of nitric oxide synthase inhibition on antinociceptive action of different doses of acetaminophen. Pharmacol Rep 2004;56:605–10.Search in Google Scholar
4. Jaeschke, H, Ramachandran, A. Acetaminophen-induced apoptosis: facts versus fiction. J Clin Transl Res 2020;6:36.Search in Google Scholar
5. Lossi, L. The concept of intrinsic versus extrinsic apoptosis. Biochem J 2022;479:357–84. https://doi.org/10.1042/bcj20210854.Search in Google Scholar PubMed
6. Masoumi, J, Jafarzadeh, A, Khorramdelazad, H, Abbasloui, M, Abdolalizadeh, J, Jamali, N. Role of Apelin/APJ axis in cancer development and progression. Adv Med Sci 2020;65:202–13. https://doi.org/10.1016/j.advms.2020.02.002.Search in Google Scholar PubMed
7. Rozwadowski, J, Borodzicz-Jażdżyk, S, Czarzasta, K, Cudnoch-Jędrzejewska, A. A review of the roles of apelin and ELABELA peptide ligands in cardiovascular disease, including heart failure and hypertension. Med Sci Monit 2022;28. https://doi.org/10.12659/msm.938112.Search in Google Scholar
8. Makav, M, Kuru, M, Aras, ŞY, Sarı, EK, Bulut, M, Alwazeer, D. The effect of hydrogen-rich water on letrozole-induced polycystic ovary syndrome in rats. Reprod Biomed Online 2023;47:103332. https://doi.org/10.1016/j.rbmo.2023.103332.Search in Google Scholar PubMed
9. OliverH, L, NiraJ, R, Farr, AL, RoseJ, R. Proteın measurement wıth the folın phenol reagent. J Biol Chem 1951;193:265–75. https://doi.org/10.1016/s0021-9258(19)52451-6.Search in Google Scholar
10. Kim, SH, Choi, HJ, Seo, H, Kwon, D, Yun, J, Jung, YS. Downregulation of glutathione-mediated detoxification capacity by binge drinking aggravates acetaminophen-induced liver injury through IRE1α ER stress signaling. Antioxidants 2021;10:1949. https://doi.org/10.3390/antiox10121949.Search in Google Scholar PubMed PubMed Central
11. Firozian, F, Karami, S, Ranjbar, A, Azandaryani, MT, Nili-Ahmadabadi, A. Improvement of therapeutic potential N-acetylcysteine in acetaminophen hepatotoxicity by encapsulation in PEGylated nano-niosomes. Life Sci 2020;255:117832. https://doi.org/10.1016/j.lfs.2020.117832.Search in Google Scholar PubMed
12. James, LP, McCullough, SS, Lamps, LW, Hinson, JA. Effect of N-acetylcysteine on acetaminophen toxicity in mice: relationship to reactive nitrogen and cytokine formation. Toxicol Sci 2003;75:458–67. https://doi.org/10.1093/toxsci/kfg181.Search in Google Scholar PubMed
13. Yang, T, Wang, H, Wang, X, Li, J, Jiang, L. The dual role of innate immune response in acetaminophen-induced liver injury. Biology 2022;11:1057. https://doi.org/10.3390/biology11071057.Search in Google Scholar PubMed PubMed Central
14. Jaeschke, H, Duan, L, Akakpo, JY, Farhood, A, Ramachandran, A. The role of apoptosis in acetaminophen hepatotoxicity. Food Chem Toxicol 2018;118:709–18. https://doi.org/10.1016/j.fct.2018.06.025.Search in Google Scholar PubMed PubMed Central
15. Principe, A, Melgar-Lesmes, P, Fernández-Varo, G, del Arbol, LR, Ros, J, Morales-Ruiz, M, et al.. The hepatic apelin system: a new therapeutic target for liver disease. Hepatology 2008;48:1193–201. https://doi.org/10.1002/hep.22467.Search in Google Scholar PubMed
16. Chen, H, Wang, L, Wang, W, Cheng, C, Zhang, Y, Zhou, Y, et al.. ELABELA and an ELABELA fragment protect against AKI. J Am Soc Nephrol 2017;28:2694–707. https://doi.org/10.1681/asn.2016111210.Search in Google Scholar PubMed PubMed Central
17. Rakhshan, K, Azizi, Y, Naderi, N, Ghardashi Afousi, A, Aboutaleb, N. ELABELA (ELA) peptide exerts cardioprotection against myocardial infarction by targeting oxidative stress and the improvement of heart function. Int J Pept Res Therapeut 2019;25:613–21. https://doi.org/10.1007/s10989-018-9707-8.Search in Google Scholar
18. Barra, NG, Chew, MV, Holloway, AC, Ashkar, AA. Interleukin-15 treatment improves glucose homeostasis and insulin sensitivity in obese mice. Diabetes Obes Metabol 2012;14:190–3. https://doi.org/10.1111/j.1463-1326.2011.01495.x.Search in Google Scholar PubMed
19. Yu, Y, Xie, M, He, YL, Xu, WQ, Zhu, S, Cao, LZ. [Role of high mobility group box 1 in adriamycin-induced apoptosis in leukemia K562 cells]. Ai Zheng 2008;27:929–33.Search in Google Scholar
20. Huang, J-Y, Lin, Y-C, Chen, H-M, Lin, JT, Kao, SH. Adenine combined with cisplatin promotes anticancer activity against hepatocellular cancer cells through AMPK-mediated p53/p21 and p38 MAPK cascades. Pharmaceuticals 2022;15:795. https://doi.org/10.3390/ph15070795.Search in Google Scholar PubMed PubMed Central
© 2025 the author(s), published by De Gruyter, Berlin/Boston
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Frontmatter
- Review Article
- Unveiling the hidden clinical and economic impact of preanalytical errors
- Research Articles
- To explore the role of hsa_circ_0053004/hsa-miR-646/CBX2 in diabetic retinopathy based on bioinformatics analysis and experimental verification
- Study on the LINC00578/miR-495-3p/RNF8 axis regulating breast cancer progression
- Comparison of two different anti-mullerian hormone measurement methods and evaluation of anti-mullerian hormone in polycystic ovary syndrome
- The evaluation of the relationship between anti angiotensin type I antibodies in hypertensive patients undergoing kidney transplantation
- Evaluation of neopterin, oxidative stress, and immune system in silicosis
- Assessment of lipocalin-1, resistin, cathepsin-D, neurokinin A, agmatine, NGF, and BDNF serum levels in children with Autism Spectrum Disorder
- Regulatory nexus in inflammation, tissue repair and immune modulation in Crimean-Congo hemorrhagic fever: PTX3, FGF2 and TNFAIP6
- Pasteur effect in leukocyte energy metabolism of patients with mild, moderate, and severe COVID-19
- Thiol-disulfide homeostasis and ischemia-modified albumin in patients with sepsis
- Myotonic dystrophy type 1 and oxidative imbalance: evaluation of ischemia-modified albumin and oxidant stress
- Antioxidant and alpha-glucosidase inhibitory activities of flavonoids isolated from fermented leaves of Camellia chrysantha (Hu) Tuyama
- Examination of the apelin signaling pathway in acetaminophen-induced hepatotoxicity in rats
- Integrating network pharmacology, in silico molecular docking and experimental validation to explain the anticancer, apoptotic, and anti-metastatic effects of cosmosiin natural product against human lung carcinoma
- Validation of Protein A chromatography: orthogonal method with size exclusion chromatography validation for mAb titer analysis
- The evaluation of the efficiency of Atellica UAS800 in detecting pathogens (rod, cocci) causing urinary tract infection
- Case Report
- Exploring inherited vitamin B responsive disorders in the Moroccan population: cutting-edge diagnosis via GC-MS profiling
- Letter to the Editor
- Letter to the Editor: “Gene mining, recombinant expression and enzymatic characterization of N-acetylglucosamine deacetylase”
Articles in the same Issue
- Frontmatter
- Review Article
- Unveiling the hidden clinical and economic impact of preanalytical errors
- Research Articles
- To explore the role of hsa_circ_0053004/hsa-miR-646/CBX2 in diabetic retinopathy based on bioinformatics analysis and experimental verification
- Study on the LINC00578/miR-495-3p/RNF8 axis regulating breast cancer progression
- Comparison of two different anti-mullerian hormone measurement methods and evaluation of anti-mullerian hormone in polycystic ovary syndrome
- The evaluation of the relationship between anti angiotensin type I antibodies in hypertensive patients undergoing kidney transplantation
- Evaluation of neopterin, oxidative stress, and immune system in silicosis
- Assessment of lipocalin-1, resistin, cathepsin-D, neurokinin A, agmatine, NGF, and BDNF serum levels in children with Autism Spectrum Disorder
- Regulatory nexus in inflammation, tissue repair and immune modulation in Crimean-Congo hemorrhagic fever: PTX3, FGF2 and TNFAIP6
- Pasteur effect in leukocyte energy metabolism of patients with mild, moderate, and severe COVID-19
- Thiol-disulfide homeostasis and ischemia-modified albumin in patients with sepsis
- Myotonic dystrophy type 1 and oxidative imbalance: evaluation of ischemia-modified albumin and oxidant stress
- Antioxidant and alpha-glucosidase inhibitory activities of flavonoids isolated from fermented leaves of Camellia chrysantha (Hu) Tuyama
- Examination of the apelin signaling pathway in acetaminophen-induced hepatotoxicity in rats
- Integrating network pharmacology, in silico molecular docking and experimental validation to explain the anticancer, apoptotic, and anti-metastatic effects of cosmosiin natural product against human lung carcinoma
- Validation of Protein A chromatography: orthogonal method with size exclusion chromatography validation for mAb titer analysis
- The evaluation of the efficiency of Atellica UAS800 in detecting pathogens (rod, cocci) causing urinary tract infection
- Case Report
- Exploring inherited vitamin B responsive disorders in the Moroccan population: cutting-edge diagnosis via GC-MS profiling
- Letter to the Editor
- Letter to the Editor: “Gene mining, recombinant expression and enzymatic characterization of N-acetylglucosamine deacetylase”


