Abstract
C15H10BrClINO2, monoclinic, P21/n (no. 14), a = 5.0083(2) Å, b = 9.7548(3) Å, c = 31.9640(10) Å, β = 92.576(2)°, V = 1560.02(9) Å3, Z = 4, Rgt(F) = 0.0416, wRref(F2) = 0.1059, T = 173 K.
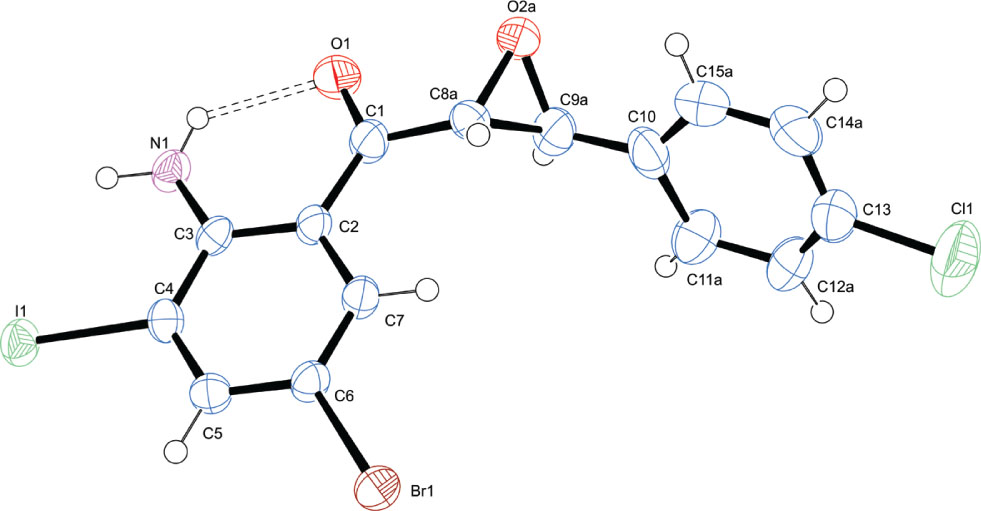
The molecular structure is shown in the figure. Table 1 contains crystallographic data and Table 2 contains the list of the atoms including atomic coordinates and displacement parameters.
Data collection and handling.
| Crystal: | Yellow block |
| Size: | 0.62 × 0.48 × 0.30 mm |
| Wavelength: | Mo Kα radiation (0.71073 Å) |
| μ: | 4.79 mm−1 |
| Diffractometer, scan mode: | Bruker D8 Venture Photon, ω |
| θmax, completeness: | 28.0°, >99% |
| N(hkl)measured, N(hkl)unique, Rint: | 31032, 3781, 0.058 |
| Criterion for Iobs, N(hkl)gt: | Iobs > 2 σ(Iobs), 3303 |
| N(param)refined: | 263 |
| Programs: | Bruker [1], WinGX/ORTEP [2], SHELX [3], PLATON [4] |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2).
| Atom | x | y | z | Uiso*/Ueq |
|---|---|---|---|---|
| C1 | 0.6931(11) | 0.6775(5) | 0.67719(17) | 0.0408(12) |
| C2 | 0.4898(9) | 0.5747(4) | 0.66531(15) | 0.0312(9) |
| C3 | 0.4385(9) | 0.4607(4) | 0.69161(14) | 0.0283(9) |
| C4 | 0.2367(9) | 0.3685(5) | 0.67779(14) | 0.0305(9) |
| C5 | 0.0928(9) | 0.3865(5) | 0.64076(14) | 0.0326(9) |
| H5 | −0.040438 | 0.32449 | 0.632563 | 0.039* |
| C6 | 0.1472(9) | 0.4984(5) | 0.61545(14) | 0.0314(9) |
| C7 | 0.3424(9) | 0.5906(5) | 0.62729(14) | 0.0322(9) |
| H7 | 0.377616 | 0.664382 | 0.609963 | 0.039* |
| C8Aa | 0.7868(15) | 0.7760(8) | 0.6414(3) | 0.037(2) |
| H8Aa | 0.793134 | 0.738745 | 0.613033 | 0.044* |
| C9Aa | 0.722(2) | 0.9235(10) | 0.6460(3) | 0.043(2) |
| H9Aa | 0.615447 | 0.952881 | 0.669366 | 0.052* |
| O2Aa | 0.9873(14) | 0.8721(7) | 0.6546(3) | 0.060(3) |
| C8Bb | 0.624(2) | 0.8232(11) | 0.6627(4) | 0.029(3) |
| H8Bb | 0.435513 | 0.84887 | 0.658135 | 0.035* |
| C9Bb | 0.821(3) | 0.8790(16) | 0.6349(5) | 0.034(3) |
| H9Bb | 0.969485 | 0.822565 | 0.625842 | 0.04* |
| O2Bb | 0.809(2) | 0.9229(10) | 0.6780(3) | 0.047(3) |
| C10 | 0.7036(15) | 1.0025(7) | 0.6060(2) | 0.063(2) |
| C11Ac | 0.445(3) | 1.0899(16) | 0.6043(5) | 0.054(3) |
| H11Ac | 0.32292 | 1.086396 | 0.625353 | 0.065* |
| C12Ac | 0.407(3) | 1.1752(16) | 0.5689(5) | 0.054(4) |
| H12Ac | 0.257531 | 1.231644 | 0.56595 | 0.065* |
| C11Bc | 0.591(3) | 1.1064(14) | 0.6093(4) | 0.042(3) |
| H11Bc | 0.537964 | 1.130583 | 0.63589 | 0.05* |
| C12Bc | 0.530(3) | 1.1945(15) | 0.5773(5) | 0.043(3) |
| H12Bc | 0.437994 | 1.274516 | 0.583214 | 0.052* |
| C13 | 0.5941(13) | 1.1729(7) | 0.5391(2) | 0.0550(15) |
| C14Ad | 0.833(3) | 1.1117(19) | 0.5456(5) | 0.074(5) |
| H14Ad | 0.968772 | 1.127614 | 0.527237 | 0.089* |
| C15Ad | 0.878(3) | 1.0277(18) | 0.5785(6) | 0.068(4) |
| H15Ad | 1.044275 | 0.985463 | 0.581677 | 0.082* |
| C14Bd | 0.719(3) | 1.0433(14) | 0.5298(3) | 0.052(3) |
| H14Bd | 0.747108 | 1.017962 | 0.502276 | 0.062* |
| C15Bd | 0.797(3) | 0.9575(14) | 0.5629(4) | 0.058(3) |
| H15Bd | 0.897594 | 0.878624 | 0.559318 | 0.07* |
| N1 | 0.5727(9) | 0.4440(5) | 0.72926(13) | 0.0352(9) |
| O1 | 0.8370(8) | 0.6731(4) | 0.70836(12) | 0.0467(9) |
| Cl1 | 0.5200(5) | 1.2815(2) | 0.49726(7) | 0.0859(6) |
| Br1 | −0.04867(11) | 0.51822(6) | 0.56198(2) | 0.04269(15) |
| I1 | 0.14893(7) | 0.19560(3) | 0.71369(2) | 0.03900(12) |
| H1A | 0.546(13) | 0.363(7) | 0.743(2) | 0.055(18)* |
| H1B | 0.713(13) | 0.491(7) | 0.734(2) | 0.048(18)* |
aOccupancy: 0.626(11), bOccupancy: 0.374(11), cOccupancy: 0.494(6), dOccupancy: 0.506(6).
Source of material
A stirred solution of 1-(2-amino-5-bromo-3-iodophenyl)-3-(4-chlorophenyl)prop-2-en-1-one (4.00 g, 4.00 mmol) in a mixture of methanol (100 mL, 1:1 v/v) and KOH (8 pellets) at 0 °C was reacted in a 100 mL round-bottomed flask. The reaction mixture was treated with H2O2 (20 mL) and stirred overnight at room temperature. The mixture was then quenched with ice-cold water (200 mL) and the product was extracted into chloroform. The combined organic layers were dried over MgSO4, filtered off and concentrated under reduced pressure on a rotary evaporator to afford the (2-amino-5-bromo-3-iodophenyl)(3-(4-chlorophenyl)oxiran-2-yl)methanone (2.10 g, 54%) as a solid; mp. 139–141 °C; νmax(ATR) 3458, 3419, 3316, 1654, 1595, 1542, 1515, 1491, 1434, 1188, 1091, 1014, 889, 819, 674, 540, 487, 420 cm−1; 1H NMR (500 MHz, CDCl3) 4.06 (1H, d, J = 2.0 Hz, α-H), 4.16 (1H, d, J = 2.0 Hz, β-H), 7.02 (2H, br s, NH2), 7.09 (2H, d, J = 8.5 Hz, H-2′,6′), 7.18 (2H, d, J = 8.5 Hz, H-3′,5′), 7.86 (1H, d, J = 2.5 Hz, H-4), 7.93 (1H, d, J = 2.0 Hz, H-6); 13C NMR (125 MHz, CDCl3) 58.6, 60.1, 87.7, 107.2, 118.0, 125.7, 128.7, 129.1, 133.6, 134.9, 140.1, 146.7, 191.9; HRMS (ES): found. 476.8628 C15H11BrClINO2+ requires 476.8608.
Experimental details
The intensity of the data was determined on a Bruker Venture D8 Photon CMOS diffractometer with graphite-monochromated MoKα1 radiation at 173 K using an Oxford Cryostream 600 cooler. Data reduction was carried out using the program SAINT+, version 6.02 [1] and empirical absorption corrections were made using SADABS [1]. The structure was solved in the WinGX [2] Suite of programs, using intrinsic phasing through SHELXT [3] and refined using SHELXL-2017 [3]. All C-bound H atoms were placed at idealized positions and refined as riding atoms with isotropic parameters 1.2 times those of their parent atoms. All N-bound H atoms were located in the difference fourier map and their coordinates and isotropic thermal parameters allowed to refine freely. The positional disorder of the epoxide group (labelled C8A/C9A/O1A and C8B/C9B/O1B) was resolved by finding alternative positions in the difference Fourier map and their site occupancies refined to 0.626(11) and 0.374(11). The positional disorder of the phenyl ring (labelled C11A/C12A/C14A/C15A and C11B/C12B/C14B/C15B) was resolved by finding alternative positions in the difference Fourier map and their site occupancies refined to 0.494(6) and 0.506(6). Diagrams and publication material were generated using ORTEP-3 [2], and PLATON [4].
Comment
Aminochalcone epoxides are important substrates for the synthesis of a wide range of natural products and biologically active molecules [5], [6], [7]. Epoxychalcones have been found to be biosynthetic intermediates for the rapid construction of complex polycyclic natural products such as flavonoids and their azaflavonoid analogues [8]. The reactivity of the oxirane ring results from its angle strain, which makes the carbon–oxygen bond weaker and more reactive towards nucleophiles than that of ethers [9]. The Weitz–Scheffer reaction, which makes use of hydrogen peroxide under alkaline conditions represents the most efficient method for the oxidation of the α,β-unsaturated ketones into α-epoxyketones [10]. This reaction in the case of 2-aminochalcones is stereospecific and occurs via syn-addition with retention of the trans-stereochemistry of the parent chalcone. The 2-aminochalcone epoxides exist exclusively in solution and solid state in trans geometry with strong intramolecular hydrogen bonding interaction between the amino and carbonyl groups [11]. The previously prepared 1-(2-amino-5-bromo-3-iodophenyl)-3-(4-chlorophenyl)prop-2-en-1-one [12] was subjected to hydrogen peroxide in the presence of aqueous sodium hydroxide in methanol at room temperature for 12 h to obtain the title compound.
Both the ketoaryl and aryl groups are in trans orientation relative to each other about the oxirane ring (O2a—C8a—C9a) with a dihedral angle C1—C8a—C9a—C10 = −155.1(7)° (see the figure). In the asymmetric unit, there is hydrogen bonding interaction involving the amino hydrogen atom as a donor and carbonyl oxygen atom as the hydrogen bond acceptor [N(1)—H(1A)⋯O(1) = 2.46(7) Å and N(1)—H(1B)⋯O(1) = 2.06 Å] classified using a R11(6) ring graph set descriptor. Both NH2 hydrogens are involved in the hydrogen bonding. The examination of the short contacts in the crystal structure shows that the molecules are aligned in parallel planes and held together via multiple weak C—H⋯O and C—H⋯N interactions and further stabilized via π⋯π stacking.
Acknowledgements
We are grateful to the University of South Africa and the National Research Foundation (NRF, SA) for financial assistance. The authors also thank Prof A. Lemmerer of University of the Witwatersrand for X-ray diffraction data using the single-crystal diffractometer purchased through the NRF Equipment Programme (UID:78572).
References
1. Bruker. APEX-3, SAINT+, Version 6.02 (Includes XPREP and SADABS). Bruker AXS Inc., Madison, WI, USA (2016).Suche in Google Scholar
2. Farrugia, L. J.: WinGX and ORTEP for Windows: an update. J. Appl. Crystallogr. 45 (2012) 849–854.10.1107/S0021889812029111Suche in Google Scholar
3. Sheldrick, G. M.: Crystal structure refinement with SHELXL. Acta Crystallogr. C71 (2015) 3–8.10.1107/S2053229614024218Suche in Google Scholar
4. Spek, A. L.: Structure validation in chemical crystallography. Acta Crystallogr. D65 (2009) 148–155.10.1107/S090744490804362XSuche in Google Scholar
5. Gong, J.; Huang, K.; Wang, F.; Yang, L.; Feng, Y.; Li, H.; Li, X.; Zeng, S.; Wu, X.; Stöckigt, J.; Zhao, Y.; Qu, J.: Preparation of two sets of 5,6,7-trioxygenated dihydroflavonol derivatives as free radical scavengers and neuronal cell protectors to oxidative damage. Bioorg. Med. Chem. 17 (2009) 3414–3425.10.1016/j.bmc.2009.03.032Suche in Google Scholar
6. Praveen, C.; Parthasarathy, K.; Kumar, P. S.; Perumal, P. T.: Azaisoflavones: synthesis, antimicrobial evaluation and binding affinity with DNA gyrase. Indian J. Chem. 54B (2015) 373–382.Suche in Google Scholar
7. Stellenboom, N.: Comparison of the inhibitory potential towards carbonic anhydrase, acetylcholinesterase and butyrylcholinesterase of chalcone and chalcone epoxide. J. Biochem. Mol. Toxicol. 33 (2009) e22240.10.1002/jbt.22240Suche in Google Scholar
8. Farooq, S.; Ngaini, Z.: One pot and two pot synthetic strategies and biological applications of epoxy-chalcones. Chem. Africa 3 (2020) 291–302.10.1007/s42250-020-00128-5Suche in Google Scholar
9. Gomes, A. R.; Varela, C. L.; Tavares-da-Silva, E. J.; Roleira, F. M. F.: Epoxide containing molecules: a good or bad drug design approach. Eur. J. Med. Chem. 201 (2020) 112327.10.1016/j.ejmech.2020.112327Suche in Google Scholar
10. Pillai, U. R.; Sahle-Demessie, E.; Varma, R. S.: Microwave-expedited olefin epoxidation over hydrotalcites using hydrogen peroxide and acetonitrile. Tetrahedron Lett. 43 (2002) 2909–2911.10.1016/S0040-4039(02)00426-4Suche in Google Scholar
11. Mphahlele, M. J.; Maluleka, M. M.; Mampa, R. M.: Elucidation of the structure of the 2-amino-3,5-dibromochalcone epoxides in solution and solid state. Crystals 9 (2019) 277–290.10.3390/cryst9060277Suche in Google Scholar
12. Mphahlele, M. J.; Maluleka, M. M.: Trifluoroacetylation of indole-chalcones derived from the 2-amino-3-(arylethynyl)-5-bromo-iodochalcones. J. Fluorine Chem. 189 (2016) 88–95.10.1016/j.jfluchem.2016.07.022Suche in Google Scholar
©2020 Marole M. Maluleka et al., published by De Gruyter, Berlin/Boston
This work is licensed under the Creative Commons Attribution 4.0 International License.
Artikel in diesem Heft
- Frontmatter
- Crystal structure of poly[tetraaqua-bis(μ4-5-(4-carboxy-benzylamino)-isophthalato-κ4O,O′:O′′:O′′′)-(μ2-4,4′-di(1H-imidazol-1-yl)-1,1′-biphenyl-κ2N:N′)dicadmium(II)], C25H22N3O8Cd
- The crystal structure of 2-(2-(2,3,4,9-tetrahydro-1H-pyrido[3,4-b]indol-2-ium-1-yl)phenoxy)acetate, C19H18N2O3
- Crystal structure of poly[aqua-μ2-4,4′-bipyridine-κ2N:N′)-μ2-bis(2-(2-((2,6-dichlorophenyl)amino)phenyl)acetato-κ2O,O′)zinc(II)], C38H28Cl4N4O4Zn
- Crystal structure of 1-(2-(1H-indol-3-yl)ethyl)-4-benzyl-3-hydroxy-3,6-diphenylpiperazine-2,5-dione, C33H29N3O3
- The crystal structure 2,2′-bipyridine-κ2N,N′-(2-(3-amino-4-chlorobenzoyl)benzoato-κ1O)-(2-(3-amino-4-chlorobenzoyl)benzoato-κ2O,O′)zinc(II) — ethanol (1/1), C40H32Cl2N4O7Zn
- Crystal structure of catena-poly[(μ3-2-carboxy-4-(3-carboxy-5-carboxylatophenoxy)benzoato-κ3O:O′:O′′)-bis(μ2-4,4′-bis(pyrid-4-yl)biphenyl-k1N)copper(II)], C60H40N4O9Cu
- The crystal structure of dimethylammonium catena-[di(μ-aqua)-bis(μ9-benzene-1,3,5-tricarboxylato)pentalithium], C20H16Li5NO13
- Crystal structure of tetraaqua-bis(3,5-di(pyridin-4-yl)-1,2,4-triazol-1-ido-κ1N)nickel(II) dihydrate, C24H28O6N10Ni
- The crystal structure of tetrakis(1-methylimidazole-κ1N)-oxido-(sulfato-κ1O)vanadium(IV), C16H24N8O5SV
- Crystal structure of methyl 2-(6,11-dioxo-2,3,6,11-tetrahydro-1H-benzo[f]pyrrolo[2,1-a]isoindole-5-carbonyl)benzoate, C24H17NO5
- Crystal structure of (E)-N′-(2-hydroxy-4-(2-(piperidin-1-yl)ethoxy)benzylidene) nicotinohydrazide monohydrate, C20H24N4O3 ⋅ H2O
- Crystal structure of poly[bis(μ3-(1-(3,5-di(1H-imidazol-1-yl)phenyl)-1H-imidazole-κ3N:N′:N′′)cobalt(II)] dinitrate — N,N-dimethylformamide (1/4), C42H52N18O10Co
- The crystal structure bis{hexakis(1-methyl-1H-imidazole-κ1N)cobalt(II)} tetrakis(μ3-oxido)-octakis(μ2-oxido)-tetradecaoxido-octamolybdate(VI), C24H36CoMo4N12O13
- Crystal structure of di-μ-nicotinato-κ2N:O; κ2O:N-bis-[aqua-bis(benzyl)(nicotinato-κ2O,O′)tin(IV)], C52H48N4O10Sn2
- Crystal structure of dichlorido-bis[2-(2-(3-(pyridin-2-yl)-1H-1,2,4-triazol-5-yl)phenoxy)benzoic acidmanganese(II) monohydrate, C40H30N8O7MnCl2
- The crystal structure of benzyl 3β-acetylglycyrrhetate, C39H54O5
- Synthesis and crystal structure of (E)-1-benzyl-3-(4-methoxystyryl)quinoxalin-2(1H)-one, C24H20N2O2
- Crystal structure of trans-dichloridobis(4-chlorophenyl-κC1)(1,10-phenanthroline-κ2N,N′)tin(IV) dimethylsulphoxide solvate, C26H22Cl4N2OSSn
- Crystal structure of phenyl(1,3,4a-triphenyl-4a,5,6,10b-tetrahydro-1H-[1,4]oxazino[2,3-c]quinolin-5-yl)methanone, C36H28N2O2
- Crystal structure of (4aS,5S,6aS,6a1S, 10aS)-4a,5,6a,6a1,9,10-hexahydro-7H-4,5-methanocyclobuta[4,5]naphtho[8a,1-b]pyran-6(2H)-one, C15H16O2
- Crystal structure of [(Z)-O-isopropyl N-(4-chlorophenyl)thiocarbamato-κS]-(triphenylphosphine-κP)-gold(I), C28H26AuClNOPS
- Crystal structure of (μ2-1,1′-bis(diphenylphosphino)ferrocene-P,P′)-bis[(Z)-O-isopropyl N-(4-chlorophenyl)thiocarbamato-S]-di-gold(I) acetonitrile di-solvate, C54H50Au2Cl2FeN2O2P2S2⋅2(C2H3N)
- Crystal structure of (6aR,6a1S,10aS)-2,4a,6a,6a1,9,10-hexahydro-7H-4,5-methanocyclobuta[4,5]naphtho[8a,1-b]pyran, C15H16O
- Crystal structure of 5,17-diformyl-25,26,27,28-tetrahydroxycalix[4]arene- dichloromethane, C31H26Cl2O6
- Crystal structure of 2-tert-butyl 1-methyl 5-{4-[(methoxycarbonyl)amino]phenyl}-2,5-dihydro-1H-pyrrole-1,2-dicarboxylate, C19H24N2O6
- Crystal structure of [2-carboxybenzene-1-thiolato-S]-(triethylphosphane-P)-gold(I), C13H20AuO2PS
- Synthesis and crystal structure of bis(5-methyl-2-aldehyde-phenolato-κ2O1,O2)copper(II), C16H14CuO4
- Crystal structure of poly[triaqua-(di(2,2′-bipyridine-κ2N,N′)-μ4-silanetetrayltetrakis(benzene-4,1-diyl)tetrakis (hydrogen phosphonato)-κ4O:O′:O′′:O′′′) dicadmium(II)], C44H42N4O15P4Cd2Si
- Crystal structure of bis[μ2-(N,N-diethylcarbamodithioato-κS:κS,κS′)]-bis(triethylphosphine-P)-di-silver(I), C22H50Ag2N2P2S4
- Crystal structure of bis[μ2-(pyrrolidine-1-carbodithioato-κS:κS,κS′)]-bis(triethylphosphine-κP)disilver(I), C22H46Ag2N2P2S4
- Crystal structure of bis[μ2-(N-(2-hydroxyethyl)-N-methylcarbamodithioato-κS:κS,κS′)]-bis(triethylphosphine-P)-di-silver(I), C20H46Ag2N2O2P2S4
- The crystal structure of (2E,2′E)-,2,2′-bis[1-(2-pyrazinyl)ethylidene]carbonimidic dihydrazide, C13H15N9
- The crystal structure of (E)-1-(quinolin-2-ylmethyl)-2-((1-(quinolin-2-ylmethyl)pyridin-2(1H)-ylidene)amino)pyridin-1-ium, C30H25BrN5
- Crystal structure of catena-poly[(μ2-1-((benzotriazol-1-yl)methyl)-1H-1,3-imdazole-κ2N:N′)-(1-((benzotriazol-1-yl)methyl)-1H-1,3-imdazole-κ1N)-(methanol-κ1O)mercury(II)] dinitrate, C21H22N12O7Hg
- Crystal structure of 1-(6-hydroxy-2-phenylbenzofuran-5-yl)ethan-1-one, C16H12O3
- The crystal structure of oxonium hexaquaaluminium disulfate hexahydrate
- Crystal structure of catena{(μ2-1,10-phenanthroline-κ4N,N,N′,N′)-(μ2-1,10-phenanthroline-κ3N,N,N′)potassium(I) {[bis(2-hydroxyethyl)iminiumyl](sulfanidyl)methyl}sulfanide hemi(1,10-phenanthroline)}, {C24H16KN4, 0.5(C12H8N2), C5H10NO2S2}
- Crystal structure of chlorido-[(N,N-di-isobutyl)dithiocarbamato-κ2S,S′]-di(4-methylbenzyl-κC)tin(IV), C25H36ClNS2Sn
- Crystal structure of chlorido-(η5-pentamethylcyclopentadienyl)-(4-chloro-4-pyridyl-2,2′:6′,2′′-terpyridine-κ2N,N′) rhodium(III) hexaflourophosphate, C31H29Cl2F6N3PRh
- The crystal structure of catena-poly[bis-(3,5-dinitro-1,2,4-triazolato-κ2N:O)-(μ2-1,4-bis(1-imidazolyl)benzene-κ2N:N′)copper(II)], C16H10CuN14O8
- Crystal structure of poly[triaqua-bis(μ3-3,3′-((5-carboxylato-1,3-phenylene)bis(oxy))dibenzoato)-tris(1,10-phenanthroline)cobalt(II)], C78H46N6O20Co3
- The crystal structure of 2,4-dihydroxybenzoic acid–nicotinamide–methanol (1/1/1), C15H18N2O6
- The crystal structure of aqua{N,N,N′,N′-tetrakis[(1H-benzimidazol-κN3) methyl]cyclohexane-1,2-diamine}lead(II) diacetate–methanol (1/2), C44H54N10O7Pb
- Crystal structure of (2-amino-5-bromo-3-iodophenyl)(3-(4-chlorophenyl)oxiran-2-yl)methanone, C15H10BrClINO2
- Synthesis and crystal structure of 3-octyl-5,5-diphenylimidazolidine-2,4-dione, C23H28N2O2
- Synthesis and crystal structure of 2-azido-N-(4-nitrophenyl)acetamide, C8H7N5O3
- Crystal structure of tert-butyl (1S,2R,5R)-2-(hydroxymethyl)-4-(4-methoxyphenyl)-6-oxa-3-azabicyclo[3.1.0]hexane-3-carboxylate, C17H23NO5
- Crystal structure of 4-[(4-methoxy-2-nitrophenyl)carbamoyl]butanoic acid, C12H14N2O6
- Crystal structure of 3-ethyl-1-[(E)-[(2E)-3-phenylprop-2-en-1-ylidene]amino]thiourea, C12H15N3S
- Crystal structure of 4,4′-bipyridin-1,1′-dium poly[bis(μ4-benzene-1,3,5-triyltris(hydrogen phosphonato-κ4O:O′:O′′:O′′′))zinc(II)], C11H11NO9P3Zn
- Crystal structure of (μ2-1,1′-bis(diphenylphosphino)butane-κ2P,P′)-bis[(Z)-N-(3-fluorophenyl)-O-methylthiocarbamato-κS]-di-gold(I), C44H42Au2F2N2O2P2S2
- Crystal structure of (μ2-1,1′-bis(diphenylphosphino)hexane-κ2P,P′)-bis[(Z)-N-(3-fluorophenyl)-O-methylthiocarbamato-κS]digold(I), C46H46Au2F2N2O2P2S2
- Crystal structure of tetrakis (N-(2-hydroxyethyl)-N-isopropylcarbamodithioato-κS,S′)-(μ2(2-(pyridin-4-yl)vinyl)pyridine-κN,N′)dicadmium(II), C36H58Cd2N6O4S8
- Crystal structure of 4-(2-(benzo[b]thiophen-2-yl)-3,3,4,4,5,5-hexafluorocyclopent-1-en-1-yl)-1,5-dimethyl-1H-pyrrole-2-carbonitrile, C20H12F6N2S
- Crystal structure of bis(octahydrocyclopenta[c]pyrrolium)pentachlorobismuthate(III), (C7NH14)2BiCl5
- The crystal structure of diaqua-tris(nitrato-κ2O,O′)-bis(4,4,5,5-tetramethyl-2-(p-pyridyl)imidazoline-1-oxyl 3-oxide-κN)samarium(III), C24H36N9O15Sm
- Synthesis and crystal structure of methyl 2-(2-((tert-butoxycarbonyl)amino)phenyl)-2-(4-oxo-4H-chromen-3-yl)acetate, C23H23NO6
- Crystal structure of O-hexyl benzoylcarbamothioate, C14H19NO2S
- Crystal structure of chlorido-(O-methyl phenylcarbamothioamide-κS)-bis(triphenylphosphane-κP)silver(I), C44H39AgClNOP2S
- Crystal structure of chlorido-(O-ethyl phenylcarbamothioamide-κS)-bis(triphenylphosphane-κP)-silver(I), C45H41AgClNOP2S
- Crystal structure of 4-[(2-methoxyphenyl)carbamoyl]butanoic acid, C12H15NO4
- Crystal structure of ethyl 4-methyl-2-oxo-5-phenyl-1,3,4-oxadiazinane-3-carboxylate, C13H16N2O4
- Crystal structure of catena-poly[diaqua(μ2-2-(hydroxymethyl)-1H-imidazole-4,5-dicarboxylato)cadmium(II)], C6H8CdN2O7
- Crystal structure of (1S)-N-(chloromethyl)-1-((4S,6aR,8aS, 8bR,9aR)-4-methoxy-6a,8a-dimethyl-1,3,4, 5,6,6a,6b,7,8,8a,9a,10,10a,10b-tetradecahydro-8bH-naphtho[2′,1′:4,5] indeno[1,2-b]oxiren-8b-yl)-N-methylethan-1-amine, C24H46ClNO5
- Crystal structure of 4-[(3,5-dichlorophenyl)carbamoyl]butanoic acid, C11H11Cl2NO3
- Crystal structure of (2Z)-2-amino-3-[(E)-[(2,4-dihydroxyphenyl)methylidene]-amino]but-2-enedinitrile, C11H8N4O2
- Crystal structure of 3-methyl-1-[(E)-(4-phenylbutan-2-ylidene)amino]thiourea, C12H17N3S
- Crystal structure of carbonyl{hydridotris[3-phenyl-5-methylpyrazol-1-yl]borato-κ3N,N′N′′}copper(I), C31H28BCuN6O
- Crystal structure of ethane-1,2-diylbis(diphenylphosphine oxide) – dihydrogenperoxide (1/2), C26H28O6P2
- Crystal structure of 2-(pyridin-2-ylamino)pyridinium chloride dibenzyldichlorostannane, [C10H10N3]Cl, C14H14Cl2Sn
- Crystal structure of 4-[(3-methoxyphenyl)carbamoyl]butanoic acid, C12H15NO4
- Crystal structure of dichlorido-bis(tri-4-tolylphosphane oxide-κO)-di(4-chlorophenyl-κC)tin(IV), C54H50Cl4O2P2Sn
- Crystal structure of dichloridodimethylbis(tri-4-tolylphosphane oxide-κO)-tin(IV), C44H48Cl2O2P2Sn
- Crystal structure of chlorido(2-methylquinolin-8-olato-κ2N,O)-bis(4-tolyl-κC)tin(IV), C24H22ClNOSn
- Crystal structure of (E)-dichloro(1-chloro-3-methoxyprop-1-en-2-yl)(4-methoxyphenyl)-λ4-tellane, C11H13Cl3O2Te
- Crystal structure of bis{N-methyl-N′-[3-(4-methoxyphenyl)-1-methylpropane-1-ylidene]carbamohydrazonothioato}zinc(II), C26H36N6O2S2Zn
- Crystal structure of (2-carboxy-4-(3-carboxy-5-carboxylatophenoxy)benzoato-κ2O,O′)bis(1,10-phenantroline-κ2N,N′)cobalt(II), C40H24N4O9Co
- The crystal structure of (3S,8R,10R,14R)-17-((2S,5S)-5-(2-hydroxypropan-2-yl)-2-methyltetrahydrofuran-2-yl)-4,4,8,10,14-pentamethyl-12-oxohexadecahydro-1H-cyclopenta[a]phenanthren-3-yl acetate, C32H52O5
- Crystal structure of (μ2-1,1′-bis(diphenylphosphino)ferrocene-κ2P,P′)-bis[(Z)N-(3-fluorophenyl)-O-methylthiocarbamato-S]digold(I) chloroform solvate, C50H42Au2F2FeN2O2P2S2, CHCl3
- Crystal structure of poly[bis(μ2-1,4-di(1H-imidazol-1-yl)benzene-κ2N:N′)-(μ2-tetraoxidomolybdato(VI)-κ2O:O′)cobalt(II)], C24H20N8O4MoCo
Artikel in diesem Heft
- Frontmatter
- Crystal structure of poly[tetraaqua-bis(μ4-5-(4-carboxy-benzylamino)-isophthalato-κ4O,O′:O′′:O′′′)-(μ2-4,4′-di(1H-imidazol-1-yl)-1,1′-biphenyl-κ2N:N′)dicadmium(II)], C25H22N3O8Cd
- The crystal structure of 2-(2-(2,3,4,9-tetrahydro-1H-pyrido[3,4-b]indol-2-ium-1-yl)phenoxy)acetate, C19H18N2O3
- Crystal structure of poly[aqua-μ2-4,4′-bipyridine-κ2N:N′)-μ2-bis(2-(2-((2,6-dichlorophenyl)amino)phenyl)acetato-κ2O,O′)zinc(II)], C38H28Cl4N4O4Zn
- Crystal structure of 1-(2-(1H-indol-3-yl)ethyl)-4-benzyl-3-hydroxy-3,6-diphenylpiperazine-2,5-dione, C33H29N3O3
- The crystal structure 2,2′-bipyridine-κ2N,N′-(2-(3-amino-4-chlorobenzoyl)benzoato-κ1O)-(2-(3-amino-4-chlorobenzoyl)benzoato-κ2O,O′)zinc(II) — ethanol (1/1), C40H32Cl2N4O7Zn
- Crystal structure of catena-poly[(μ3-2-carboxy-4-(3-carboxy-5-carboxylatophenoxy)benzoato-κ3O:O′:O′′)-bis(μ2-4,4′-bis(pyrid-4-yl)biphenyl-k1N)copper(II)], C60H40N4O9Cu
- The crystal structure of dimethylammonium catena-[di(μ-aqua)-bis(μ9-benzene-1,3,5-tricarboxylato)pentalithium], C20H16Li5NO13
- Crystal structure of tetraaqua-bis(3,5-di(pyridin-4-yl)-1,2,4-triazol-1-ido-κ1N)nickel(II) dihydrate, C24H28O6N10Ni
- The crystal structure of tetrakis(1-methylimidazole-κ1N)-oxido-(sulfato-κ1O)vanadium(IV), C16H24N8O5SV
- Crystal structure of methyl 2-(6,11-dioxo-2,3,6,11-tetrahydro-1H-benzo[f]pyrrolo[2,1-a]isoindole-5-carbonyl)benzoate, C24H17NO5
- Crystal structure of (E)-N′-(2-hydroxy-4-(2-(piperidin-1-yl)ethoxy)benzylidene) nicotinohydrazide monohydrate, C20H24N4O3 ⋅ H2O
- Crystal structure of poly[bis(μ3-(1-(3,5-di(1H-imidazol-1-yl)phenyl)-1H-imidazole-κ3N:N′:N′′)cobalt(II)] dinitrate — N,N-dimethylformamide (1/4), C42H52N18O10Co
- The crystal structure bis{hexakis(1-methyl-1H-imidazole-κ1N)cobalt(II)} tetrakis(μ3-oxido)-octakis(μ2-oxido)-tetradecaoxido-octamolybdate(VI), C24H36CoMo4N12O13
- Crystal structure of di-μ-nicotinato-κ2N:O; κ2O:N-bis-[aqua-bis(benzyl)(nicotinato-κ2O,O′)tin(IV)], C52H48N4O10Sn2
- Crystal structure of dichlorido-bis[2-(2-(3-(pyridin-2-yl)-1H-1,2,4-triazol-5-yl)phenoxy)benzoic acidmanganese(II) monohydrate, C40H30N8O7MnCl2
- The crystal structure of benzyl 3β-acetylglycyrrhetate, C39H54O5
- Synthesis and crystal structure of (E)-1-benzyl-3-(4-methoxystyryl)quinoxalin-2(1H)-one, C24H20N2O2
- Crystal structure of trans-dichloridobis(4-chlorophenyl-κC1)(1,10-phenanthroline-κ2N,N′)tin(IV) dimethylsulphoxide solvate, C26H22Cl4N2OSSn
- Crystal structure of phenyl(1,3,4a-triphenyl-4a,5,6,10b-tetrahydro-1H-[1,4]oxazino[2,3-c]quinolin-5-yl)methanone, C36H28N2O2
- Crystal structure of (4aS,5S,6aS,6a1S, 10aS)-4a,5,6a,6a1,9,10-hexahydro-7H-4,5-methanocyclobuta[4,5]naphtho[8a,1-b]pyran-6(2H)-one, C15H16O2
- Crystal structure of [(Z)-O-isopropyl N-(4-chlorophenyl)thiocarbamato-κS]-(triphenylphosphine-κP)-gold(I), C28H26AuClNOPS
- Crystal structure of (μ2-1,1′-bis(diphenylphosphino)ferrocene-P,P′)-bis[(Z)-O-isopropyl N-(4-chlorophenyl)thiocarbamato-S]-di-gold(I) acetonitrile di-solvate, C54H50Au2Cl2FeN2O2P2S2⋅2(C2H3N)
- Crystal structure of (6aR,6a1S,10aS)-2,4a,6a,6a1,9,10-hexahydro-7H-4,5-methanocyclobuta[4,5]naphtho[8a,1-b]pyran, C15H16O
- Crystal structure of 5,17-diformyl-25,26,27,28-tetrahydroxycalix[4]arene- dichloromethane, C31H26Cl2O6
- Crystal structure of 2-tert-butyl 1-methyl 5-{4-[(methoxycarbonyl)amino]phenyl}-2,5-dihydro-1H-pyrrole-1,2-dicarboxylate, C19H24N2O6
- Crystal structure of [2-carboxybenzene-1-thiolato-S]-(triethylphosphane-P)-gold(I), C13H20AuO2PS
- Synthesis and crystal structure of bis(5-methyl-2-aldehyde-phenolato-κ2O1,O2)copper(II), C16H14CuO4
- Crystal structure of poly[triaqua-(di(2,2′-bipyridine-κ2N,N′)-μ4-silanetetrayltetrakis(benzene-4,1-diyl)tetrakis (hydrogen phosphonato)-κ4O:O′:O′′:O′′′) dicadmium(II)], C44H42N4O15P4Cd2Si
- Crystal structure of bis[μ2-(N,N-diethylcarbamodithioato-κS:κS,κS′)]-bis(triethylphosphine-P)-di-silver(I), C22H50Ag2N2P2S4
- Crystal structure of bis[μ2-(pyrrolidine-1-carbodithioato-κS:κS,κS′)]-bis(triethylphosphine-κP)disilver(I), C22H46Ag2N2P2S4
- Crystal structure of bis[μ2-(N-(2-hydroxyethyl)-N-methylcarbamodithioato-κS:κS,κS′)]-bis(triethylphosphine-P)-di-silver(I), C20H46Ag2N2O2P2S4
- The crystal structure of (2E,2′E)-,2,2′-bis[1-(2-pyrazinyl)ethylidene]carbonimidic dihydrazide, C13H15N9
- The crystal structure of (E)-1-(quinolin-2-ylmethyl)-2-((1-(quinolin-2-ylmethyl)pyridin-2(1H)-ylidene)amino)pyridin-1-ium, C30H25BrN5
- Crystal structure of catena-poly[(μ2-1-((benzotriazol-1-yl)methyl)-1H-1,3-imdazole-κ2N:N′)-(1-((benzotriazol-1-yl)methyl)-1H-1,3-imdazole-κ1N)-(methanol-κ1O)mercury(II)] dinitrate, C21H22N12O7Hg
- Crystal structure of 1-(6-hydroxy-2-phenylbenzofuran-5-yl)ethan-1-one, C16H12O3
- The crystal structure of oxonium hexaquaaluminium disulfate hexahydrate
- Crystal structure of catena{(μ2-1,10-phenanthroline-κ4N,N,N′,N′)-(μ2-1,10-phenanthroline-κ3N,N,N′)potassium(I) {[bis(2-hydroxyethyl)iminiumyl](sulfanidyl)methyl}sulfanide hemi(1,10-phenanthroline)}, {C24H16KN4, 0.5(C12H8N2), C5H10NO2S2}
- Crystal structure of chlorido-[(N,N-di-isobutyl)dithiocarbamato-κ2S,S′]-di(4-methylbenzyl-κC)tin(IV), C25H36ClNS2Sn
- Crystal structure of chlorido-(η5-pentamethylcyclopentadienyl)-(4-chloro-4-pyridyl-2,2′:6′,2′′-terpyridine-κ2N,N′) rhodium(III) hexaflourophosphate, C31H29Cl2F6N3PRh
- The crystal structure of catena-poly[bis-(3,5-dinitro-1,2,4-triazolato-κ2N:O)-(μ2-1,4-bis(1-imidazolyl)benzene-κ2N:N′)copper(II)], C16H10CuN14O8
- Crystal structure of poly[triaqua-bis(μ3-3,3′-((5-carboxylato-1,3-phenylene)bis(oxy))dibenzoato)-tris(1,10-phenanthroline)cobalt(II)], C78H46N6O20Co3
- The crystal structure of 2,4-dihydroxybenzoic acid–nicotinamide–methanol (1/1/1), C15H18N2O6
- The crystal structure of aqua{N,N,N′,N′-tetrakis[(1H-benzimidazol-κN3) methyl]cyclohexane-1,2-diamine}lead(II) diacetate–methanol (1/2), C44H54N10O7Pb
- Crystal structure of (2-amino-5-bromo-3-iodophenyl)(3-(4-chlorophenyl)oxiran-2-yl)methanone, C15H10BrClINO2
- Synthesis and crystal structure of 3-octyl-5,5-diphenylimidazolidine-2,4-dione, C23H28N2O2
- Synthesis and crystal structure of 2-azido-N-(4-nitrophenyl)acetamide, C8H7N5O3
- Crystal structure of tert-butyl (1S,2R,5R)-2-(hydroxymethyl)-4-(4-methoxyphenyl)-6-oxa-3-azabicyclo[3.1.0]hexane-3-carboxylate, C17H23NO5
- Crystal structure of 4-[(4-methoxy-2-nitrophenyl)carbamoyl]butanoic acid, C12H14N2O6
- Crystal structure of 3-ethyl-1-[(E)-[(2E)-3-phenylprop-2-en-1-ylidene]amino]thiourea, C12H15N3S
- Crystal structure of 4,4′-bipyridin-1,1′-dium poly[bis(μ4-benzene-1,3,5-triyltris(hydrogen phosphonato-κ4O:O′:O′′:O′′′))zinc(II)], C11H11NO9P3Zn
- Crystal structure of (μ2-1,1′-bis(diphenylphosphino)butane-κ2P,P′)-bis[(Z)-N-(3-fluorophenyl)-O-methylthiocarbamato-κS]-di-gold(I), C44H42Au2F2N2O2P2S2
- Crystal structure of (μ2-1,1′-bis(diphenylphosphino)hexane-κ2P,P′)-bis[(Z)-N-(3-fluorophenyl)-O-methylthiocarbamato-κS]digold(I), C46H46Au2F2N2O2P2S2
- Crystal structure of tetrakis (N-(2-hydroxyethyl)-N-isopropylcarbamodithioato-κS,S′)-(μ2(2-(pyridin-4-yl)vinyl)pyridine-κN,N′)dicadmium(II), C36H58Cd2N6O4S8
- Crystal structure of 4-(2-(benzo[b]thiophen-2-yl)-3,3,4,4,5,5-hexafluorocyclopent-1-en-1-yl)-1,5-dimethyl-1H-pyrrole-2-carbonitrile, C20H12F6N2S
- Crystal structure of bis(octahydrocyclopenta[c]pyrrolium)pentachlorobismuthate(III), (C7NH14)2BiCl5
- The crystal structure of diaqua-tris(nitrato-κ2O,O′)-bis(4,4,5,5-tetramethyl-2-(p-pyridyl)imidazoline-1-oxyl 3-oxide-κN)samarium(III), C24H36N9O15Sm
- Synthesis and crystal structure of methyl 2-(2-((tert-butoxycarbonyl)amino)phenyl)-2-(4-oxo-4H-chromen-3-yl)acetate, C23H23NO6
- Crystal structure of O-hexyl benzoylcarbamothioate, C14H19NO2S
- Crystal structure of chlorido-(O-methyl phenylcarbamothioamide-κS)-bis(triphenylphosphane-κP)silver(I), C44H39AgClNOP2S
- Crystal structure of chlorido-(O-ethyl phenylcarbamothioamide-κS)-bis(triphenylphosphane-κP)-silver(I), C45H41AgClNOP2S
- Crystal structure of 4-[(2-methoxyphenyl)carbamoyl]butanoic acid, C12H15NO4
- Crystal structure of ethyl 4-methyl-2-oxo-5-phenyl-1,3,4-oxadiazinane-3-carboxylate, C13H16N2O4
- Crystal structure of catena-poly[diaqua(μ2-2-(hydroxymethyl)-1H-imidazole-4,5-dicarboxylato)cadmium(II)], C6H8CdN2O7
- Crystal structure of (1S)-N-(chloromethyl)-1-((4S,6aR,8aS, 8bR,9aR)-4-methoxy-6a,8a-dimethyl-1,3,4, 5,6,6a,6b,7,8,8a,9a,10,10a,10b-tetradecahydro-8bH-naphtho[2′,1′:4,5] indeno[1,2-b]oxiren-8b-yl)-N-methylethan-1-amine, C24H46ClNO5
- Crystal structure of 4-[(3,5-dichlorophenyl)carbamoyl]butanoic acid, C11H11Cl2NO3
- Crystal structure of (2Z)-2-amino-3-[(E)-[(2,4-dihydroxyphenyl)methylidene]-amino]but-2-enedinitrile, C11H8N4O2
- Crystal structure of 3-methyl-1-[(E)-(4-phenylbutan-2-ylidene)amino]thiourea, C12H17N3S
- Crystal structure of carbonyl{hydridotris[3-phenyl-5-methylpyrazol-1-yl]borato-κ3N,N′N′′}copper(I), C31H28BCuN6O
- Crystal structure of ethane-1,2-diylbis(diphenylphosphine oxide) – dihydrogenperoxide (1/2), C26H28O6P2
- Crystal structure of 2-(pyridin-2-ylamino)pyridinium chloride dibenzyldichlorostannane, [C10H10N3]Cl, C14H14Cl2Sn
- Crystal structure of 4-[(3-methoxyphenyl)carbamoyl]butanoic acid, C12H15NO4
- Crystal structure of dichlorido-bis(tri-4-tolylphosphane oxide-κO)-di(4-chlorophenyl-κC)tin(IV), C54H50Cl4O2P2Sn
- Crystal structure of dichloridodimethylbis(tri-4-tolylphosphane oxide-κO)-tin(IV), C44H48Cl2O2P2Sn
- Crystal structure of chlorido(2-methylquinolin-8-olato-κ2N,O)-bis(4-tolyl-κC)tin(IV), C24H22ClNOSn
- Crystal structure of (E)-dichloro(1-chloro-3-methoxyprop-1-en-2-yl)(4-methoxyphenyl)-λ4-tellane, C11H13Cl3O2Te
- Crystal structure of bis{N-methyl-N′-[3-(4-methoxyphenyl)-1-methylpropane-1-ylidene]carbamohydrazonothioato}zinc(II), C26H36N6O2S2Zn
- Crystal structure of (2-carboxy-4-(3-carboxy-5-carboxylatophenoxy)benzoato-κ2O,O′)bis(1,10-phenantroline-κ2N,N′)cobalt(II), C40H24N4O9Co
- The crystal structure of (3S,8R,10R,14R)-17-((2S,5S)-5-(2-hydroxypropan-2-yl)-2-methyltetrahydrofuran-2-yl)-4,4,8,10,14-pentamethyl-12-oxohexadecahydro-1H-cyclopenta[a]phenanthren-3-yl acetate, C32H52O5
- Crystal structure of (μ2-1,1′-bis(diphenylphosphino)ferrocene-κ2P,P′)-bis[(Z)N-(3-fluorophenyl)-O-methylthiocarbamato-S]digold(I) chloroform solvate, C50H42Au2F2FeN2O2P2S2, CHCl3
- Crystal structure of poly[bis(μ2-1,4-di(1H-imidazol-1-yl)benzene-κ2N:N′)-(μ2-tetraoxidomolybdato(VI)-κ2O:O′)cobalt(II)], C24H20N8O4MoCo


