Abstract
[C15H6Cl6O4], monoclinic, Cc (no. 9), a = 12.0980(11) Å, b = 10.0628(9) Å, c = 15.1518(14) Å, β = 104.231(1)°, V = 1788.0(3) Å3, Z = 4, Rgt(F) = 0.0214, wRref(F2) = 0.0568, T = 296(2) K.
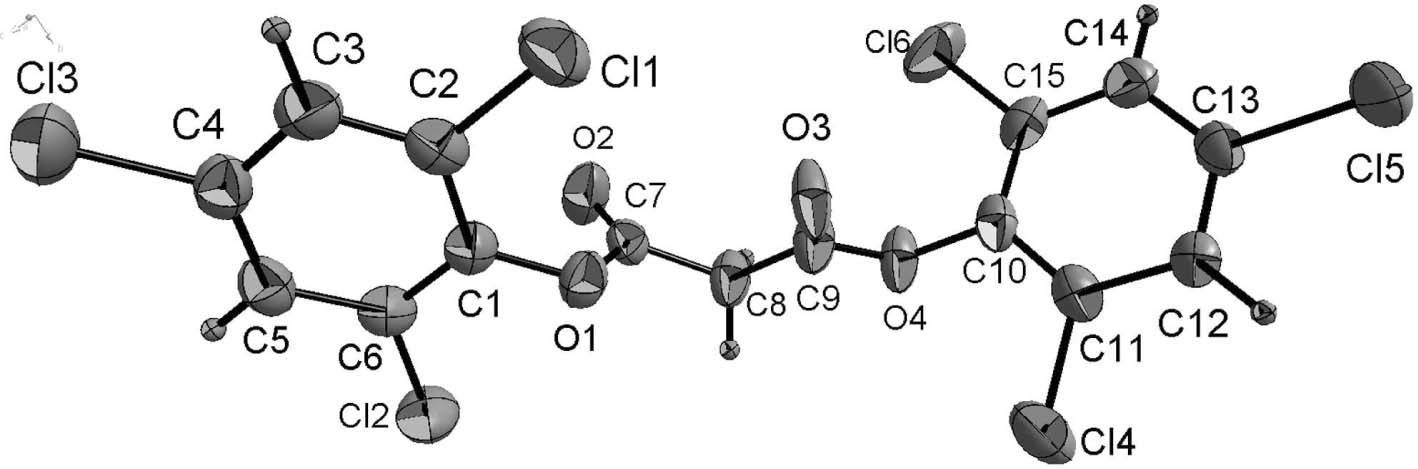
The crystal structure is shown in the figure. Tables 1 and 2 contain details on crystal structure and measurement conditions and a list of the atoms including atomic coordinates and displacement parameters.
Data collection and handling.
| Crystal: | Colorless block |
| Size: | 0.16 × 0.14 × 0.12 mm |
| Wavelength: | Mo Kα radiation (0.71073 Å) |
| μ: | 1.0 mm−1 |
| Diffractometer, scan mode: | Bruker Apex-II, φ and ω-scans |
| θmax, completeness: | 25.5°, >99% |
| N(hkl)measured, N(hkl)unique, Rint: | 6761, 3023, 0.016 |
| Criterion for Iobs, N(hkl)gt: | Iobs > 2 σ(Iobs), 2952 |
| N(param)refined: | 227 |
| Programs: | Bruker programs [1], SHELX [2] |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2).
| Atom | x | y | z | Uiso*/Ueq |
|---|---|---|---|---|
| C1 | 0.4028(2) | 0.7439(3) | 0.7211(2) | 0.0404(6) |
| C2 | 0.3300(2) | 0.6358(3) | 0.7001(2) | 0.0447(6) |
| C3 | 0.3337(3) | 0.5341(3) | 0.7616(2) | 0.0492(7) |
| H3 | 0.2842 | 0.4621 | 0.7478 | 0.059* |
| C4 | 0.4131(3) | 0.5419(3) | 0.8445(2) | 0.0488(7) |
| C5 | 0.4875(3) | 0.6471(3) | 0.8675(2) | 0.0461(7) |
| H5 | 0.5403 | 0.6504 | 0.9236 | 0.055* |
| C6 | 0.4809(2) | 0.7476(3) | 0.8043(2) | 0.0418(6) |
| C7 | 0.4609(2) | 0.8445(3) | 0.6012(2) | 0.0409(6) |
| C8 | 0.4453(3) | 0.9649(3) | 0.5412(2) | 0.0512(7) |
| H8A | 0.4608 | 1.0433 | 0.5794 | 0.061* |
| H8B | 0.5012 | 0.9621 | 0.5049 | 0.061* |
| C9 | 0.3289(2) | 0.9783(3) | 0.4783(2) | 0.0507(8) |
| C10 | 0.2370(2) | 1.0893(3) | 0.3437(2) | 0.0424(6) |
| C11 | 0.1700(2) | 1.1991(3) | 0.3461(2) | 0.0425(6) |
| C12 | 0.0793(2) | 1.2288(3) | 0.2735(2) | 0.0417(6) |
| H12 | 0.0345 | 1.3036 | 0.2747 | 0.050* |
| C13 | 0.0568(2) | 1.1451(3) | 0.19938(19) | 0.0410(6) |
| C14 | 0.1216(3) | 1.0341(3) | 0.1951(2) | 0.0463(7) |
| H14 | 0.1049 | 0.9785 | 0.1445 | 0.056* |
| C15 | 0.2121(2) | 1.0079(3) | 0.2683(2) | 0.0475(7) |
| O1 | 0.39339(17) | 0.8502(2) | 0.66100(15) | 0.0474(5) |
| O2 | 0.5245(2) | 0.7569(3) | 0.59956(17) | 0.0664(7) |
| O3 | 0.2442(2) | 0.9227(4) | 0.4817(2) | 0.0933(11) |
| O4 | 0.33483(17) | 1.0684(2) | 0.41368(16) | 0.0538(6) |
| Cl1 | 0.23312(8) | 0.62950(10) | 0.59519(6) | 0.0650(2) |
| Cl2 | 0.57314(7) | 0.88156(8) | 0.83113(6) | 0.0610(2) |
| Cl3 | 0.42004(10) | 0.41507(9) | 0.92305(7) | 0.0733(3) |
| Cl4 | 0.19913(7) | 1.30229(10) | 0.44010(6) | 0.0663(3) |
| Cl5 | −0.05775(7) | 1.18000(8) | 0.10804(6) | 0.0606(2) |
| Cl6 | 0.29540(9) | 0.86970(9) | 0.26502(10) | 0.0843(3) |
Source of materials
Pyrimidinones are usually synthesized by using 2-aminopyridine and bis(2,4,6-trichlorophenyl)malonate. Based on the literature synthesis, we have improved the route. The synthesis process of bis(2,4,6-trichlorophenyl) malonate was carried out using malonic acid as starting material. Malonic acid (4 g, 38.46 mmol), oxalyl chloride (5.38 mL, 57.69 mmol) were added in DCM (50 mL); the mixtrue was stirred well for 1 h at room temperature, then we added 1 drop of DMF, and continued stirring until the mixture became clear. 2,4,6-trichlorophenol (11.3 g, 57.69 mmol) was added, the reaction solution stirred at room temperature overnight. The product was concentrated under reduced pressure. Finally, methanol (20 mL) was added until a white solid precipitated then the resulting solution was filtered under reduced pressure. The raw product (10.5 g) was recrystallied from dichloromethane. m.p: 149–152 °C. Elemental Anal. Calcd. (%) for C15H6Cl6O4 (462.90): C, 38.92; H, 1.37. Found (%): C, 38.18; H, 1.50.
Experimental details
All H atoms were included in calculated positions and refined as riding atoms, with C—H = 0.90−0.97 Å with Uiso(H) = 1.5 Ueq(C) for methyl H atoms and 1.2 Ueq(C) for all other H atoms. The Flack-Parsons parameter is 0.039(18) based on 1283 quotients [2].
Comment
Pyrimidine and pyrimidopyrimidine derivatives is an important medicine [3], and its synthesis is usually performed by using 2-aminopyridine and bis(2,4,6-trichlorophenyl)malonate [4], [5].
Nowadays, many pyrimidine and pyrimidopyrimidine derivatives are of interest in the search for new pharmaceutical candidates because they may show distinguished pharmaceutical properties, such as: antiviral, antibacterial, anti-HIV, antiallergic, and antitumoral activities [6]. Pyrimidines have a prominent pharmacophore prevailing in many heterocyclic natural products. During the past two decades, several pyrimidine derivatives were found to have widespread clinical applications including chemotherapy of AIDS.
Herein we report the synthesis and crystal structure of the intermediate of the pyrimidone synthesis [7]: bis(2,4,6-trichlorophenyl) malonate. In the molecule of the title compound (Fig.), bond lengths and angles are in the expected ranges for malonates [8, 9] . Two aryl rings were bound to the both sides of malonic acid group. The dihedral angle between the benzene rings is 67.8°. Further, the dihedral angle between the central CH2—C(O)—O segment and the phenyl ring is 83.8°.
Acknowledgement
X-ray data were collected at Instrumental Analysis Center Nanchang Hangkong University, Nanchang, 330063, People’s Republic of China.
This research has been supported by the China National Key R&D Program during the 13th Five-year Plan Period (Grant No.2017YFD0301604) the National Natural Science Foundation of China (Grant No. 21562022), the Research Foundation of Educational Department of Jiangxi Province [GJJ170274,160382] and the Research Foundation of company [9131206709].
References
1. Bruker. APEX2, SAINT and SADABS. Brucker AXS Inc., Madison, WI, USA (2009).Search in Google Scholar
2. Sheldrick, G. M.: A short history of SHELX. Acta Crystallogr. A64 (2008) 112–122.10.1107/S0108767307043930Search in Google Scholar
3. Huang, Q. C.; Liu, J. Y.: Progress in pyrimidine diketone TRPA1 inhibitors. Biotech World 6 (2015) 10.Search in Google Scholar
4. Marshall, A. J.; Lill, C. L.; Chao, M.; Kolekar, S. V.; Lee, W. J.; Marshall, E. S.; Baguley, B. C.; Shepherd, P. R.; Denny, W. A.; Flanagan, J. U.: Exploring the isoform selectivity of TGX-221 related pyrido[1,2-a]pyrimidinone-based class IAPI 3-kinase inhibitors: synthesis, biological evaluation and molecular modelling. Bioorg. Med. Chem. 23 (2015) 3796–3808.10.1016/j.bmc.2015.03.073Search in Google Scholar
5. Park, D. S.; Jo, E.; Choi, J.; Lee, M.; Kim, S.; Kim, H. Y.; Nam, J.; Ahn, S.; Hwang, J. Y.; Windisch, M. P.: Characterization and structure-activity relationship study of iminodipyridinopyrimidines as novel hepatitis C virus inhibitor. Eur. J. Med. Chem. 140 (2017) 65–73.10.1016/j.ejmech.2017.09.010Search in Google Scholar
6. Gebauer, M. G.; Mckinlay, C.; Gready, J. E.: Synthesis of quaternised 2-aminopyrimido[4,5-d]pyrimidin-4(3H)-ones and their biological activity with dihydrofolate reductase. Eur. J. Med. Chem. 38 (2003) 719–728.10.1016/S0223-5234(03)00140-5Search in Google Scholar
7. Guellue, M.; Dincsoenmez, A.; Oezyavs, O.: Facile synthesis of novel pyrimido[1,2-a]pyrimidin-4-ones from highly reactive malonates. Eur. J. Org. Chem. 41 (2010) 2113–2120.10.1002/chin.201036161Search in Google Scholar
8. Curt, W.; Jeff, B.; Karl, A. B.; Colin, H. L.: Bis(2,4,6-trichlorophenyl) 2-ethylmalonate. Acta Crystallogr. E60 (2004) o137–o138.10.1107/S1600536803028812Search in Google Scholar
9. Devarajegowda, H. C.; Suchetan, P. A.; Sreenivasa, S.; Srinivasa, H. T.; Palakshamurthy, B. S.: Crystal structure of bis(4-methoxyphenyl) malonate. Acta Crystallogr. E71 (2015) o330.10.1107/S2056989015006891Search in Google Scholar PubMed PubMed Central
©2019 Shi Yun-Lian et al., published by De Gruyter, Berlin/Boston
This work is licensed under the Creative Commons Attribution 4.0 Public License.
Articles in the same Issue
- Cover and Frontmatter
- Crystal structure of 1H-indole-5-carboxylic acid – 4,4′-bipyridine (2/1), C14H11N2O2
- Crystal structure of ethyl 2-amino-4-(4-ethoxyphenyl)-5-oxo-4H,5H-pyrano[3,2-c] chromene-3-carboxylate, C23H21NO6
- Crystal structure of ethyl 2-amino-4-(4-bromothiophen-2-yl)-5-oxo-5,6,7,8-tetrahydro-4H-chromene-3-carboxylate, C16H16BrNO4S
- The crystal structure of 6-amino-2-methyl-8-(4-(methylthio)phenyl)-2,3,8,8a-tetrahydroisoquinoline-5,7,7(1H)-tricarbonitrile – ethanol (1/1), C20H19N5S
- Crystal structure of 6-amino-8-(4-isopropylphenyl)-2-methyl-2,3,8,8a-tetrahydroisoquinoline-5,7,7(1H)-tricarbonitrile-ethanol (1/1), C24H29N5O
- Crystal structure of 1,1′-(ethane-1,2-diyl)bis(3-ethyl-1H-imidazol-3-ium)bis(hexafluorido phosphate), C12H20F12N4P2
- Crystal structure of dimethyl (3aS,6R,6aS,7S)-2-pivaloyl-2,3-dihydro-1H,6H,7H-3a,6:7,9a-diepoxybenzo[de]isoquinoline-3a1,6a-dicarboxylate, C21H25NO8
- Crystal structure of methyl 4-(4-bromothiophen-2-yl)-2,7,7-trimethyl-5-oxo-1,4,5,6,7,8-hexahydroquinoline-3-carboxylate, C18H20BrNO3S
- Hydrothermal synthesis and crystal structure of catena-poly[bis(4-((pyridin-4-ylmethyl)amino)benzoato-κ3N:O,O′)zinc(II) – 1,2-di(pyridin-4-yl)ethene – water (1/1/1), C38H34N6O5Zn
- The crystal structure of 1,2-dimethyl-3,4-dinitrobenzene, C8H8N2O4
- Synthesis and crystal structure of trans-tetraaqua-bis(3-(((7-hydroxy-3-(4-hydroxy-3-sulfonatophenyl)-4-oxo-4H-chromen-8-yl)methyl)ammonio)propanoato-κO)zinc(II) tetrahydrate, C38H48N2O26S2Zn
- Crystal structure of diaqua-bis(μ2-6-chloropyridin-2-olato-κ3N,O:O)-tetrakis(chloropyridin-2-olato-κ1O)-bis(penanthroline-κ2N,N′)diterbium(III), C54H38Cl6Tb2N10O8
- Crystal structure of oxidobis(piperidine-1-carbodithioato-κ2S,S′)vanadium(IV), C12H20N2OS4V
- Crystal structure of 2-((tert-butyldimethylsilyl)oxy)-5-methylisophthalaldehyde, C15H22O3Si
- Crystal structure of catena-poly[tetraiodido-(μ2-1,4-bis(2-methyl-1H-imidazol-1-yl)benzene-κ2N:N′)dimercury(II)], C14H14Hg2I4N4
- Crystal structure of tetrakis(n-butyl)-(μ2-1,2-bis(2-oxidobenzoyl)hydrazine-1,2-diido-κ6N,O,O′:N′,O′′,O′′′)ditin(IV), C30H44N2O4Sn2
- Crystal structure of ethyl 2-amino-4-(4-hydroxy-3-methoxyphenyl)-7-methyl-5-oxo-4H,5H-pyrano-[4,3-b]pyran-3-carboxylate, C19H19NO7
- Crystal structure of 3-aminopyrazine-2-carbohydrazide, C5H7N5O
- Crystal structure of ethanol-bis(N-((5-(ethoxycarbonyl)-3,4-dimethyl-1H-pyrrol-2-yl)methylene)benzohydrazonato-κ2N,O)copper(II), C36H42N6O7Cu
- Crystal structure of 3-methyl-2-oxo-2H-chromen-7-yl propionate, C13H12O4
- Crystal structure of 2-(dimethylamino)ethyl 4-aminobenzoate, C11H16N2O2
- Crystal structure of 3-(benzo[d]thiazol-2-ylamino)isobenzofuran-1(3H)-one, C15H10N2O2S
- Crystal structure of 3-((1H-benzo[d]imidazol-2-yl)amino)-2-(1H-benzo[d]imidazol-2-yl)isoindolin-1-one, C22H16N6O
- Crystal structure of (2,2′-bipyridine-κ2N,N′)bis(4-(dimethylamino)phenyldiphenylphosphane-κP)copper(I) tetrafluoroborate, C50H48BCuF4N4P2
- Crystal structure of citric acid–acetonitrile (1/1), C8H11NO7
- Crystal structure of diethyl 2-(4-methoxyphenyl)-1-phenyl-1,2-dihydropyridine-3,5-dicarboxylate, C24H25NO5
- The crystal structure of poly[triaqua-bis(μ3-2,5-dihydroxyterephthalato-κ4O,O′:O′′:O′′′)-(μ4-oxalato-κ4O,O′:O′′,O′′′)cerium(III)], C9H10CeO11
- Crystal structure of 1-(5-(anthracen-9-yl)-3-(4-hydroxyphenyl)-4,5-dihydro-1H-pyrazol-1-yl)propan-1-one, C26H22N2O2
- Synthesis and crystal structure of 5-(8-(((2-carboxyethyl)ammonio)methyl)-7-hydroxy-4-oxo-4H-chromen-3-yl)-2-hydroxybenzenesulfonate trihydrate, C19H23NO12S
- Crystal structure of rac-trans-6,6′-((cyclohexane-1,2-diylbis(azanylylidene))bis(methanylylidene))bis(2-bromophenolato-κ4N,N′,O,O′)-bis(methanol)cobalt(III) chloride, C22H25Br2Co8N2O4Cl
- Crystal structure of 1-((R)-(2′-(dimethylamino)-[1,1′-binaphthalen]-2-yl))-3-((S)-2-hydroxy-1-phenylethyl)thiourea, C31H29N3OS
- Crystal structure and photochemical property of 1,8-bis(p-tolylthio)pyrene, C30H22S2
- Crystal structure of 2-(2-(2-amino-6-chloro-9H-purin-9-yl)ethyl)propane-1,3-diyl diacetate, C14H18ClN5O4
- Crystal structure of ethyl 5-amino-1-(pyridin-2-yl)-1H-pyrazole-4-carboxylate, C11H12N4O2
- Crystal structure of trichloro-(4-chloro-2,6-bis(diphenylmethyl)-N-((pyridin-2-yl)methylene)aniline)-aluminum dichloromethane solvate, C39H31AlCl6N2
- Bis(ethanol-κO)-bis(6-aminopicolinato-κ2N,O)magnesium(II), C16H22O6N4Mg
- Crystal structure of catena-poly[aqua-(μ2-1,7-dicarba-closo-dodecaborane-1,7-dicarboxylato-κ2O:O′)-(1,10-phenanthrolin-κ2N,N′)copper(II)], C16H20B10CuN2O5
- Crystal structure of (1,2-dicarba-closo-dodecaborane-1,2-dithiolato κ2S,S′)-bis(1,10-phenanthroline κ2N,N′)zinc(II), C26H26B10Zn4S2
- Crystal structure of diaqua-bis(1,10-phenanthroline-κ2N,N′)-bis(1,7-dicarba-closo-dodecaborane-1,7-dicarboxylato-κ3O,O′:O′′) dicobalt(II) — ethanol (1/1), C34H46B20Co2N4O11
- Crystal structure of ((5,5′-dimethoxy-2,2′-(1,2-phenylenebis(nitrilomethylidyne)))diphenolato-κ4O,N,O′,N′)copper(II), C22H18N2CuO4
- Crystal structure of 1-(5-bromo-2-(4-methoxyphenyl)-1H-indol-7-yl)ethan-1-ol, C17H14BrNO2
- Crystal structure of (E)-2-(((6-bromopyridin-2-yl)methylene)amino)-3′,6′-bis(diethylamino)spiro[isoindoline-1,9′-xanthen]-3-one, C34H34N5O2Br
- Crystal structure of (Z)-2-((adamantan-1-ylimino)methyl)-5-methoxyphenol, C18H23NO2
- Crystal structure of bis((E)-2-ethoxy-6-(((2-hydroxyethyl)imino)methyl)phenolato-κ2N,O)copper(II), C22H28N2CuO6
- Crystal structure of 2,3-diphenyl-5,6-bis(4-methoxyphenyl)pyrazine, C30H24N2O2
- Crystal structure of dichlorido bis[1-((2,4-dimethyl-1H-imidazol-1-yl)methyl)-1H-benzo[d][1,2,3]triazole-κN]cadmium(II), Cd(C12H13N5)2Cl2
- The crystal structure of 1,5-di(naphthalen-2-yl)-3-(pyridin-2-yl)pentane-1,5-dione, C30H23NO2
- The crystal structure of 2-((3-methylthiophen-2-yl)methylene)malononitrile, C9H6N2S
- The crystal structure of 1,4-dinitroso-2,3,5,6-tetraacetoxy-piperazine, C12H16N4O10
- Crystal structure of bis(2,4,6-trichlorophenyl) malonate, C15H6Cl6O4
- The crystal structure of trans-dichlorido-bis(pyridine-2-carboxylato-κ2N,O)platinum(IV), C12H8Cl2N2O4Pt
- Crystal structure of 3-nitroquinoline 1-oxide, C9H6N2O3
- Crystal structure of 2-(piperidin-1-ium-4-yl)-1H-benzo[d]imidazol-3-ium dichloride dihydrate, C12H21Cl2N3O2
- Crystal structure of (4S,4aS,6aR,6bR,12aS,12bR,14aS,14bR)-3,3,6a,6b,9,9,12a-heptamethyloctadecahydro-1H,3H-4,14b-ethanophenanthro[1,2-h]isochromene, C30H50O
- Crystal structure of (E)-4-((2-fluoro-3-(trifluoromethyl)benzylidene)amino)-3-methyl-1H-1,2,4-triazole-5(4H)-thione, C11H8F4N4S
- Crystal structure of 5-(4-fluorophenyl)-4-methyl-2,4-dihydro-3H-1,2,4-triazole-3-thione, C9H8FN3S
- Crystal structure of catena-poly[(1-(4-fluorophenyl)-N–(5-((trimethylstannyl)thio)-1,3,4-thiadiazol-2-yl)methanimine], (C12H14FN3S2Sn)n
- The crystal structure of 4-(methoxycarbonyl)benzoic acid, C9H8O4
- The crystal structure of N,N′-(6-(thiophen-2-yl)-1,3,5-triazine-2,4-diyl)bis(2-methylpropane-2-sulfonamide) – ethyl acetate(2/1), C34H54N10O6S6
- Crystal structure of N′-(1-(2-hydroxyphenyl)ethylidene)-5-methyl-1-phenyl-1H-1,2,3-triazole-4-carbohydrazide, C18H17N5O2
- Crystal structure of 3-(4-methoxyphenyl)-1-phenylprop-2-yn-1-one, C16H12O2
- Crystal structure of N′-(1-(benzofuran-2-yl)ethylidene)-2-cyanoacetohydrazide, C13H11N3O2
- Crystal structure of hexa-μ2-chlorido-μ4-oxido-tetrakis(1-vinyl-1H-imidazole-κN)tetracopper(II), C20H24Cu4Cl6N8O
- Crystal structure of N′-((1E,2E)-4-(7-methoxy-2-oxo-2H-chromen-8-yl)-2-methylbut-2-en-1-ylidene)-4-methylbenzenesulfonohydrazide, C22H22O5N2S
- Crystal structure of 2-acetyl pyrene, C18H12O
Articles in the same Issue
- Cover and Frontmatter
- Crystal structure of 1H-indole-5-carboxylic acid – 4,4′-bipyridine (2/1), C14H11N2O2
- Crystal structure of ethyl 2-amino-4-(4-ethoxyphenyl)-5-oxo-4H,5H-pyrano[3,2-c] chromene-3-carboxylate, C23H21NO6
- Crystal structure of ethyl 2-amino-4-(4-bromothiophen-2-yl)-5-oxo-5,6,7,8-tetrahydro-4H-chromene-3-carboxylate, C16H16BrNO4S
- The crystal structure of 6-amino-2-methyl-8-(4-(methylthio)phenyl)-2,3,8,8a-tetrahydroisoquinoline-5,7,7(1H)-tricarbonitrile – ethanol (1/1), C20H19N5S
- Crystal structure of 6-amino-8-(4-isopropylphenyl)-2-methyl-2,3,8,8a-tetrahydroisoquinoline-5,7,7(1H)-tricarbonitrile-ethanol (1/1), C24H29N5O
- Crystal structure of 1,1′-(ethane-1,2-diyl)bis(3-ethyl-1H-imidazol-3-ium)bis(hexafluorido phosphate), C12H20F12N4P2
- Crystal structure of dimethyl (3aS,6R,6aS,7S)-2-pivaloyl-2,3-dihydro-1H,6H,7H-3a,6:7,9a-diepoxybenzo[de]isoquinoline-3a1,6a-dicarboxylate, C21H25NO8
- Crystal structure of methyl 4-(4-bromothiophen-2-yl)-2,7,7-trimethyl-5-oxo-1,4,5,6,7,8-hexahydroquinoline-3-carboxylate, C18H20BrNO3S
- Hydrothermal synthesis and crystal structure of catena-poly[bis(4-((pyridin-4-ylmethyl)amino)benzoato-κ3N:O,O′)zinc(II) – 1,2-di(pyridin-4-yl)ethene – water (1/1/1), C38H34N6O5Zn
- The crystal structure of 1,2-dimethyl-3,4-dinitrobenzene, C8H8N2O4
- Synthesis and crystal structure of trans-tetraaqua-bis(3-(((7-hydroxy-3-(4-hydroxy-3-sulfonatophenyl)-4-oxo-4H-chromen-8-yl)methyl)ammonio)propanoato-κO)zinc(II) tetrahydrate, C38H48N2O26S2Zn
- Crystal structure of diaqua-bis(μ2-6-chloropyridin-2-olato-κ3N,O:O)-tetrakis(chloropyridin-2-olato-κ1O)-bis(penanthroline-κ2N,N′)diterbium(III), C54H38Cl6Tb2N10O8
- Crystal structure of oxidobis(piperidine-1-carbodithioato-κ2S,S′)vanadium(IV), C12H20N2OS4V
- Crystal structure of 2-((tert-butyldimethylsilyl)oxy)-5-methylisophthalaldehyde, C15H22O3Si
- Crystal structure of catena-poly[tetraiodido-(μ2-1,4-bis(2-methyl-1H-imidazol-1-yl)benzene-κ2N:N′)dimercury(II)], C14H14Hg2I4N4
- Crystal structure of tetrakis(n-butyl)-(μ2-1,2-bis(2-oxidobenzoyl)hydrazine-1,2-diido-κ6N,O,O′:N′,O′′,O′′′)ditin(IV), C30H44N2O4Sn2
- Crystal structure of ethyl 2-amino-4-(4-hydroxy-3-methoxyphenyl)-7-methyl-5-oxo-4H,5H-pyrano-[4,3-b]pyran-3-carboxylate, C19H19NO7
- Crystal structure of 3-aminopyrazine-2-carbohydrazide, C5H7N5O
- Crystal structure of ethanol-bis(N-((5-(ethoxycarbonyl)-3,4-dimethyl-1H-pyrrol-2-yl)methylene)benzohydrazonato-κ2N,O)copper(II), C36H42N6O7Cu
- Crystal structure of 3-methyl-2-oxo-2H-chromen-7-yl propionate, C13H12O4
- Crystal structure of 2-(dimethylamino)ethyl 4-aminobenzoate, C11H16N2O2
- Crystal structure of 3-(benzo[d]thiazol-2-ylamino)isobenzofuran-1(3H)-one, C15H10N2O2S
- Crystal structure of 3-((1H-benzo[d]imidazol-2-yl)amino)-2-(1H-benzo[d]imidazol-2-yl)isoindolin-1-one, C22H16N6O
- Crystal structure of (2,2′-bipyridine-κ2N,N′)bis(4-(dimethylamino)phenyldiphenylphosphane-κP)copper(I) tetrafluoroborate, C50H48BCuF4N4P2
- Crystal structure of citric acid–acetonitrile (1/1), C8H11NO7
- Crystal structure of diethyl 2-(4-methoxyphenyl)-1-phenyl-1,2-dihydropyridine-3,5-dicarboxylate, C24H25NO5
- The crystal structure of poly[triaqua-bis(μ3-2,5-dihydroxyterephthalato-κ4O,O′:O′′:O′′′)-(μ4-oxalato-κ4O,O′:O′′,O′′′)cerium(III)], C9H10CeO11
- Crystal structure of 1-(5-(anthracen-9-yl)-3-(4-hydroxyphenyl)-4,5-dihydro-1H-pyrazol-1-yl)propan-1-one, C26H22N2O2
- Synthesis and crystal structure of 5-(8-(((2-carboxyethyl)ammonio)methyl)-7-hydroxy-4-oxo-4H-chromen-3-yl)-2-hydroxybenzenesulfonate trihydrate, C19H23NO12S
- Crystal structure of rac-trans-6,6′-((cyclohexane-1,2-diylbis(azanylylidene))bis(methanylylidene))bis(2-bromophenolato-κ4N,N′,O,O′)-bis(methanol)cobalt(III) chloride, C22H25Br2Co8N2O4Cl
- Crystal structure of 1-((R)-(2′-(dimethylamino)-[1,1′-binaphthalen]-2-yl))-3-((S)-2-hydroxy-1-phenylethyl)thiourea, C31H29N3OS
- Crystal structure and photochemical property of 1,8-bis(p-tolylthio)pyrene, C30H22S2
- Crystal structure of 2-(2-(2-amino-6-chloro-9H-purin-9-yl)ethyl)propane-1,3-diyl diacetate, C14H18ClN5O4
- Crystal structure of ethyl 5-amino-1-(pyridin-2-yl)-1H-pyrazole-4-carboxylate, C11H12N4O2
- Crystal structure of trichloro-(4-chloro-2,6-bis(diphenylmethyl)-N-((pyridin-2-yl)methylene)aniline)-aluminum dichloromethane solvate, C39H31AlCl6N2
- Bis(ethanol-κO)-bis(6-aminopicolinato-κ2N,O)magnesium(II), C16H22O6N4Mg
- Crystal structure of catena-poly[aqua-(μ2-1,7-dicarba-closo-dodecaborane-1,7-dicarboxylato-κ2O:O′)-(1,10-phenanthrolin-κ2N,N′)copper(II)], C16H20B10CuN2O5
- Crystal structure of (1,2-dicarba-closo-dodecaborane-1,2-dithiolato κ2S,S′)-bis(1,10-phenanthroline κ2N,N′)zinc(II), C26H26B10Zn4S2
- Crystal structure of diaqua-bis(1,10-phenanthroline-κ2N,N′)-bis(1,7-dicarba-closo-dodecaborane-1,7-dicarboxylato-κ3O,O′:O′′) dicobalt(II) — ethanol (1/1), C34H46B20Co2N4O11
- Crystal structure of ((5,5′-dimethoxy-2,2′-(1,2-phenylenebis(nitrilomethylidyne)))diphenolato-κ4O,N,O′,N′)copper(II), C22H18N2CuO4
- Crystal structure of 1-(5-bromo-2-(4-methoxyphenyl)-1H-indol-7-yl)ethan-1-ol, C17H14BrNO2
- Crystal structure of (E)-2-(((6-bromopyridin-2-yl)methylene)amino)-3′,6′-bis(diethylamino)spiro[isoindoline-1,9′-xanthen]-3-one, C34H34N5O2Br
- Crystal structure of (Z)-2-((adamantan-1-ylimino)methyl)-5-methoxyphenol, C18H23NO2
- Crystal structure of bis((E)-2-ethoxy-6-(((2-hydroxyethyl)imino)methyl)phenolato-κ2N,O)copper(II), C22H28N2CuO6
- Crystal structure of 2,3-diphenyl-5,6-bis(4-methoxyphenyl)pyrazine, C30H24N2O2
- Crystal structure of dichlorido bis[1-((2,4-dimethyl-1H-imidazol-1-yl)methyl)-1H-benzo[d][1,2,3]triazole-κN]cadmium(II), Cd(C12H13N5)2Cl2
- The crystal structure of 1,5-di(naphthalen-2-yl)-3-(pyridin-2-yl)pentane-1,5-dione, C30H23NO2
- The crystal structure of 2-((3-methylthiophen-2-yl)methylene)malononitrile, C9H6N2S
- The crystal structure of 1,4-dinitroso-2,3,5,6-tetraacetoxy-piperazine, C12H16N4O10
- Crystal structure of bis(2,4,6-trichlorophenyl) malonate, C15H6Cl6O4
- The crystal structure of trans-dichlorido-bis(pyridine-2-carboxylato-κ2N,O)platinum(IV), C12H8Cl2N2O4Pt
- Crystal structure of 3-nitroquinoline 1-oxide, C9H6N2O3
- Crystal structure of 2-(piperidin-1-ium-4-yl)-1H-benzo[d]imidazol-3-ium dichloride dihydrate, C12H21Cl2N3O2
- Crystal structure of (4S,4aS,6aR,6bR,12aS,12bR,14aS,14bR)-3,3,6a,6b,9,9,12a-heptamethyloctadecahydro-1H,3H-4,14b-ethanophenanthro[1,2-h]isochromene, C30H50O
- Crystal structure of (E)-4-((2-fluoro-3-(trifluoromethyl)benzylidene)amino)-3-methyl-1H-1,2,4-triazole-5(4H)-thione, C11H8F4N4S
- Crystal structure of 5-(4-fluorophenyl)-4-methyl-2,4-dihydro-3H-1,2,4-triazole-3-thione, C9H8FN3S
- Crystal structure of catena-poly[(1-(4-fluorophenyl)-N–(5-((trimethylstannyl)thio)-1,3,4-thiadiazol-2-yl)methanimine], (C12H14FN3S2Sn)n
- The crystal structure of 4-(methoxycarbonyl)benzoic acid, C9H8O4
- The crystal structure of N,N′-(6-(thiophen-2-yl)-1,3,5-triazine-2,4-diyl)bis(2-methylpropane-2-sulfonamide) – ethyl acetate(2/1), C34H54N10O6S6
- Crystal structure of N′-(1-(2-hydroxyphenyl)ethylidene)-5-methyl-1-phenyl-1H-1,2,3-triazole-4-carbohydrazide, C18H17N5O2
- Crystal structure of 3-(4-methoxyphenyl)-1-phenylprop-2-yn-1-one, C16H12O2
- Crystal structure of N′-(1-(benzofuran-2-yl)ethylidene)-2-cyanoacetohydrazide, C13H11N3O2
- Crystal structure of hexa-μ2-chlorido-μ4-oxido-tetrakis(1-vinyl-1H-imidazole-κN)tetracopper(II), C20H24Cu4Cl6N8O
- Crystal structure of N′-((1E,2E)-4-(7-methoxy-2-oxo-2H-chromen-8-yl)-2-methylbut-2-en-1-ylidene)-4-methylbenzenesulfonohydrazide, C22H22O5N2S
- Crystal structure of 2-acetyl pyrene, C18H12O


